Scalable Production of High-Purity Indanones via Novel Palladium-Catalyzed Carbonylative Cyclization
Scalable Production of High-Purity Indanones via Novel Palladium-Catalyzed Carbonylative Cyclization
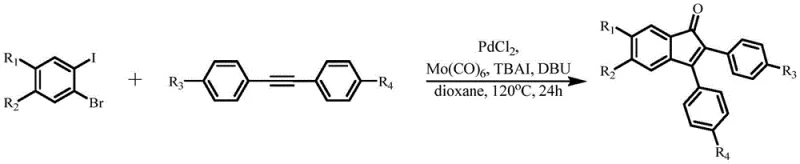
The pharmaceutical and fine chemical industries continuously seek robust methodologies for constructing complex heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. Patent CN114890883A introduces a significant advancement in the synthesis of indanone derivatives, a privileged structural motif found in numerous therapeutic agents including antidepressants and antispasmodics. This technology leverages a palladium-catalyzed carbonylative cyclization strategy that utilizes o-dihalobenzene series compounds and diphenylacetylene series compounds as key starting materials. Unlike traditional approaches that often rely on hazardous gaseous carbon monoxide, this novel method employs molybdenum hexacarbonyl (Mo(CO)6) as a safe and convenient solid carbonyl source. The process operates under relatively mild thermal conditions in a 1,4-dioxane solvent system, facilitated by a catalytic amount of PdCl2 alongside specific additives like TBAI and DBU. This innovation not only streamlines the synthetic route but also enhances the safety profile of the manufacturing process, making it highly attractive for the production of high-purity pharmaceutical intermediates.
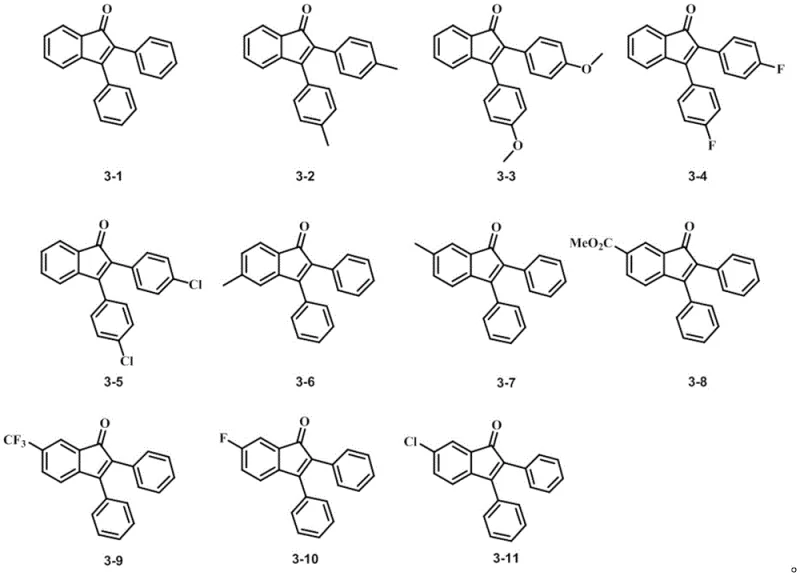
The versatility of this synthetic platform is evidenced by the broad scope of substrates it can accommodate, allowing for the generation of a diverse library of functionalized indanones. As illustrated in the structural array above, the method tolerates a wide range of substituents including alkyl, alkoxy, halo, and ester groups at various positions on the aromatic rings. This structural diversity is crucial for medicinal chemists aiming to optimize the pharmacokinetic properties of lead compounds. The ability to introduce electron-withdrawing groups such as trifluoromethyl or halogens, as well as electron-donating groups like methoxy, without compromising the reaction efficiency underscores the robustness of this catalytic system. For procurement managers and supply chain heads, this flexibility means that a single standardized protocol can be adapted to produce multiple distinct intermediates, thereby simplifying inventory management and reducing the need for specialized equipment for different product lines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indanone core has often relied on Friedel-Crafts acylation or transition-metal catalyzed carbonylation using gaseous carbon monoxide. While effective in some contexts, these conventional methods present significant logistical and safety challenges, particularly when scaling up for commercial production. The use of gaseous CO requires specialized high-pressure reactors and rigorous safety protocols to prevent leakage and exposure, which drastically increases capital expenditure and operational complexity. Furthermore, traditional Friedel-Crafts reactions often suffer from poor regioselectivity and generate substantial amounts of acidic waste, necessitating costly downstream purification and waste treatment processes. The harsh reaction conditions associated with these older methods can also limit the compatibility with sensitive functional groups, forcing chemists to employ additional protection and deprotection steps that lengthen the synthetic timeline and reduce overall atom economy. These factors collectively contribute to higher manufacturing costs and longer lead times, creating bottlenecks in the supply chain for critical API intermediates.
The Novel Approach
The methodology described in patent CN114890883A offers a transformative solution by replacing gaseous CO with the solid surrogate Mo(CO)6, effectively decoupling the synthesis from high-pressure infrastructure requirements. This shift allows the reaction to proceed in standard glassware or stainless steel reactors at atmospheric pressure, significantly lowering the barrier to entry for manufacturing facilities. The reaction is conducted at a moderate temperature of 120°C in 1,4-dioxane, which provides a stable environment for the catalytic cycle to proceed efficiently over a 24-hour period. The inclusion of TBAI and DBU plays a pivotal role in stabilizing the active palladium species and neutralizing acidic byproducts, respectively, ensuring consistent performance across different substrate batches. This approach not only mitigates safety risks but also simplifies the workup procedure, as the removal of solid residues is generally more straightforward than scrubbing toxic gases. By addressing the fundamental safety and operational hurdles of traditional carbonylation, this novel route enables a more agile and cost-effective production model for complex organic intermediates.
Mechanistic Insights into Pd-Catalyzed Carbonylative Cyclization
At the heart of this synthesis lies a sophisticated palladium-catalyzed mechanism that orchestrates the formation of two new carbon-carbon bonds and one carbon-oxygen bond in a single operational step. The catalytic cycle initiates with the oxidative addition of the palladium(0) species into the carbon-iodine bond of the o-dihalobenzene substrate, which is typically more reactive than the carbon-bromine bond. This selectivity is crucial for controlling the regiochemistry of the subsequent steps. Following oxidative addition, the coordination and insertion of carbon monoxide, released in situ from the thermal decomposition of Mo(CO)6, generates an acyl-palladium intermediate. This acyl species then undergoes a migratory insertion with the alkyne moiety of the diphenylacetylene derivative, forming a new vinyl-palladium bond. The final ring-closing step involves the intramolecular nucleophilic attack or reductive elimination that constructs the five-membered indanone ring, regenerating the active palladium catalyst to continue the cycle. The precise tuning of ligands and additives ensures that each of these elementary steps proceeds with high fidelity, minimizing the formation of side products such as homocoupling dimers or uncyclized ketones.
Impurity control in this system is largely governed by the stability of the catalytic species and the efficiency of the base. The presence of TBAI (tetrabutylammonium iodide) is instrumental in maintaining the palladium in a highly active nano-dispersed state, preventing the precipitation of inactive palladium black which would otherwise halt the reaction and lead to incomplete conversion. Incomplete conversion is a major source of impurities, as unreacted starting materials can be difficult to separate from the product due to similar polarity. Furthermore, the use of DBU (1,8-diazabicyclo[5.4.0]undec-7-ene) as a non-nucleophilic base effectively scavenges the hydrogen halide byproducts generated during the cyclization. If left unchecked, these acidic species could promote the decomposition of sensitive functional groups or catalyze polymerization of the alkyne substrates. By maintaining a neutral to slightly basic environment throughout the 24-hour reaction window, the process ensures that the final crude mixture contains a high proportion of the desired indanone, thereby reducing the burden on downstream purification columns and enhancing the overall purity profile of the isolated material.
How to Synthesize Indanone Derivatives Efficiently
Implementing this synthesis requires careful attention to reagent stoichiometry and atmospheric control to maximize yield and reproducibility. The protocol dictates a specific molar ratio of substrates to catalyst and additives, typically employing a slight excess of the o-dihalobenzene relative to the alkyne to drive the reaction to completion. The reaction must be performed under an inert nitrogen atmosphere to prevent oxidation of the phosphine-free palladium catalyst and the sensitive alkyne substrates. Monitoring the reaction progress via thin-layer chromatography (TLC) is recommended to determine the optimal quenching time, although the standard 24-hour duration at 120°C has been proven effective for a wide range of substrates. Detailed standard operating procedures regarding specific mixing orders and temperature ramping rates are essential for transferring this chemistry from the bench to the pilot plant.
- Mix o-dihalobenzene derivatives, diphenylacetylene compounds, Mo(CO)6, TBAI, DBU, and PdCl2 catalyst in 1,4-dioxane solvent under inert atmosphere.
- Heat the reaction mixture to 120°C and stir for 24 hours to facilitate the carbonylative cyclization process.
- Purify the crude product mixture through dilution, washing, drying, and flash column chromatography to isolate the target indanone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the manufacturing infrastructure required. By eliminating the need for high-pressure carbon monoxide cylinders and specialized gas handling systems, facilities can utilize existing standard reactor trains, thereby avoiding significant capital investment. This flexibility allows for faster deployment of production lines and reduces the regulatory burden associated with handling toxic gases. Moreover, the use of commercially available and stable reagents like Mo(CO)6 and PdCl2 ensures a reliable supply chain, as these materials are produced at scale by multiple global suppliers, mitigating the risk of single-source bottlenecks. The robustness of the reaction conditions also implies a lower rate of batch failures, leading to more predictable production schedules and improved on-time delivery performance for downstream customers.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the elimination of expensive high-pressure equipment and the reduction in safety compliance costs associated with toxic gas handling. Additionally, the high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, which reduces the consumption of silica gel and solvents during the purification phase. The use of a solid carbonyl source also improves atom economy compared to methods requiring large excesses of gaseous reagents to maintain pressure. These factors collectively contribute to a lower cost of goods sold (COGS), allowing for more competitive pricing in the global market for pharmaceutical intermediates without sacrificing margin.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable solid reagents rather than compressed gases significantly enhances supply chain resilience. Mo(CO)6 and the organic substrates can be stored in standard warehouses without special ventilation or pressure containment, simplifying logistics and inventory management. The tolerance of the reaction to various functional groups means that a single production line can be easily switched between different indanone derivatives with minimal changeover time. This agility allows manufacturers to respond quickly to fluctuating market demands and urgent orders from pharmaceutical clients, ensuring continuity of supply even during periods of raw material volatility.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns well with green chemistry principles by avoiding the release of carbon monoxide into the atmosphere. The waste stream is primarily composed of organic solvents and solid salts, which are easier to treat and dispose of compared to heavy metal-contaminated acidic waste from Friedel-Crafts processes. The scalability of the reaction is supported by its homogeneous nature and moderate temperature profile, which facilitates heat transfer in larger reactors. This makes the transition from kilogram-scale development to multi-ton commercial production straightforward, ensuring that the supply can grow in tandem with the clinical or commercial success of the final drug product.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this indanone synthesis technology. These insights are derived directly from the experimental data and embodiments disclosed in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What is the primary advantage of using Mo(CO)6 over gaseous CO in this synthesis?
A: Using Mo(CO)6 as a solid carbonyl source eliminates the need for high-pressure gas equipment and significantly reduces safety risks associated with handling toxic carbon monoxide gas, making the process more suitable for standard laboratory and pilot plant settings.
Q: What types of substituents are tolerated on the benzene rings during this reaction?
A: The reaction demonstrates excellent functional group tolerance, accommodating electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as fluoro, chloro, trifluoromethyl, and ester groups on both the dihalobenzene and diphenylacetylene substrates.
Q: How does the addition of TBAI and DBU impact the reaction efficiency?
A: TBAI acts as an additive to stabilize the generated nano-palladium species, preventing catalyst aggregation, while DBU serves as a robust base to neutralize acid byproducts, collectively ensuring high catalytic turnover and consistent yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the drug development lifecycle. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of indanone derivatives meets the exacting standards required by global pharmaceutical regulators. Our commitment to process excellence means we can leverage advanced catalytic technologies like the one described in CN114890883A to deliver superior products with consistent quality profiles.
We invite you to collaborate with us to optimize your supply chain for indanone-based projects. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you accelerate your project timelines with our reliable manufacturing capabilities and deep technical expertise in complex organic synthesis.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
