Advanced Iron Catalyst Technology for Commercial Pyridine Derivative Manufacturing
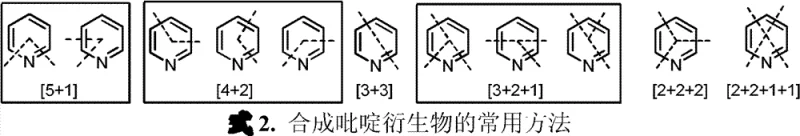
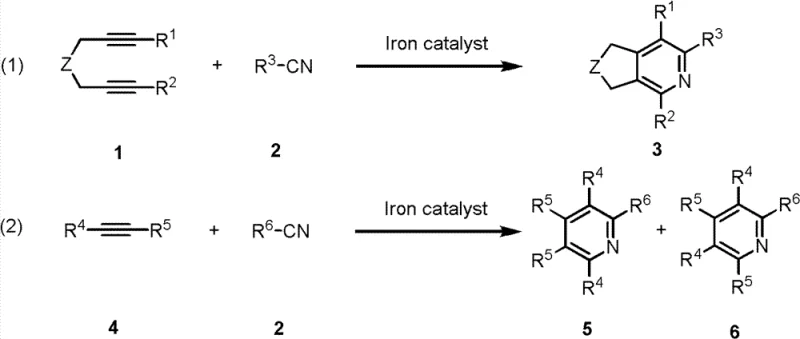
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways for constructing heterocyclic scaffolds, which serve as the backbone for countless active pharmaceutical ingredients. Patent CN102872915A introduces a groundbreaking iron catalyst system designed specifically for the preparation of pyridine derivatives through [2+2+2] cycloaddition reactions. This technology represents a significant departure from traditional methods that rely on expensive and toxic noble metals, offering a robust alternative for the synthesis of complex pyridine structures found in drugs like Atazanavir and Gleevec. By utilizing cheap and readily available iron salts combined with bisphosphine ligands, this innovation addresses critical pain points in modern chemical manufacturing, including cost efficiency, environmental compliance, and supply chain stability. The ability to catalyze the reaction of diynes or monoynes with nitriles under mild conditions opens new avenues for producing high-purity intermediates with exceptional atom economy.
Pyridine derivatives are indispensable building blocks in the synthesis of agrochemicals, pharmaceuticals, and functional materials, yet their construction has historically been plagued by inefficient methodologies. Conventional approaches often require harsh acidic or alkaline conditions that generate substantial inorganic waste and suffer from poor atom utilization. The [2+2+2] cycloaddition strategy highlighted in this patent splits the pyridine ring into three diatomic fragments, achieving up to 100% atom utilization by simultaneously forming two carbon-carbon bonds and one carbon-nitrogen bond without generating waste by-products. This green chemistry approach not only aligns with increasingly stringent environmental regulations but also simplifies the purification process, thereby reducing the overall operational burden on manufacturing facilities. The transition towards such atom-economical processes is essential for companies aiming to reduce their carbon footprint while maintaining high production throughput.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyridine rings has relied heavily on transition metal catalytic systems based on cobalt, rhodium, or ruthenium, which present significant logistical and economic challenges for large-scale production. These noble metal catalysts are not only prohibitively expensive due to their scarcity but also pose severe toxicity risks that necessitate complex and costly removal steps to meet pharmaceutical purity standards. Furthermore, many traditional methods operate under high temperatures or require stoichiometric amounts of acids and bases, leading to the generation of large volumes of hazardous waste that complicate disposal and increase environmental compliance costs. The difficulty in controlling regioselectivity with these older systems often results in mixtures of isomers, requiring energy-intensive separation processes that further erode profit margins and extend lead times for final product delivery.
The Novel Approach
The iron catalyst system described in patent CN102872915A offers a transformative solution by replacing scarce noble metals with abundant, non-toxic iron, fundamentally altering the cost structure of pyridine derivative manufacturing. This novel approach utilizes a combination of iron salts, bisphosphine ligands like dppp or dppe, and reducing agents to achieve high reactivity and specificity under remarkably mild conditions ranging from room temperature to 100°C. The system demonstrates exceptional versatility, capable of catalyzing reactions with various substrates including diynes and monoynes to produce diverse substituted pyridines with high yields. By eliminating the need for expensive metal scavengers and reducing waste generation, this method significantly streamlines the production workflow, making it an ideal candidate for companies seeking to optimize their supply chain resilience and reduce dependency on volatile precious metal markets.
Mechanistic Insights into Iron-Catalyzed [2+2+2] Cycloaddition
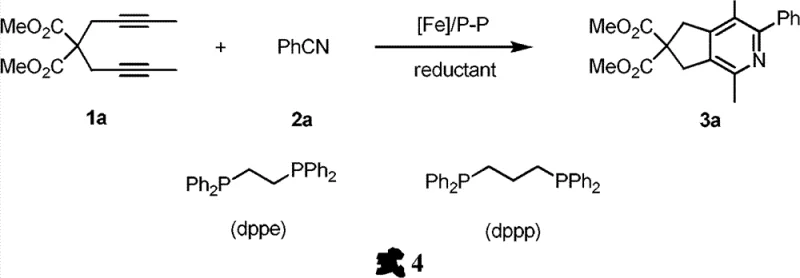
The core of this technological advancement lies in the unique mechanistic pathway enabled by the iron-bisphosphine complex, which facilitates the [2+2+2] cycloaddition with remarkable efficiency and control. The reaction proceeds through the coordination of the alkyne and nitrile substrates to the low-valent iron center generated in situ by the reduction of iron salts, promoting the sequential formation of metallacycle intermediates that eventually collapse to form the aromatic pyridine ring. This mechanism allows for the precise assembly of the heterocyclic core in a single step, bypassing the multi-step sequences typically required in classical heterocyclic chemistry. The use of specific ligands such as 1,3-bis(diphenylphosphine)propane ensures that the catalytic cycle remains stable and active over extended periods, supporting high turnover numbers that are critical for industrial viability.
Regioselectivity is a paramount concern in the synthesis of substituted pyridines, as the position of substituents dictates the biological activity and physical properties of the final pharmaceutical agent. The iron catalyst system exhibits superior control over regioselectivity, particularly in reactions involving monoalkynes, where the ratio of the desired isomer to the undesired byproduct can be maintained at levels greater than 9:1. This high degree of selectivity minimizes the formation of difficult-to-remove impurities, thereby simplifying downstream purification and ensuring that the final product meets stringent quality specifications required by regulatory bodies. The ability to tune the electronic and steric properties of the ligand system further enhances this control, allowing chemists to tailor the reaction outcomes for specific target molecules with high precision.
How to Synthesize Pyridine Derivatives Efficiently
Implementing this synthesis route requires careful attention to the preparation of the active catalyst species and the control of reaction parameters to maximize yield and purity. The process begins with the in situ generation of the active iron catalyst by mixing an iron salt precursor with a bisphosphine ligand and a reducing agent in an anhydrous solvent under an inert atmosphere. Once the catalyst is activated, the alkyne and nitrile substrates are introduced, and the mixture is allowed to react at controlled temperatures for a specified duration to ensure complete conversion. Detailed standardized synthesis steps see the guide below.
- Prepare the catalyst system by mixing iron salt precursors like FeCl3 with bisphosphine ligands such as dppp and a reducing agent.
- Combine the activated catalyst with diyne or monoalkyne substrates and nitrile reactants in a solvent like tetrahydrofuran.
- Maintain the reaction mixture at temperatures between room temperature and 100°C for 6 to 48 hours to achieve high yields.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iron-catalyzed technology translates into tangible strategic benefits that extend far beyond simple reaction chemistry. The substitution of noble metals with iron drastically reduces the raw material costs associated with catalyst procurement, insulating the manufacturing process from the price volatility often seen in the precious metals market. Additionally, the mild reaction conditions reduce energy consumption and lower the requirements for specialized high-pressure or high-temperature equipment, resulting in substantial capital expenditure savings and improved operational safety profiles. The simplified workup procedures also mean faster batch turnover times, allowing facilities to respond more agilely to market demand fluctuations without compromising on product quality or compliance standards.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts such as rhodium or cobalt removes a significant cost driver from the bill of materials, while the absence of heavy metal residues negates the need for costly purification steps like metal scavenging. This dual saving mechanism ensures that the overall cost of goods sold is significantly lowered, improving margin potential for high-volume pharmaceutical intermediates. Furthermore, the use of common solvents and readily available reagents minimizes procurement complexity and reduces the risk of supply disruptions caused by specialized chemical shortages.
- Enhanced Supply Chain Reliability: Iron is one of the most abundant elements on earth, ensuring a stable and secure supply of the primary catalytic metal that is not subject to the geopolitical constraints affecting rare earth or precious metal mining. The robustness of the catalyst system allows for consistent performance across different batches, reducing the variability that often leads to production delays and quality investigations. This reliability is crucial for maintaining continuous supply to downstream customers, particularly in the pharmaceutical sector where interruption can have severe consequences for drug availability.
- Scalability and Environmental Compliance: The green chemistry attributes of this process, including high atom economy and reduced waste generation, facilitate easier regulatory approval and environmental permitting for new manufacturing lines. The mild operating conditions make the process inherently safer and easier to scale from laboratory to commercial production volumes without the need for extensive re-engineering of reactor systems. This scalability ensures that companies can rapidly ramp up production to meet commercial demand while maintaining a strong environmental, social, and governance (ESG) profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iron-catalyzed pyridine synthesis technology in industrial settings. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation to ensure accuracy and relevance for decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this process into existing manufacturing workflows.
Q: What are the advantages of using iron catalysts over traditional noble metals for pyridine synthesis?
A: Iron catalysts offer significant cost reductions due to the abundance of iron compared to cobalt or rhodium, while providing high atom economy and reduced environmental toxicity in pharmaceutical intermediate manufacturing.
Q: What is the regioselectivity performance of this iron-catalyzed system?
A: The system demonstrates excellent regioselectivity, particularly in monoalkyne reactions where the ratio of desired isomers can exceed 9:1, minimizing downstream purification costs.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the mild reaction conditions ranging from room temperature to 100°C and the use of common solvents like THF make the process highly suitable for commercial scale-up and continuous manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyridine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iron-catalyzed technology for the production of high-value pharmaceutical intermediates and fine chemicals. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of pyridine derivatives meets the exacting standards required by global pharmaceutical clients. We are committed to leveraging advanced catalytic technologies to deliver superior product quality and consistency.
We invite procurement leaders and technical directors to collaborate with us to explore how this cost-effective synthesis route can optimize your supply chain and reduce manufacturing expenses. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific product portfolio, demonstrating the economic benefits of switching to this iron-based system. Please contact our technical procurement team to request specific COA data and route feasibility assessments for your target molecules, and let us help you secure a competitive advantage in the global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
