Advanced Manufacturing of Cyclopropylacetylene Intermediates for Pharmaceutical Applications
Advanced Manufacturing of Cyclopropylacetylene Intermediates for Pharmaceutical Applications
The pharmaceutical industry constantly seeks robust synthetic routes for complex scaffolds, particularly those containing strained rings like cyclopropanes which are prevalent in modern antiviral therapeutics. Patent CN1129567C discloses a groundbreaking methodology for the preparation of cyclopropylacetylene derivatives, specifically targeting intermediates useful for synthesizing compounds with anti-HIV activity, such as the benzoxazinone derivative L-743726. This technology represents a significant leap forward in process chemistry, addressing long-standing issues regarding yield and scalability that have plagued traditional synthesis methods. By leveraging a unique sequence involving protected propargyl alcohol derivatives and specific propane dihalides, the process ensures high purity and exceptional conversion rates. For R&D directors and procurement specialists alike, understanding the nuances of this patent is critical for securing a reliable pharmaceutical intermediates supplier capable of delivering high-purity API precursors consistently.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of cyclopropylacetylene has been fraught with inefficiencies that hinder industrial adoption. One prominent conventional method involves the reaction of 5-chloropentyne with n-butyllithium, a process documented to provide a disappointing yield of merely 57 percent for the starting material itself, creating a bottleneck before the synthesis even truly begins. Another established route utilizes cyclopropylmethyl ketone reacted with phosphorus pentachloride to generate 1,1-dichloro-1-cyclopropylethane, followed by dehydrochlorination; however, this pathway is notorious for generating a multitude of difficult-to-separate by-products, drastically reducing the overall yield of the target product. These legacy methods are chemically cumbersome, often requiring harsh conditions that compromise safety and increase waste disposal costs, making them unsuitable for the rigorous demands of modern GMP manufacturing environments where consistency is paramount.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a sophisticated strategy starting with protected propargyl alcohol derivatives, which offer superior stability and handling characteristics compared to free alkynes. 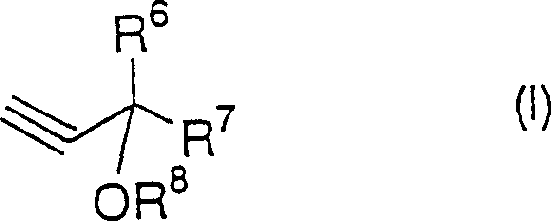 . The core innovation lies in reacting these stable precursors with propane derivatives, such as 1-bromo-3-chloropropane, in the presence of a controlled amount of strong base. This method allows for the precise construction of the carbon skeleton prior to ring closure, minimizing side reactions. Experimental data from the patent demonstrates that this route can achieve isolated yields exceeding 90 percent, as seen in Example 1 where a 91.6 percent yield was obtained. This dramatic improvement in efficiency translates directly into cost reduction in pharmaceutical intermediates manufacturing, providing a compelling economic argument for adopting this technology over obsolete methods.
. The core innovation lies in reacting these stable precursors with propane derivatives, such as 1-bromo-3-chloropropane, in the presence of a controlled amount of strong base. This method allows for the precise construction of the carbon skeleton prior to ring closure, minimizing side reactions. Experimental data from the patent demonstrates that this route can achieve isolated yields exceeding 90 percent, as seen in Example 1 where a 91.6 percent yield was obtained. This dramatic improvement in efficiency translates directly into cost reduction in pharmaceutical intermediates manufacturing, providing a compelling economic argument for adopting this technology over obsolete methods.
Mechanistic Insights into Base-Mediated Cyclization and Retro-Ethynylation
The chemical elegance of this process is rooted in its multi-step mechanistic pathway which carefully manages reactivity to prevent polymerization or decomposition. Initially, the protected propargyl alcohol is deprotonated by a strong base such as lithium amide or n-butyllithium at low temperatures, typically between -50°C and -40°C, to generate a nucleophilic acetylide species. This species then undergoes alkylation with the dihalide propane derivative. Depending on the stoichiometry of the base used, the reaction can be steered towards either a linear acetylene derivative or directly towards the cyclized product. 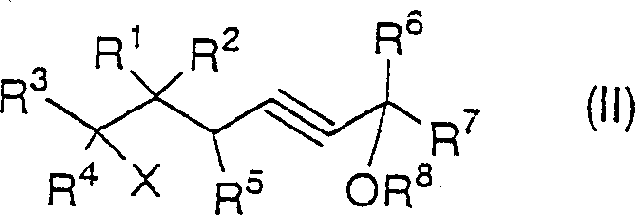 . When excess base (greater than 2 equivalents) is employed, the system favors the formation of the cyclopropane ring through an intramolecular nucleophilic substitution, effectively closing the three-membered ring with high stereochemical integrity.
. When excess base (greater than 2 equivalents) is employed, the system favors the formation of the cyclopropane ring through an intramolecular nucleophilic substitution, effectively closing the three-membered ring with high stereochemical integrity. 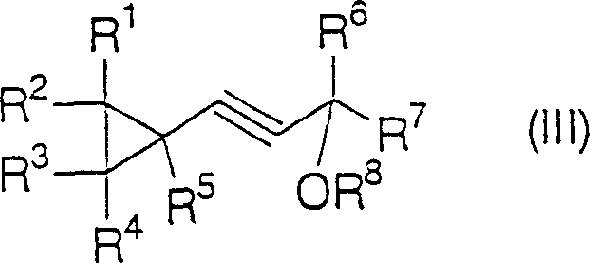 . This control over the reaction trajectory is vital for impurity control, ensuring that the final product profile remains clean and易于 purification.
. This control over the reaction trajectory is vital for impurity control, ensuring that the final product profile remains clean and易于 purification.
Following the cyclization, the process employs a strategic deprotection step to reveal the hydroxyl group, followed by a retro-ethynylation reaction. This final transformation is particularly ingenious as it releases the volatile and potentially unstable cyclopropylacetylene moiety only at the very end of the synthesis, thereby minimizing degradation losses during upstream processing. The use of catalytic amounts of base in this final step facilitates the cleavage of the carbon-carbon bond to release the target acetylene while generating a stable ketone by-product that is easily separated. This mechanistic design significantly enhances the overall mass balance of the process. For technical teams evaluating route feasibility, this level of mechanistic control suggests a robust process capable of maintaining stringent purity specifications even when scaled to multi-ton quantities, reducing the risk of batch failures due to trace impurities.
How to Synthesize Cyclopropylacetylene Derivatives Efficiently
Implementing this synthesis requires precise adherence to the reaction conditions outlined in the patent to maximize yield and safety. The process begins with the careful preparation of the reaction vessel under an inert atmosphere, followed by the sequential addition of reagents at strictly controlled low temperatures to manage the exothermic nature of the lithiation steps. Solvent selection is also critical, with tetrahydrofuran and liquid ammonia proving effective for solubilizing the intermediates while maintaining the reactivity of the organometallic species. The detailed standardized synthesis steps见下方的指南 outline the exact molar ratios and temperature ramps necessary to reproduce the high yields reported in the examples, serving as a foundational protocol for process development teams aiming to transfer this technology to pilot or production scales.
- React protected propargyl alcohol derivatives with propane dihalides using strong bases like lithium amide or butyllithium at low temperatures.
- Isolate the resulting cyclopropane derivatives and perform deprotection of the hydroxyl group using acid or fluoride sources.
- Subject the deprotected cyclopropyl propargyl alcohol to retro-ethynylation conditions with catalytic base to release the final cyclopropylacetylene.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers substantial strategic benefits for procurement managers and supply chain heads tasked with securing critical raw materials. The shift away from low-yielding, by-product-heavy conventional methods towards this high-efficiency pathway fundamentally alters the cost structure of producing these valuable intermediates. By eliminating the need for complex purification trains required to remove the numerous by-products of older methods, the process inherently reduces manufacturing costs and shortens production cycles. Furthermore, the reliance on readily available commodity chemicals like 1-bromo-3-chloropropane and standard protecting group reagents mitigates supply risk, ensuring that production is not held hostage by the scarcity of exotic starting materials. This reliability is essential for maintaining continuous supply chains for downstream API manufacturing.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the dramatic increase in chemical yield, which moves from roughly 57 percent in legacy methods to over 90 percent in this new protocol. This near-doubling of efficiency means that for every kilogram of input material, significantly more product is generated, effectively lowering the cost per unit of active intermediate. Additionally, the process avoids the use of expensive transition metal catalysts or hazardous reagents like phosphorus pentachloride, which require specialized handling and waste treatment infrastructure. The elimination of these costly inputs, combined with the simplified work-up procedures described in the patent examples, results in substantial cost savings that can be passed down the supply chain or retained as margin.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by complex syntheses that are prone to failure or variability. This method enhances reliability by utilizing robust reaction conditions that are less sensitive to minor fluctuations in parameters, provided the temperature controls are maintained. The use of stable protected intermediates allows for potential storage and batching strategies that decouple upstream synthesis from the final volatile steps, providing greater flexibility in production scheduling. For a reliable pharmaceutical intermediates supplier, this means the ability to commit to longer-term contracts with confidence, knowing that the underlying chemistry is proven to be stable and reproducible across multiple batches as evidenced by the consistent yields in Examples 1 through 8.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and waste management, but this route is designed with scalability in mind. The reaction temperatures, while low, are within the range of standard industrial cryogenic capabilities (-50°C to 0°C), avoiding the extreme conditions that limit scale-up. Moreover, the cleaner reaction profile generates less hazardous waste, simplifying environmental compliance and reducing the burden on effluent treatment plants. The ability to distill the final product directly from the reaction mixture in some embodiments further streamlines the isolation process, reducing solvent consumption and energy usage, which aligns with modern green chemistry principles and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical disclosures within patent CN1129567C, providing a factual basis for decision-making. Understanding these details helps stakeholders assess the fit of this technology within their existing manufacturing portfolios and identify potential areas for process optimization or customization.
Q: What are the primary advantages of this synthesis route over conventional methods?
A: Unlike conventional methods using 5-chloropentyne which suffer from low yields (around 57%), this patented process achieves yields exceeding 90% by utilizing stable protected propargyl alcohol precursors and controlled cyclization.
Q: How is the cyclopropane ring formed in this specific pathway?
A: The cyclopropane ring is constructed via an intramolecular nucleophilic substitution where a lithiated acetylene intermediate attacks a terminal halide on a three-carbon chain, facilitated by strong bases like lithium amide.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes commercially available reagents like 1-bromo-3-chloropropane and operates at manageable temperatures (-50°C to 0°C), making it highly scalable for commercial manufacturing of API intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclopropylacetylene Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to meeting stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of cyclopropylacetylene derivatives meets the exacting standards required for pharmaceutical applications. Our expertise in handling sensitive organometallic reactions and low-temperature processes positions us as an ideal partner for companies seeking to leverage this advanced synthesis technology.
We invite you to engage with our technical procurement team to discuss how we can support your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how implementing this patented route can optimize your budget without compromising quality. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a transparent and collaborative path forward in securing your supply of these essential chemical building blocks.
