Advanced Stereoselective Synthesis of Gemcitabine Hydrochloride for Commercial Scale-Up
Introduction to Advanced Nucleoside Manufacturing
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical oncology therapeutics, particularly for complex nucleoside analogs like 2'-deoxy-2',2'-difluoro-beta-cytidine hydrochloride, widely known as Gemcitabine Hydrochloride. Patent CN101381387A discloses a groundbreaking stereoselective synthesis method that addresses longstanding challenges in producing this vital active pharmaceutical ingredient (API). The structural complexity of Gemcitabine, characterized by its difluoro substitution at the 2' position of the ribofuranose ring, demands precise stereochemical control to ensure therapeutic efficacy and safety. 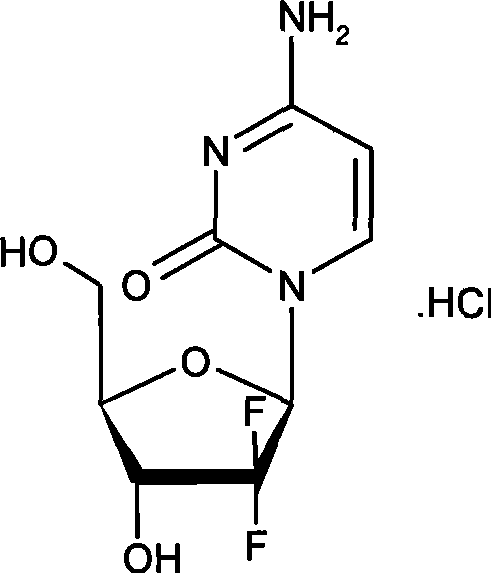 Traditional synthetic routes often struggle with low yields, poor stereoselectivity, or the requirement for hazardous and expensive reagents, creating bottlenecks in the global supply chain. This new methodology offers a transformative approach by optimizing the glycosylation step through a unique kinetic resolution strategy, thereby enhancing both the economic viability and the environmental profile of large-scale production.
Traditional synthetic routes often struggle with low yields, poor stereoselectivity, or the requirement for hazardous and expensive reagents, creating bottlenecks in the global supply chain. This new methodology offers a transformative approach by optimizing the glycosylation step through a unique kinetic resolution strategy, thereby enhancing both the economic viability and the environmental profile of large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing Gemcitabine have been plagued by significant technical and economic hurdles that hinder efficient industrial application. For instance, earlier patents such as US5371230 and US5401838 describe routes involving the activation of the 1-position hydroxyl group to form methanesulfonate intermediates, which notoriously require reaction temperatures lower than -80°C to achieve acceptable stereoselectivity. Maintaining such cryogenic conditions on a multi-ton scale is energetically prohibitive and introduces substantial operational risks and equipment costs for manufacturing facilities. Furthermore, alternative methods utilizing 1-halo ribofuranose intermediates, as seen in US5744597, often result in a beta-to-alpha anomer ratio of no more than 2:1, necessitating complex and yield-reducing purification steps to isolate the therapeutically active beta-isomer. 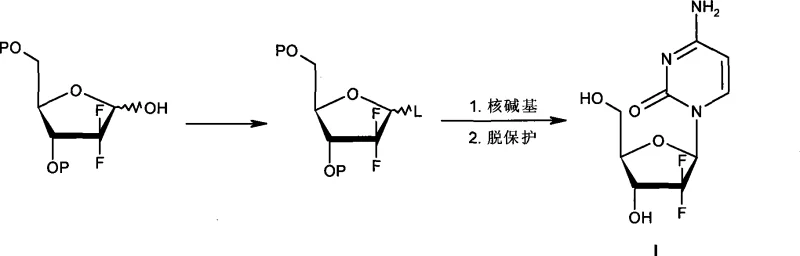 Other sophisticated routes reported in documents like WO2006070985 attempt to improve selectivity but rely on exorbitantly expensive reagents such as diphenoxy phosphoryl chloride and require excessive molar equivalents of the nucleobase, sometimes up to 14 equivalents, which drastically inflates raw material costs and waste generation.
Other sophisticated routes reported in documents like WO2006070985 attempt to improve selectivity but rely on exorbitantly expensive reagents such as diphenoxy phosphoryl chloride and require excessive molar equivalents of the nucleobase, sometimes up to 14 equivalents, which drastically inflates raw material costs and waste generation.
The Novel Approach
The innovative process detailed in CN101381387A circumvents these obstacles by introducing a streamlined workflow that eliminates the need for intermediate isolation and extreme temperature control. Instead of striving for a pure single isomer at the halogenation stage, this method cleverly exploits the differential reaction rates of the alpha and beta methanesulfonate isomers when treated with sodium halide. The process generates a mixture containing the desired alpha-halide alongside unreacted alpha-methanesulfonate, which is then subjected directly to the glycosylation reaction with silylated cytosine without any intervening purification. This "telescoped" approach not only simplifies the operational procedure but also significantly improves the overall conversion rate of raw materials by preventing losses typically associated with multiple isolation steps. By operating at moderate temperatures between 90°C and 135°C during the coupling phase, the method ensures high reproducibility and safety, making it exceptionally well-suited for the rigorous demands of commercial API manufacturing.
Mechanistic Insights into Kinetic Resolution and Glycosylation
The core chemical ingenuity of this synthesis lies in the kinetic resolution observed during the initial halogenation step, where the beta-methanesulfonate precursor reacts nearly twice as fast as its alpha counterpart. When the starting mixture of alpha and beta methanesulfonates is treated with a sodium halide, such as sodium bromide, in the presence of a quaternary amine phase-transfer catalyst, the beta-isomer is rapidly converted into the alpha-halide species. Monitoring this reaction via HPLC allows operators to halt the process precisely when the beta-methanesulfonate has been substantially consumed, leaving behind a mixture enriched with the alpha-halide and the original alpha-methanesulfonate. 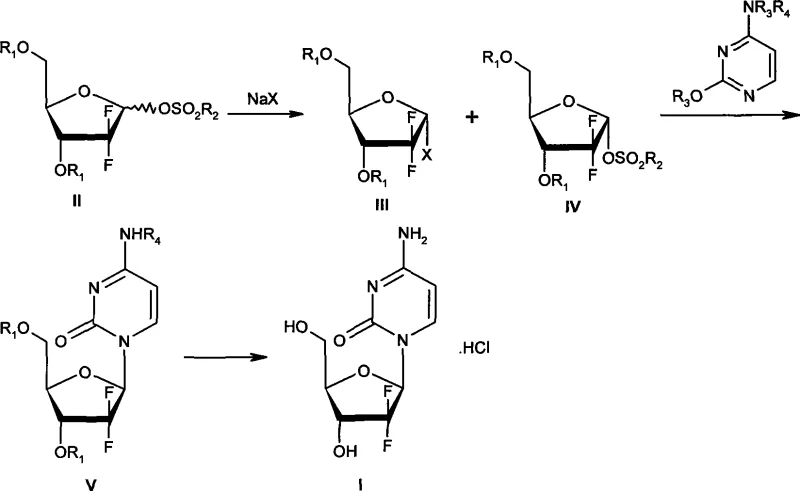 This specific composition of the intermediate mixture is crucial because the unreacted alpha-methanesulfonate does not interfere detrimentally with the subsequent nucleophilic substitution; instead, it can participate or remain inert without compromising the stereochemical outcome of the final nucleoside. The subsequent glycosylation involves the attack of the silylated cytosine on the anomeric carbon, facilitated by the specific solvent system of alkanes and high-boiling ethers, which promotes the formation of the beta-N-glycosidic bond with high fidelity.
This specific composition of the intermediate mixture is crucial because the unreacted alpha-methanesulfonate does not interfere detrimentally with the subsequent nucleophilic substitution; instead, it can participate or remain inert without compromising the stereochemical outcome of the final nucleoside. The subsequent glycosylation involves the attack of the silylated cytosine on the anomeric carbon, facilitated by the specific solvent system of alkanes and high-boiling ethers, which promotes the formation of the beta-N-glycosidic bond with high fidelity.
Impurity control is inherently built into this mechanism through the selective reactivity and the specific workup procedures described in the patent embodiments. The use of phase-transfer catalysts like tetrabutyl ammonium bromide ensures homogeneous reaction conditions that minimize side reactions such as elimination or degradation of the sensitive difluoro sugar moiety. Furthermore, the final deprotection and salification steps utilize saturated ammonia in methanol followed by careful pH adjustment with hydrochloric acid, which effectively removes protecting groups while inducing crystallization of the pure beta-anomer. The result is a product with a beta-isomer content exceeding 99.8% and alpha-isomer content below 0.1%, meeting the stringent purity specifications required by pharmacopeial standards like USP29 without the need for preparative chromatography. This high level of stereochemical purity is achieved because the reaction pathway thermodynamically and kinetically favors the formation of the beta-configuration during the coupling and crystallization phases.
How to Synthesize Gemcitabine Hydrochloride Efficiently
Implementing this synthesis requires careful attention to the stoichiometry of the halogenating agent and the precise monitoring of the reaction progress to maximize the yield of the key alpha-halide intermediate. The process begins with the reaction of the methanesulfonate mixture with sodium halide in a solvent such as tetrahydrofuran or DMF, followed by the direct addition of the crude mixture to the silylated base. Detailed standardized operating procedures regarding temperature ramps, addition rates, and crystallization parameters are essential to replicate the high success rates reported in the patent examples.
- React the mixture of alpha/beta methanesulfonates with sodium halide and a phase-transfer catalyst to selectively convert the beta-isomer into the alpha-halide.
- Directly couple the resulting crude mixture containing the alpha-halide and unreacted alpha-methanesulfonate with silylated cytosine at elevated temperatures.
- Perform deprotection using saturated ammonia in methanol followed by salification with hydrochloric acid to crystallize the high-purity beta-anomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers profound strategic advantages by fundamentally altering the cost structure and risk profile of Gemcitabine production. The elimination of cryogenic cooling requirements removes a major capital expenditure barrier, allowing manufacturers to utilize standard reactor vessels rather than specialized low-temperature equipment, which significantly lowers the barrier to entry for contract manufacturing organizations. Additionally, the ability to skip the isolation and purification of the unstable alpha-halide intermediate reduces the total number of processing steps, leading to a shorter manufacturing cycle time and reduced labor costs per kilogram of finished API. The reduction in the molar equivalent of the nucleobase required, from 14 equivalents in some prior art down to 10 equivalents in this method, directly translates to substantial savings in raw material procurement, especially given the high cost of specialized heterocyclic bases.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the simplification of the workflow and the use of commodity chemicals rather than exotic reagents. By avoiding the use of expensive phosphorylating agents and eliminating the need for energy-intensive low-temperature operations, the overall utility and material costs are drastically reduced. The telescoping of the halogenation and glycosylation steps means that solvents and reagents are utilized more efficiently, minimizing waste disposal costs and maximizing the throughput of existing production assets. This lean manufacturing approach ensures that the final cost of goods sold (COGS) is optimized, providing a competitive edge in the generic oncology market.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the robustness of the reaction conditions, which are less susceptible to minor fluctuations in temperature or mixing compared to cryogenic processes. The use of stable, commercially available starting materials like sodium bromide and standard protecting groups ensures that the supply chain is not vulnerable to shortages of niche specialty chemicals. Furthermore, the high conversion rates and minimal need for reprocessing batches due to failed purifications mean that production schedules are more predictable and lead times for customers can be consistently met. This stability is critical for pharmaceutical companies managing just-in-time inventory for life-saving cancer medications.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial tonnage is straightforward because it avoids the engineering complexities associated with maintaining -80°C across large reactor volumes. The solvent systems employed, such as heptane and phenyl ether mixtures, are manageable within standard solvent recovery units, facilitating compliance with environmental regulations regarding volatile organic compound (VOC) emissions. The reduction in chemical waste generated by skipping purification steps aligns with green chemistry principles, helping manufacturers meet increasingly strict sustainability goals and reducing the environmental footprint of their API production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and validation of this specific synthetic pathway for Gemcitabine Hydrochloride. These answers are derived directly from the experimental data and claims presented in the patent documentation to provide clarity on the method's feasibility and performance characteristics. Understanding these nuances is vital for technical teams evaluating the transfer of this technology to their own manufacturing sites.
Q: How does this method improve stereoselectivity compared to traditional routes?
A: This method leverages kinetic resolution during the halogenation step, where the beta-methanesulfonate reacts significantly faster than the alpha-isomer. By stopping the reaction at the optimal point, a mixture rich in the desired alpha-halide precursor is obtained, which subsequently yields a high ratio of the beta-nucleoside (greater than 99.8%) without requiring difficult chromatographic separations.
Q: Does this process require cryogenic conditions like -80°C?
A: No, one of the primary advantages of this patented technology is the elimination of extreme cryogenic conditions. Unlike prior art methods that necessitate temperatures below -80°C to control stereochemistry, this process operates effectively at room temperature or under reflux conditions, drastically reducing energy consumption and equipment complexity.
Q: What are the cost benefits of not separating the intermediate?
A: By utilizing the crude mixture of the alpha-halide and unreacted alpha-methanesulfonate directly in the glycosylation step, the process eliminates an entire purification unit operation. This reduces solvent usage, minimizes material loss associated with isolation, and shortens the overall cycle time, leading to significant operational expenditure savings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gemcitabine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner for the supply of high-quality oncology intermediates like Gemcitabine Hydrochloride. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global pharmaceutical markets with consistency. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch meets or exceeds the requirements set forth by international pharmacopeias. Our commitment to technical excellence allows us to navigate the complexities of nucleoside chemistry effectively, delivering products that support the uninterrupted treatment of patients worldwide.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can align with your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall procurement expenses. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that our solutions are perfectly tailored to support your drug development and commercialization timelines.
