Advanced Manufacturing of Cyclopropylacetylene Derivatives for Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust synthetic pathways for complex scaffolds, particularly those containing strained ring systems like cyclopropane which are prevalent in modern antiviral therapeutics. Patent CN1191856A discloses a groundbreaking methodology for the preparation of cyclopropylacetylene derivatives, which serve as critical building blocks for compounds exhibiting anti-HIV activity, such as the oxazinone L-743726. This technical disclosure addresses long-standing challenges in constructing the acetylene functionality adjacent to a sensitive cyclopropane ring without inducing ring-opening side reactions. The core innovation lies in a multi-step sequence starting from cyclopropylacrylic acid derivatives, proceeding through halogenation, and concluding with a base-mediated elimination. 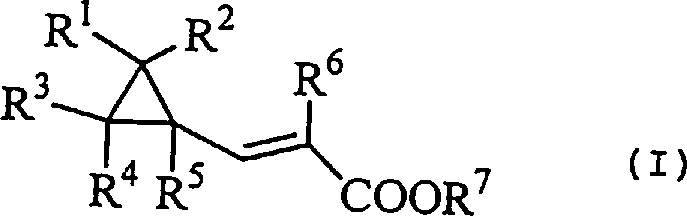 As illustrated in the general structure above, the versatility of the R-groups allows for significant structural diversity, enabling the synthesis of a broad library of intermediates essential for drug discovery and process development teams aiming to optimize lead compounds.
As illustrated in the general structure above, the versatility of the R-groups allows for significant structural diversity, enabling the synthesis of a broad library of intermediates essential for drug discovery and process development teams aiming to optimize lead compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cyclopropylacetylene has been plagued by significant operational and economic hurdles that hinder industrial adoption. Traditional routes often rely on the reaction of cyclopropylmethyl ketones with phosphorus pentachloride followed by dehydrochlorination, a process notorious for generating complex mixtures of by-products and suffering from low target compound yields. Another common approach involves the use of n-butyllithium or lithium diisopropylamide to react with chloropentyne; however, these organometallic reagents are not only prohibitively expensive but also pose severe safety risks regarding pyrophoricity and moisture sensitivity on a large scale. Furthermore, Wittig reaction pathways utilizing carbon tetrabromide and triphenylphosphine produce stoichiometric amounts of triphenylphosphine oxide, a by-product that is notoriously difficult to separate from the desired product, thereby complicating downstream purification and increasing waste disposal costs substantially.
The Novel Approach
In stark contrast to these legacy methods, the process outlined in CN1191856A offers a streamlined and economically viable alternative that circumvents the need for hazardous organometallics and difficult purifications. By utilizing a cyclopropylacrylic acid derivative as the foundational substrate, the invention enables a controlled transformation where the double bond is first converted into a dihalo-species and subsequently eliminated to form the triple bond. This strategy operates under much milder conditions, typically avoiding the extreme temperatures required for carbene generation or the cryogenic conditions needed for lithiation. The ability to use common halogenating agents like bromine or chlorine, followed by treatment with standard inorganic or alkoxide bases, drastically simplifies the reactor setup and reduces the overall cost of goods sold, making it an attractive option for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Halogenation-Elimination Cascade
The mechanistic elegance of this synthesis lies in its stepwise construction of the alkyne moiety while preserving the integrity of the adjacent cyclopropane ring. The process initiates with the condensation of cyclopropanecarbaldehyde with esters or malonic acid derivatives to form the acrylic acid precursor, a reaction that can be driven to completion by azeotropic removal of water. Subsequently, the introduction of a halogenating agent adds across the double bond to yield a dihalocyclopropylpropionic acid derivative. This halogenated intermediate is pivotal, as it sets the stage for the final elimination. When treated with a base, such as potassium tert-butoxide or sodium methoxide, the molecule undergoes a double dehydrohalogenation. Crucially, this proceeds via a cyclopropylvinyl derivative intermediate, which can be isolated or carried forward in situ. 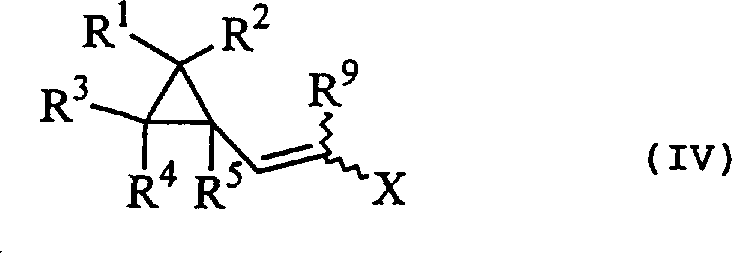 The formation of this vinyl species, as shown in the structure above, confirms the stepwise nature of the elimination, allowing chemists to monitor reaction progress and control stereochemistry before the final conversion to the acetylene.
The formation of this vinyl species, as shown in the structure above, confirms the stepwise nature of the elimination, allowing chemists to monitor reaction progress and control stereochemistry before the final conversion to the acetylene.
From an impurity control perspective, this mechanism offers distinct advantages over direct alkyne formation methods. The stepwise halogenation and elimination allow for the identification and removal of mono-eliminated by-products before the final step, ensuring a cleaner crude profile. The patent data indicates that by carefully selecting the base strength and reaction temperature, one can minimize ring-opening side reactions which are thermodynamically favorable in highly strained systems. For instance, using weaker bases at lower temperatures favors the isolation of the vinyl intermediate, while stronger bases at elevated temperatures drive the reaction to the final alkyne. This tunability provides process chemists with a powerful lever to optimize yield and purity, ensuring that the final active pharmaceutical ingredient meets stringent regulatory specifications for residual halogens and isomeric impurities.
How to Synthesize Cyclopropylacetylene Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and safety. The process begins with the preparation of the cyclopropylacrylic acid derivative, followed by halogenation and final elimination. Detailed operational protocols regarding solvent selection, stoichiometry, and workup procedures are critical for successful replication. The following guide outlines the standardized synthesis steps derived from the patent examples to assist technical teams in process validation.
- Condense cyclopropanecarbaldehyde with an ester or malonic acid in the presence of a base to form cyclopropylacrylic acid derivatives.
- React the resulting acrylic acid derivative with a halogenating agent such as bromine or chlorine to generate a dihalocyclopropylpropionic acid intermediate.
- Treat the halogenated intermediate with a strong base like potassium tert-butoxide to effect double elimination, yielding the final cyclopropylacetylene product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route represents a strategic opportunity to enhance supply security and reduce manufacturing overheads. By eliminating the dependency on volatile and expensive organolithium reagents, the process stabilizes the raw material supply chain against market fluctuations associated with specialty metal reagents. Furthermore, the use of commodity chemicals such as bromine, chlorine, and common alkoxides ensures that sourcing is straightforward and less susceptible to geopolitical disruptions. The simplified purification process, which avoids the generation of tenacious phosphine oxide waste, translates directly into reduced waste treatment costs and shorter cycle times, allowing for faster throughput in multipurpose manufacturing facilities.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of high-cost reagents with inexpensive commodity chemicals. By removing the need for n-butyllithium and triphenylphosphine, the direct material costs are significantly lowered. Additionally, the avoidance of complex chromatographic separations required to remove phosphine oxides reduces solvent consumption and labor hours. The ability to perform the reaction in standard solvents like dichloromethane or alcohols further contributes to cost efficiency, as these solvents are easily recovered and recycled within existing distillation infrastructure, leading to substantial overall cost savings in the production of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is markedly improved due to the widespread availability of the key starting materials and reagents. Cyclopropanecarbaldehyde and simple esters are produced by numerous global suppliers, reducing the risk of single-source bottlenecks. The robustness of the reaction conditions, which tolerate a broader range of temperatures and moisture levels compared to organometallic processes, minimizes the risk of batch failures due to minor environmental deviations. This reliability ensures consistent delivery schedules and reduces the need for excessive safety stock, thereby optimizing inventory management and working capital for downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this process aligns well with green chemistry principles by reducing the E-factor through minimized waste generation. The absence of heavy metal catalysts and the use of recyclable solvents simplify the environmental permitting process for new production lines. The reaction exotherms are manageable with standard cooling systems, facilitating safe scale-up from pilot plant to commercial tonnage without requiring specialized cryogenic reactors. This ease of scale-up ensures that supply can be rapidly ramped to meet clinical trial demands or commercial launch volumes without significant capital expenditure on new equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cyclopropylacetylene synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation to provide clarity on process capabilities and limitations.
Q: What are the primary advantages of this synthesis route over conventional methods?
A: Unlike conventional methods requiring expensive organolithium reagents or generating difficult-to-remove triphenylphosphine oxide waste, this process utilizes readily available halogenating agents and standard bases, significantly simplifying purification and reducing raw material costs.
Q: Can this process be scaled for industrial production of API intermediates?
A: Yes, the reaction conditions are mild, typically ranging from -20°C to 100°C, and utilize common organic solvents, making the process highly amenable to commercial scale-up from kilogram to multi-ton quantities without specialized cryogenic equipment.
Q: What is the expected purity profile of the final cyclopropylacetylene derivative?
A: Through optimized distillation and crystallization steps described in the patent examples, the final product can achieve purity levels exceeding 99%, with specific isomer control (Z/E ratio) manageable through base selection and reaction temperature.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclopropylacetylene Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory synthesis to industrial manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of cyclopropylacetylene derivative meets the highest standards of quality and consistency required by global regulatory bodies.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your timeline and optimize your budget for high-purity pharmaceutical intermediates.
