Advanced Manufacturing of Imatinib: A Safe and Cost-Effective Synthetic Route for Global Pharma Supply Chains
Advanced Manufacturing of Imatinib: A Safe and Cost-Effective Synthetic Route for Global Pharma Supply Chains
The pharmaceutical industry is constantly seeking robust, scalable, and economically viable pathways for the production of critical oncology therapeutics such as Imatinib. Patent CN102382100A introduces a transformative preparation method that addresses long-standing safety and cost inefficiencies inherent in legacy manufacturing processes. By utilizing N-(3-guanidino-4-methylphenyl)-4-(4-methyl-piperazine-1-methyl) benzamide and 3-dimethylamino-1-(3-pyridyl)-2-propene-1-one as key intermediates, this novel approach circumvents the need for hazardous halogenating agents and precious metal catalysts. For R&D directors and procurement managers alike, this represents a significant opportunity to optimize the supply chain for this high-value API intermediate. The method not only ensures safer preparation conditions but also delivers a product that requires no cis-trans isomer screening, thereby streamlining the entire production lifecycle from raw material sourcing to final crystallization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Imatinib, such as those disclosed in patents like WO2006/042362A and CN1630648A, often rely on chemically aggressive and operationally dangerous reagents that pose substantial risks to both personnel and the environment. For instance, certain prior art methods utilize phosphoryl chloride (POCl3) during the synthesis of pyrimidine intermediates, a substance known for its high toxicity and corrosive nature, which necessitates stringent environmental controls and specialized waste treatment infrastructure. Furthermore, other established pathways depend on the use of trimethylaluminium for ammonolysis reactions; this reagent is pyrophoric and reacts violently with water, creating severe safety hazards that complicate scale-up operations in standard chemical plants. Additionally, processes employing palladium catalysis introduce the burden of removing trace heavy metals to meet rigorous pharmaceutical purity standards, adding complex purification steps that inflate both time and cost. These conventional methods often result in the formation of isomeric impurities, such as the 10% isomer noted in some literature, which are difficult to separate and can compromise the quality of the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast, the methodology outlined in CN102382100A offers a streamlined and inherently safer alternative that leverages readily available starting materials like 4-methyl-3-nitroaniline. This innovative route bypasses the need for toxic halogenation and pyrophoric alkylation by employing a sequence of condensation, reduction, and cyclization reactions that proceed under mild and controllable conditions. The process utilizes common organic solvents such as dioxane, ethanol, and isopropanol, which are easy to handle, store, and recover, significantly reducing the operational complexity associated with solvent management. By avoiding the use of precious metal catalysts and hazardous reagents, this new approach not only mitigates safety risks but also simplifies the regulatory compliance landscape for manufacturers. The elimination of cis-trans isomer formation is a particularly critical advancement, as it removes the need for resource-intensive chromatographic separations, allowing for a more direct and efficient path to high-purity Imatinib suitable for commercial distribution.
Mechanistic Insights into the Multi-Step Synthesis of Imatinib
The core of this patented technology lies in a carefully orchestrated five-step sequence that builds the complex Imatinib scaffold with high precision and yield. The synthesis initiates with the condensation of 4-methyl-3-nitroaniline with chloromethyl benzoyl chloride in the presence of anhydrous potassium carbonate, forming the nitro-amide backbone. This is followed by a crucial reduction step where hydrazine hydrate is employed to convert the nitro group into a primary amine, a transformation that proceeds efficiently in ethanol under reflux without the generation of heavy metal waste. Subsequently, the amino intermediate undergoes guanidinylation with cyanamide under acidic conditions to install the essential guanidine moiety, followed by neutralization and crystallization to isolate the key intermediate with high purity. The final stage involves the reaction of this guanidine derivative with 3-dimethylamino-1-(3-pyridyl)-2-propene-1-one, an enaminone species prepared separately from 3-acetylpyridine and DMF-DMA, to close the pyrimidine ring and complete the Imatinib structure.
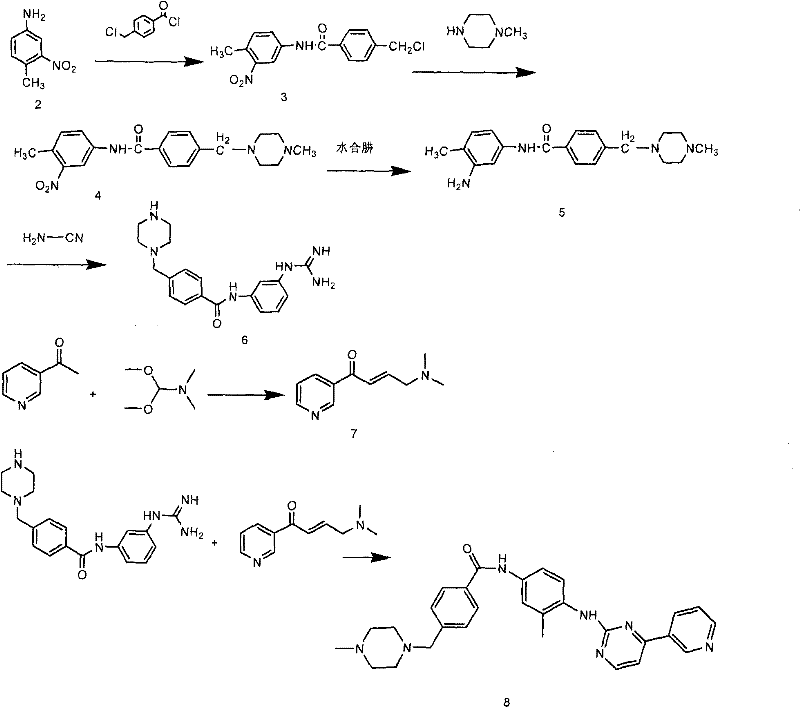
From a mechanistic perspective, the control of reaction parameters is vital to ensuring the high selectivity observed in this process. The reduction step using hydrazine hydrate is particularly noteworthy for its ability to selectively reduce the nitro group while leaving the amide linkage intact, a selectivity that is often challenging to achieve with catalytic hydrogenation methods that might inadvertently reduce other sensitive functional groups. Furthermore, the cyclization step is conducted in isoamyl alcohol with water removal, driving the equilibrium towards the formation of the desired pyrimidine ring and minimizing the formation of open-chain byproducts. The purification strategy, which involves conversion to the mesylate salt followed by recrystallization from ethanol, effectively removes residual organic impurities and ensures the final product meets the stringent melting point and HPLC purity specifications required for pharmaceutical applications. This level of control over the reaction pathway underscores the robustness of the method for large-scale manufacturing.
How to Synthesize Imatinib Efficiently
Implementing this synthesis route requires strict adherence to the specified stoichiometry and temperature profiles to maximize yield and minimize impurity formation. The process is designed to be scalable, moving seamlessly from laboratory benchtop quantities to multi-ton production campaigns with consistent quality outcomes. Operators must pay close attention to the exothermic nature of the initial acylation and the subsequent guanidinylation steps, ensuring adequate cooling and stirring to maintain reaction homogeneity. The detailed standardized synthesis steps, including specific reagent grades, addition rates, and workup procedures, are outlined in the comprehensive guide below to assist technical teams in replicating this high-efficiency protocol.
- Condense 4-methyl-3-nitroaniline with chloromethyl benzoyl chloride and N-methylpiperazine to form the nitro-amide intermediate.
- Reduce the nitro group to an amino group using hydrazine hydrate in ethanol under reflux conditions.
- React the amino intermediate with cyanamide to form the guanidine derivative, followed by cyclization with 3-dimethylamino-1-(3-pyridyl)-2-propene-1-one.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling economic and logistical benefits that directly impact the bottom line. By shifting away from reagents that are subject to strict regulatory controls or volatile market pricing, such as palladium catalysts and specialized halogenating agents, manufacturers can achieve a more stable and predictable cost structure. The reliance on commodity chemicals like 4-methyl-3-nitroaniline and common solvents ensures a reliable supply of raw materials, reducing the risk of production delays caused by sourcing bottlenecks. Furthermore, the simplified purification process, which avoids complex isomer separation, translates into shorter batch cycles and higher throughput, allowing facilities to respond more agilely to market demand fluctuations without compromising on product quality or safety standards.
- Cost Reduction in Manufacturing: The most significant financial advantage of this method stems from the complete elimination of expensive precious metal catalysts and the avoidance of hazardous reagents that require specialized disposal protocols. By utilizing cost-effective reagents like hydrazine hydrate and cyanamide, the direct material cost per kilogram of Imatinib is substantially lowered compared to prior art methods. Additionally, the high yields reported in the patent embodiments, such as the 90% yield in the final cyclization step, mean that less raw material is wasted, further enhancing the overall economic efficiency of the process. The reduction in waste treatment costs, due to the absence of heavy metals and toxic halogens, adds another layer of savings that improves the total cost of ownership for the manufacturing facility.
- Enhanced Supply Chain Reliability: The use of widely available starting materials ensures that the supply chain is resilient against disruptions that often plague the sourcing of specialty fine chemicals. Since the process does not depend on single-source suppliers for exotic catalysts or reagents, procurement teams can diversify their vendor base and negotiate more favorable terms. The robustness of the reaction conditions, which tolerate a range of temperatures between 0°C and 100°C, also means that the process is less sensitive to minor variations in utility supply, ensuring consistent production schedules even in facilities with varying infrastructure capabilities. This reliability is crucial for maintaining continuous supply to downstream formulation partners and meeting contractual delivery obligations.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is significantly greener, facilitating easier permitting and compliance with increasingly stringent global environmental regulations. The absence of pyrophoric reagents like trimethylaluminium reduces the need for specialized fire suppression systems and inert atmosphere handling equipment, lowering capital expenditure for plant upgrades. The solvents used, such as ethanol and isopropanol, are biodegradable and easier to recycle, supporting sustainability goals and reducing the carbon footprint of the manufacturing operation. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the manufacturer, making it a more attractive partner for global pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Imatinib synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the practical aspects of adopting this technology. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing production portfolios.
Q: How does this new method improve safety compared to traditional Imatinib synthesis?
A: This method eliminates the use of highly toxic phosphoryl chloride (POCl3) and pyrophoric trimethylaluminium, replacing them with safer reagents like hydrazine hydrate and cyanamide under mild temperature conditions (0-100°C).
Q: Does this process require expensive purification steps for isomers?
A: No, a key advantage of this patent (CN102382100A) is that the reactants do not require cis-trans screening, significantly simplifying the downstream purification process and reducing overall production time.
Q: What are the primary cost drivers eliminated in this synthetic route?
A: The process avoids the use of precious metal palladium catalysts and complex halogenation agents, relying instead on commodity chemicals and common solvents like ethanol and dioxane, which drastically lowers the raw material cost profile.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imatinib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner who can deliver high-quality intermediates with consistent reliability. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and efficiency. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Imatinib intermediate we produce meets the highest international standards for safety and efficacy. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions required by this novel synthesis route, providing a seamless transition from development to full-scale manufacturing.
We invite you to collaborate with us to explore how this advanced preparation method can optimize your supply chain and reduce your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic goals for reliable API intermediate sourcing.
