Optimizing Imatinib Production: A Technical Analysis of High-Purity Synthesis and Commercial Scalability
The pharmaceutical landscape for oncology treatments demands rigorous adherence to purity standards, particularly for critical tyrosine kinase inhibitors like Imatinib. The patent documentation CN101899035B outlines a transformative preparation method designed to overcome the persistent limitations of legacy synthesis routes. This technical disclosure details a robust amidation coupling strategy that consistently delivers product purity exceeding 99.5%, with single impurity levels rigorously controlled below 0.10%. For R&D directors and procurement specialists evaluating supply chain resilience, this methodology represents a significant leap forward in process chemistry. By integrating efficient solvent recovery systems and a novel crystallization protocol, the process addresses both the economic and regulatory pressures facing modern API manufacturing. The ability to produce high-purity Imatinib with reduced environmental impact positions this technology as a cornerstone for reliable API intermediate supplier networks aiming to meet global pharmacopeial standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to Imatinib synthesis, as referenced in prior art such as EP0564409 and various US patents, have struggled with inherent inefficiencies that compromise commercial viability. Traditional protocols frequently rely on excessive volumes of pyridine solvent, often utilizing ratios as high as 40:1 relative to the substrate mass, without effective mechanisms for solvent recovery. This not only inflates the cost reduction in pharmaceutical manufacturing but also creates substantial environmental burdens due to hazardous waste generation. Furthermore, the resulting crude products from these conventional routes typically exhibit purity levels hovering between 97% and 98%, failing to meet the stringent ICH Q3A requirements for bulk drug substances which mandate purity greater than 99.0%. The presence of unquantified foreign matter and high residual solvent content necessitates additional, costly purification steps that delay time-to-market and complicate the commercial scale-up of complex polymer additives or pharmaceutical intermediates.
The Novel Approach
In stark contrast, the innovative method described in the patent data introduces a streamlined workflow that optimizes both reaction efficiency and downstream processing. By employing a precise molar ratio of reactants and utilizing acetonitrile or recyclable pyridine as solvents, the process drastically simplifies the operational complexity. The introduction of a specific work-up procedure involving water addition, ethyl acetate extraction, and controlled pH neutralization allows for the effective removal of by-products before the final crystallization step. This approach ensures that the final Imatinib crystals precipitate with exceptional clarity and structural integrity. The elimination of cumbersome purification stages translates directly into substantial cost savings and a more sustainable manufacturing footprint. For supply chain heads, this means reducing lead time for high-purity pharmaceutical intermediates while ensuring a consistent supply of material that requires minimal reprocessing before formulation.
Mechanistic Insights into Amidation Coupling and Crystallization
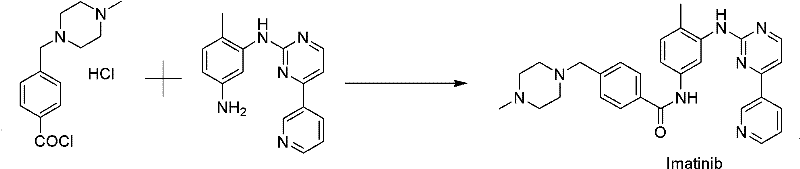
The core of this synthesis lies in the nucleophilic substitution reaction between 4-[(4-methyl-1-piperazinyl)methyl]benzoyl chloride dihydrochloride and N-(5-amino-2-methylphenyl)-4-(3-pyridyl)-2-pyrimidinamine. The reaction is facilitated by triethylamine, which acts as an acid-binding agent to neutralize the hydrochloride salt and drive the amidation forward. Maintaining the reaction temperature within the range of -5 to 50°C is critical for controlling the kinetics of the coupling and preventing the formation of thermal degradation by-products. The choice of solvent plays a pivotal role in solubilizing the reactants while maintaining a homogeneous reaction mixture that favors the formation of the desired amide bond. This careful control of reaction parameters ensures high conversion rates, as evidenced by yields reaching up to 85% in optimized embodiments, providing a robust foundation for industrial application.
Following the coupling reaction, the purification mechanism employs a sophisticated liquid-liquid extraction and pH-swing crystallization strategy. The removal of the solvent under reduced pressure allows for immediate recycling, aligning with green chemistry principles. Subsequent addition of water and extraction with ethyl acetate effectively partitions lipophilic impurities into the organic phase, leaving the desired product in the aqueous layer. The critical step involves neutralizing the aqueous phase to a pH of 9-10 using ammonia water, which deprotonates the molecule and reduces its solubility. The addition of saturated fatty alcohol then induces supersaturation, prompting the nucleation and growth of high-purity Imatinib crystals. This precise control over the crystallization environment is what enables the suppression of single impurities to levels below 0.10%.
How to Synthesize Imatinib Efficiently
Implementing this synthesis route requires strict adherence to the stoichiometric ratios and temperature controls defined in the patent specifications to ensure reproducibility at scale. The process begins with the preparation of the reaction vessel under inert atmosphere, followed by the sequential addition of the amine precursor and the acid chloride derivative in the presence of the base. Operators must monitor the reaction progress via HPLC to determine the exact endpoint before proceeding to the solvent recovery and extraction phases. The detailed standardized synthetic steps see the guide below for a comprehensive breakdown of the operational parameters required to achieve the reported purity and yield metrics consistently.
- React 4-[(4-methyl-1-piperazinyl)methyl]benzoyl chloride dihydrochloride with N-(5-amino-2-methylphenyl)-4-(3-pyridyl)-2-pyrimidinamine in acetonitrile or pyridine at -5 to 50°C using triethylamine.
- Remove solvent under reduced pressure for recycling, add water to residues, and extract impurities using ethyl acetate to separate organic and aqueous phases.
- Neutralize the aqueous phase to pH 9-10 with ammonia water and add saturated fatty alcohol to precipitate high-purity Imatinib crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this optimized synthesis method offers tangible strategic benefits beyond mere technical specifications. The primary advantage lies in the significant reduction of raw material costs driven by the ability to recover and reuse solvents, which traditionally constitute a major portion of variable manufacturing expenses. By minimizing solvent consumption and eliminating the need for expensive chromatographic purification, the overall cost of goods sold is drastically lowered. This economic efficiency allows for more competitive pricing structures without compromising on the quality of the high-purity OLED material or pharmaceutical intermediate supplied to downstream partners. Additionally, the simplified workflow reduces the dependency on specialized equipment, thereby lowering capital expenditure requirements for facility upgrades.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the implementation of solvent recycling loops directly correlate to a leaner cost structure. By avoiding the use of heavy metals, the expensive and time-consuming processes associated with metal scavenging and validation are completely removed from the production line. This qualitative shift in process design ensures that resources are allocated towards value-added activities rather than waste management. Consequently, manufacturers can achieve substantial cost savings while maintaining a competitive edge in the global market for specialty chemicals.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply continuity by reducing the risk of batch failures associated with complex purification steps. The use of readily available starting materials and common solvents like acetonitrile ensures that raw material sourcing remains stable even during market fluctuations. Furthermore, the mild reaction conditions reduce the likelihood of safety incidents that could disrupt production schedules. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream drug manufacturers receive their materials on schedule to maintain their own production timelines.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with embodiments demonstrating successful scaling from gram to multi-kilogram quantities without loss of efficiency. The reduction in solvent waste and the absence of toxic heavy metals simplify the environmental compliance landscape, making it easier to obtain necessary regulatory approvals for expansion. This facilitates the commercial scale-up of complex pharmaceutical intermediates, allowing producers to rapidly respond to increased market demand. The eco-friendly nature of the process also aligns with corporate sustainability goals, enhancing the brand reputation of the supplier in an increasingly environmentally conscious industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-purity Imatinib synthesis method. These insights are derived directly from the patent data and are intended to clarify the operational advantages and quality benchmarks associated with this technology. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this method improve impurity profiles compared to conventional synthesis?
A: Conventional methods often yield purity between 97%-98% with significant residual solvents. This optimized protocol utilizes a specific extraction and pH-controlled crystallization sequence to achieve purity greater than 99.5% with single impurities below 0.10%, meeting strict ICH Q3A guidelines.
Q: What are the solvent advantages in this preparation method?
A: Unlike prior art requiring large volumes of non-recyclable pyridine, this method allows for the use of acetonitrile or pyridine which can be recovered and recycled after the reaction. This significantly reduces raw material consumption and environmental waste load.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction conditions are mild (-5 to 50°C) and the operation process is simplified without requiring complex chromatography. The high yield (up to 85%) and robust crystallization steps make it highly adaptable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imatinib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of delivering high-quality intermediates that meet the rigorous demands of the global pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to verify that every batch of Imatinib we produce adheres to the highest international standards. Our dedication to technical excellence allows us to support your R&D and commercialization efforts with confidence and reliability.
We invite you to collaborate with us to optimize your supply chain and achieve your production goals. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can enhance your project's success. Let us be your partner in driving innovation and efficiency in the production of life-saving medicines.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
