Advanced Manufacturing of Cyclopropylacetylene Derivatives for Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust synthetic pathways for complex intermediates, particularly those serving as building blocks for potent antiviral agents. Patent CN1070833C introduces a groundbreaking methodology for the preparation of cyclopropylacetylene derivatives, which are critical precursors in the synthesis of compounds possessing a cyclopropane skeleton, such as the anti-HIV active oxazinone L-743726. This technology addresses long-standing challenges in organic synthesis by providing a route that operates under mild conditions while achieving high yields, a combination rarely found in traditional acetylene chemistry. The core innovation lies in transforming readily available cyclopropylacrylic acid derivatives into the target acetylene structures through a controlled halogenation and elimination sequence. 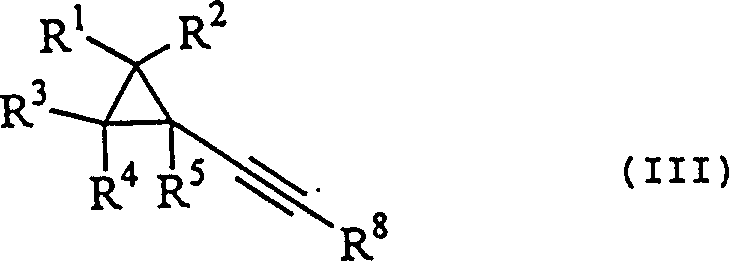 For procurement specialists and R&D directors, this represents a significant opportunity to secure a reliable cyclopropylacetylene supplier capable of delivering high-purity materials without the volatility associated with older synthetic methods. The ability to produce these derivatives efficiently directly impacts the cost structure and supply continuity of downstream API manufacturing.
For procurement specialists and R&D directors, this represents a significant opportunity to secure a reliable cyclopropylacetylene supplier capable of delivering high-purity materials without the volatility associated with older synthetic methods. The ability to produce these derivatives efficiently directly impacts the cost structure and supply continuity of downstream API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cyclopropylacetylene has been plagued by significant technical and economic hurdles that hinder industrial scalability. Conventional Method 1 involves the reaction of cyclopropylmethyl ketone with phosphorus pentachloride followed by dehydrochlorination, a process notorious for generating substantial amounts of by-products and suffering from low target compound productivity. Method 2 relies on the use of 5-chloropentyne reacting with n-butyllithium; while chemically feasible, this approach necessitates the use of expensive and hazardous organolithium reagents, posing severe safety risks and cost burdens for large-scale operations. Furthermore, Method 3 utilizes a Wittig reaction involving carbon tetrabromide and triphenylphosphine, which inevitably produces large quantities of triphenylphosphine oxide. The separation and removal of this oxide by-product are notoriously troublesome and costly, often requiring extensive chromatography or recrystallization steps that reduce overall throughput. These limitations collectively render traditional methods unsuitable for modern, cost-sensitive pharmaceutical manufacturing environments where efficiency and safety are paramount.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a cyclopropylacrylic acid derivative as a pivotal intermediate, bypassing the need for hazardous organometallics or wasteful phosphine reagents. This method begins with the conversion of cyclopropanecarbaldehyde derivatives into cyclopropylacrylic acid derivatives, which are then subjected to a halogenation reaction to form halocyclopropylpropionic acid derivatives. 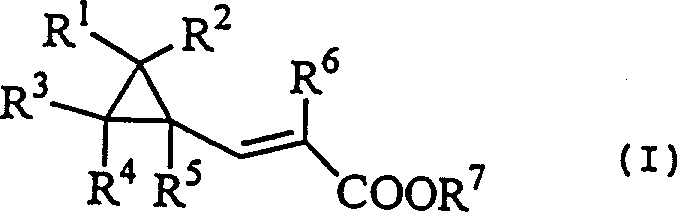 The subsequent reaction of these halogenated intermediates with a base facilitates a smooth elimination process to yield the desired cyclopropylacetylene. This pathway is advantageous because it avoids the aggressive conditions that typically lead to the ring-opening of the sensitive cyclopropane moiety. By leveraging mild reaction temperatures and standard halogenating agents, this novel approach ensures high selectivity and yield. For a reliable agrochemical intermediate supplier or pharma partner, adopting this route means drastically simplified purification protocols and a significant reduction in raw material costs, directly translating to enhanced commercial viability.
The subsequent reaction of these halogenated intermediates with a base facilitates a smooth elimination process to yield the desired cyclopropylacetylene. This pathway is advantageous because it avoids the aggressive conditions that typically lead to the ring-opening of the sensitive cyclopropane moiety. By leveraging mild reaction temperatures and standard halogenating agents, this novel approach ensures high selectivity and yield. For a reliable agrochemical intermediate supplier or pharma partner, adopting this route means drastically simplified purification protocols and a significant reduction in raw material costs, directly translating to enhanced commercial viability.
Mechanistic Insights into Halogenation-Elimination Sequence
The mechanistic elegance of this process lies in its ability to construct the acetylene functionality without compromising the integrity of the adjacent cyclopropane ring, a feat that requires precise control over reaction parameters. The synthesis initiates with the condensation of a cyclopropanecarbaldehyde derivative with an ester in the presence of a base, such as sodium methoxide or pyridine, to generate the unsaturated cyclopropylacrylic acid derivative. 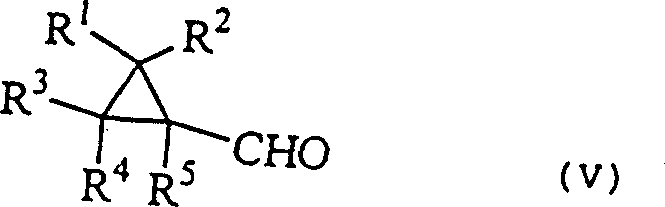 This step is critical as it establishes the carbon backbone required for the subsequent transformations. The resulting acrylic derivative is then treated with a halogenating agent, preferably bromine or chlorine, at controlled temperatures ranging from -20°C to 40°C. This halogenation adds across the double bond to form a dihalo-intermediate. The final transformation involves a double elimination reaction induced by a strong base, such as potassium tert-butoxide or sodium hydroxide. This base abstracts protons to eliminate two molecules of hydrogen halide, thereby forming the triple bond. The careful selection of base strength and solvent polarity is essential to drive the reaction to completion while preventing nucleophilic attack on the cyclopropane ring, which could lead to ring-opening side reactions.
This step is critical as it establishes the carbon backbone required for the subsequent transformations. The resulting acrylic derivative is then treated with a halogenating agent, preferably bromine or chlorine, at controlled temperatures ranging from -20°C to 40°C. This halogenation adds across the double bond to form a dihalo-intermediate. The final transformation involves a double elimination reaction induced by a strong base, such as potassium tert-butoxide or sodium hydroxide. This base abstracts protons to eliminate two molecules of hydrogen halide, thereby forming the triple bond. The careful selection of base strength and solvent polarity is essential to drive the reaction to completion while preventing nucleophilic attack on the cyclopropane ring, which could lead to ring-opening side reactions.
Impurity control is another cornerstone of this mechanistic design, ensuring the production of high-purity cyclopropylacetylene derivatives suitable for sensitive biological applications. The patent explicitly notes that side reactions, such as the electrophilic ring-opening of the cyclopropane by bromine, are minimized by maintaining specific temperature ranges and stoichiometric ratios. Furthermore, the process allows for the isolation of the intermediate cyclopropylvinyl derivative, which can be purified before the final elimination step. This modular approach enables manufacturers to remove impurities early in the sequence, preventing their carryover into the final product. By utilizing standard workup procedures like aqueous washing and distillation, the process effectively removes inorganic salts and residual solvents. This rigorous control over the reaction pathway ensures that the final API intermediate meets stringent purity specifications, reducing the risk of batch failures and ensuring consistent quality for downstream drug synthesis.
How to Synthesize Cyclopropylacetylene Efficiently
The operational protocol for synthesizing these valuable derivatives is designed for reproducibility and safety, making it accessible for both laboratory optimization and plant-scale production. The process begins with the preparation of the acrylic intermediate, followed by halogenation and final elimination, with each step optimized for maximum conversion. Detailed standardized synthesis steps are provided below to guide technical teams in implementing this efficient route. Adhering to these guidelines ensures that the delicate balance between reactivity and selectivity is maintained throughout the synthesis. Operators should pay close attention to temperature control during the halogenation phase, as exothermic reactions must be managed carefully to prevent thermal runaway. Additionally, the choice of solvent plays a crucial role in solubility and reaction kinetics, with options ranging from hydrocarbons to halogenated solvents depending on the specific substrate. Proper quenching and extraction techniques are also vital to isolate the product cleanly, ensuring that the final material is free from residual bases or halogens that could affect stability.
- Condense cyclopropanecarbaldehyde derivatives with esters in the presence of a base to form cyclopropylacrylic acid derivatives.
- React the resulting acrylic derivatives with a halogenating agent to generate dihalo-cyclopropylpropionic acid intermediates.
- Treat the halogenated intermediate with a strong base to induce double elimination, yielding the final cyclopropylacetylene product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers transformative benefits for procurement managers and supply chain heads looking to optimize their sourcing strategies for complex intermediates. The shift away from expensive and hazardous reagents like n-butyllithium towards common halogens and bases results in substantial cost savings in pharmaceutical intermediates manufacturing. By eliminating the need for cryogenic conditions and specialized handling equipment for pyrophoric materials, the process significantly lowers capital expenditure and operational overhead. Furthermore, the avoidance of triphenylphosphine oxide by-products removes a major bottleneck in waste management and purification, streamlining the production timeline. These efficiencies contribute to a more resilient supply chain, reducing the risk of delays caused by reagent shortages or complex waste disposal regulations.
- Cost Reduction in Manufacturing: The replacement of high-cost organolithium reagents with inexpensive halogenating agents and alkali bases drives down the direct material cost per kilogram. Additionally, the simplified purification process reduces solvent consumption and energy usage associated with extensive chromatography or recrystallization. This holistic reduction in processing complexity allows for a more competitive pricing structure without compromising on quality. The ability to run reactions at near-ambient temperatures further decreases utility costs, making the overall process economically superior to legacy methods.
- Enhanced Supply Chain Reliability: The starting materials, such as cyclopropanecarbaldehyde and common esters, are widely available commodities with stable global supply chains. This availability mitigates the risk of production stoppages due to raw material scarcity, a common issue with specialized organometallic reagents. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor variations in input quality, ensuring consistent output even when sourcing from multiple vendors. This reliability is crucial for maintaining continuous API production schedules and meeting tight delivery deadlines.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial application in mind, allowing for seamless transition from pilot batches to multi-ton commercial production. The use of standard solvents and the generation of manageable inorganic by-products simplify waste treatment and align with increasingly strict environmental regulations. By minimizing the generation of hazardous waste and avoiding persistent organic pollutants, this method supports sustainable manufacturing practices. This environmental compatibility not only reduces compliance costs but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the patent's technical disclosures and beneficial effects. These insights are intended to clarify the operational advantages and feasibility of adopting this technology for large-scale production. Understanding these nuances helps stakeholders make informed decisions about integrating this process into their existing manufacturing portfolios. The answers reflect a deep understanding of the chemical principles involved and the practical realities of industrial organic synthesis.
Q: Why is this new method superior to traditional organolithium routes?
A: Traditional methods often rely on expensive and hazardous reagents like n-butyllithium or generate difficult-to-remove byproducts like triphenylphosphine oxide. This patented process utilizes mild halogenation and elimination steps, significantly reducing raw material costs and simplifying purification workflows.
Q: How does this process ensure the stability of the cyclopropane ring?
A: The cyclopropane ring is highly strained and prone to electrophilic ring-opening. This method carefully controls reaction conditions, specifically temperature and halogenating agents, to prevent ring opening while successfully forming the acetylene functionality, ensuring high structural integrity.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process operates under mild temperatures ranging from -20°C to 100°C and uses common industrial solvents. The avoidance of cryogenic conditions and pyrophoric reagents makes it inherently safer and more scalable for multi-ton manufacturing compared to legacy methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclopropylacetylene Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of cyclopropylacetylene derivatives performs consistently in your downstream synthesis. Our expertise in managing complex chemical transformations allows us to navigate the intricacies of this patented process, delivering value through both technical excellence and operational efficiency.
We invite you to collaborate with us to leverage these advanced manufacturing capabilities for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain security and reduce your overall cost of goods sold. Let us be your partner in turning complex chemistry into commercial success.
