Advanced Palladium-Catalyzed Synthesis of Indanone Derivatives for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of Indanone Derivatives for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient routes to access complex heterocyclic scaffolds, particularly indanone derivatives which serve as critical building blocks for bioactive molecules. A significant breakthrough in this domain is detailed in Chinese Patent CN110028394B, which discloses a highly efficient method for synthesizing indanone and its derivatives through a palladium-catalyzed three-component coupling carbonylation reaction. This innovative approach addresses long-standing challenges in organic synthesis by utilizing readily available starting materials and mild reaction conditions to construct the indenone core with exceptional atom economy. For R&D directors and procurement specialists, this technology represents a paradigm shift away from cumbersome multi-step sequences towards a streamlined, one-pot protocol that significantly enhances process viability. The ability to form three new carbon-carbon bonds in a single operation not only reduces waste but also simplifies the purification workflow, offering substantial potential for cost optimization in the manufacturing of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of indanone compounds has relied heavily on classical methodologies such as the Friedel-Crafts acylation or transition metal-catalyzed cyclizations that often suffer from significant operational drawbacks. Traditional routes typically necessitate the pre-introduction of carbonyl functional group precursors, such as acyl or aldehyde groups, onto the aromatic substrate prior to the cyclization step. This requirement inherently complicates the synthetic pathway, forcing chemists to engage in multi-step preparations to generate these specialized starting materials, which are frequently difficult to purchase commercially and expensive to produce. Furthermore, these conventional methods often struggle with regioselectivity issues and harsh reaction conditions that can degrade sensitive functional groups, leading to lower overall yields and complicated impurity profiles. The reliance on stoichiometric amounts of Lewis acids or harsh reagents also generates substantial chemical waste, creating environmental burdens and increasing the cost of waste disposal for large-scale production facilities.
The Novel Approach
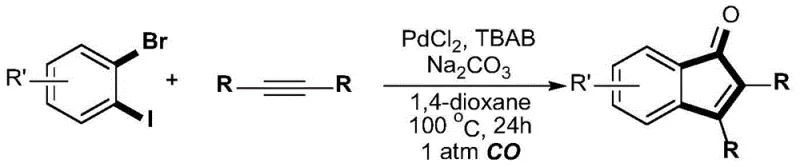
In stark contrast to these legacy methods, the technology described in patent CN110028394B introduces a transformative strategy that utilizes carbon monoxide directly as the carbonyl source, thereby eliminating the need for pre-functionalized carbonyl precursors entirely. This novel approach employs a palladium-catalyzed multicomponent reaction involving o-bromoiodobenzene compounds, dihydrocarbylacetylene compounds, and carbon monoxide gas under relatively mild thermal conditions. By integrating the carbonylation step directly into the cyclization process, this method achieves the construction of the indanone skeleton in a single pot, effectively collapsing what was previously a multi-step sequence into one efficient operation. The use of inexpensive and abundant carbon monoxide gas not only aligns with green chemistry principles by improving atom economy but also drastically reduces the raw material costs associated with specialized acylating agents. This streamlined process ensures higher overall efficiency and provides a robust platform for the rapid generation of diverse indanone libraries for drug discovery and development programs.
Mechanistic Insights into Pd-Catalyzed Multicomponent Carbonylation
The success of this synthetic methodology relies on a sophisticated palladium catalytic cycle that orchestrates the sequential formation of multiple carbon-carbon bonds with high precision. The mechanism initiates with the oxidative addition of the palladium catalyst, specifically palladium chloride in the presence of the tetrabutylammonium bromide additive, into the carbon-iodine bond of the o-bromoiodobenzene substrate. This selective activation is crucial as it distinguishes between the iodine and bromine atoms on the aromatic ring, setting the stage for the subsequent migratory insertion of the alkyne moiety. Following the alkyne insertion, the carbon monoxide molecule inserts into the palladium-carbon bond, introducing the carbonyl functionality directly into the growing molecular framework. The cycle concludes with an intramolecular reductive elimination that closes the five-membered ring, regenerating the active palladium species and releasing the desired 2,3-disubstituted indenone product. This elegant mechanistic pathway ensures that the reaction proceeds with high fidelity, minimizing side reactions and maximizing the yield of the target scaffold.
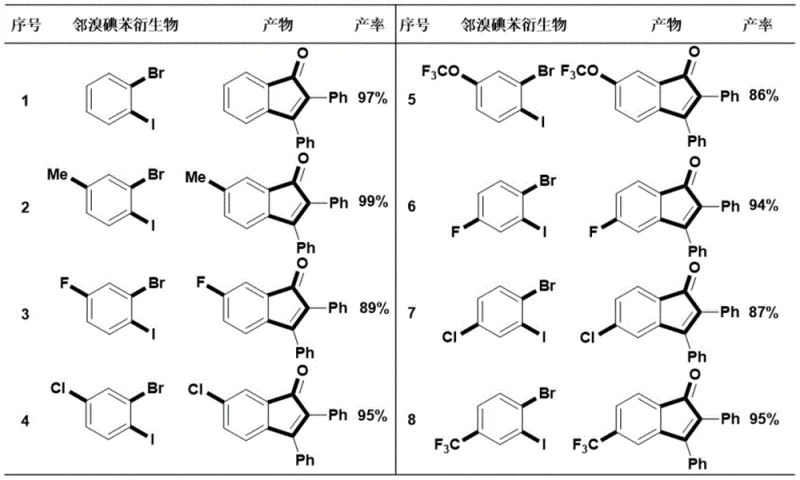
Beyond the core mechanism, the robustness of this catalytic system is evidenced by its remarkable tolerance towards a wide array of functional groups, which is a critical parameter for pharmaceutical process chemistry. The reaction conditions are sufficiently mild to accommodate electron-donating substituents such as methyl and methoxy groups, as well as electron-withdrawing groups like fluoro, chloro, and trifluoromethyl moieties without compromising the reaction efficiency. This broad substrate scope implies that the impurity profile remains manageable even when synthesizing complex derivatives, as the catalyst does not promote unwanted side reactions like dehalogenation or polymerization of the alkyne. For quality control teams, this predictability translates to easier purification processes, often achievable through standard column chromatography or crystallization techniques. The ability to maintain high yields across such a diverse set of substrates underscores the versatility of this method, making it an ideal candidate for the late-stage functionalization of advanced intermediates where structural complexity is already high.
How to Synthesize 2,3-Diarylindenone Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, operators must adhere to specific procedural guidelines that ensure safety and reproducibility while maximizing yield. The process begins with the careful preparation of the reaction vessel, typically a Schlenk tube, which must be thoroughly dried to prevent moisture from deactivating the palladium catalyst or interfering with the base. The precise stoichiometry of reagents, including the o-bromoiodobenzene, dialkylacetylene, palladium chloride catalyst, tetrabutylammonium bromide additive, and sodium carbonate base, is critical for driving the reaction to completion. Once the reagents are loaded, the atmosphere must be rigorously exchanged from air to carbon monoxide to establish the necessary environment for the carbonylation step, followed by heating at 100°C for 24 hours to facilitate the coupling. Detailed standardized operating procedures for this synthesis are provided below to guide technical teams through the execution of this high-value transformation.
- Combine dialkylacetylene, PdCl2 catalyst, TBAB additive, and Na2CO3 base in a dry Schlenk tube, then purge with carbon monoxide.
- Add o-bromoiodobenzene and anhydrous 1,4-dioxane under 1 atm CO atmosphere, then heat the mixture to 100°C for 24 hours.
- Quench the reaction with saturated ammonium chloride, extract with ethyl acetate, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing and supply chain perspective, the adoption of this palladium-catalyzed carbonylation method offers profound advantages that extend well beyond simple chemical yield improvements. The primary economic driver is the substitution of expensive, custom-synthesized carbonyl precursors with commodity-grade carbon monoxide gas, which is widely available and cost-effective on an industrial scale. This shift fundamentally alters the cost structure of indanone manufacturing, removing the margin layers associated with multi-step precursor synthesis and allowing procurement managers to negotiate better terms for bulk raw materials. Furthermore, the simplification of the synthetic route from multiple steps to a single pot significantly reduces the consumption of solvents, energy, and labor hours, leading to a leaner and more agile production process. These efficiencies collectively contribute to a more resilient supply chain capable of responding rapidly to market demands for key pharmaceutical intermediates without the bottlenecks typical of complex linear syntheses.
- Cost Reduction in Manufacturing: The elimination of pre-functionalized carbonyl precursors removes a major cost center, as these specialized reagents often command high prices due to their complex synthesis and low commercial availability. By utilizing simple o-bromoiodobenzenes and alkynes alongside cheap carbon monoxide, the direct material costs are drastically lowered, enabling significant savings in the overall cost of goods sold. Additionally, the one-pot nature of the reaction minimizes the need for intermediate isolation and purification steps, which are traditionally resource-intensive and prone to material loss. This consolidation of steps reduces the consumption of silica gel, solvents, and processing time, further enhancing the economic viability of the process for large-scale commercial production.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials such as substituted benzene derivatives and internal alkynes ensures a secure and consistent supply of inputs for manufacturing operations. Unlike proprietary intermediates that may be sourced from a single vendor with long lead times, the reagents for this process are commodity chemicals with robust global supply networks. This diversification of supply sources mitigates the risk of production delays caused by raw material shortages, providing supply chain heads with greater confidence in meeting delivery commitments. The stability of the reagents also simplifies inventory management, as they do not require specialized storage conditions or have short shelf lives that could lead to waste.
- Scalability and Environmental Compliance: The reaction operates under mild conditions of 100°C and atmospheric pressure of carbon monoxide, which greatly simplifies the engineering requirements for scaling up from laboratory to pilot and commercial plants. The absence of extreme temperatures or high-pressure equipment reduces capital expenditure on specialized reactors and enhances operational safety, making the technology accessible to a wider range of manufacturing facilities. Moreover, the high atom economy and reduced solvent usage align with increasingly stringent environmental regulations, lowering the burden of waste treatment and disposal. This environmental compatibility not only reduces compliance costs but also enhances the corporate sustainability profile of the manufacturing entity, which is increasingly important for partnerships with major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this indanone synthesis technology, derived directly from the experimental data and specifications outlined in the patent documentation. These insights are intended to clarify the practical aspects of the reaction for technical teams evaluating its adoption for their specific project needs. Understanding these nuances is essential for ensuring a smooth technology transfer and successful scale-up.
Q: What are the primary advantages of this indanone synthesis method over traditional Friedel-Crafts reactions?
A: Unlike traditional methods requiring complex pre-functionalized substrates and multi-step sequences, this patent describes a one-pot process that constructs three new C-C bonds simultaneously using cheap carbon monoxide as the carbonyl source.
Q: Does this catalytic system tolerate diverse functional groups on the aromatic rings?
A: Yes, the methodology demonstrates excellent functional group compatibility, successfully accommodating electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as fluoro, chloro, and trifluoromethyl substituents.
Q: Is the reaction condition suitable for large-scale industrial production?
A: The process utilizes mild conditions (100°C, 1 atm CO) and commercially available reagents like PdCl2 and sodium carbonate, making it highly scalable and economically viable for manufacturing pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indanone Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed carbonylation technology for the efficient production of high-purity indanone derivatives used in advanced pharmaceutical applications. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to full-scale manufacturing is seamless and risk-mitigated. Our state-of-the-art facilities are equipped to handle the specific safety and operational requirements of carbonylation reactions, including the safe management of carbon monoxide gas and the rigorous control of reaction parameters to meet stringent purity specifications. With our dedicated rigorous QC labs, we guarantee that every batch of indanone intermediate delivered meets the highest standards of quality and consistency required by global regulatory bodies.
We invite you to leverage our technical expertise to optimize your supply chain and reduce your overall manufacturing costs through the adoption of this superior synthetic route. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements and project timelines. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can support your long-term strategic goals in the development of next-generation therapeutic agents.
