Revolutionizing Quinoline Production: A Metal-Free DMSO Participation Strategy for Scalable Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing nitrogen-containing heterocycles, particularly quinoline derivatives which serve as pivotal scaffolds in numerous bioactive molecules. A groundbreaking advancement in this domain is detailed in Chinese Patent CN114031552A, which discloses a novel method for synthesizing substituted quinoline derivatives through the direct participation of dimethyl sulfoxide (DMSO). This technology represents a paradigm shift from conventional transition-metal catalyzed routes, offering a metal-free, one-pot oxidative cyclization strategy that utilizes aromatic aldehydes and aromatic amines as primary starting materials. By leveraging DMSO not merely as a solvent but as an active carbon source, this invention addresses critical pain points regarding raw material availability, environmental impact, and process complexity. For R&D directors and procurement managers alike, this patent signals a potential transformation in how high-purity quinoline intermediates are sourced and manufactured, promising enhanced supply chain stability and significant reductions in production overheads.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of quinoline compounds has relied heavily on classical methodologies such as the Skraup synthesis or the Combes condensation, which often necessitate harsh reaction conditions including concentrated sulfuric acid and high temperatures, leading to severe safety hazards and substantial waste generation. Furthermore, modern catalytic approaches frequently depend on expensive and scarce transition metal catalysts, such as rare earth elements, copper trifluoromethanesulfonate, or iron complexes, which introduce significant cost burdens and complicate downstream purification due to strict regulatory limits on residual heavy metals in pharmaceutical ingredients. As illustrated in prior art reaction schemes, many existing methods require specialized substrates like unstable imines or electron-withdrawing alkynes, which are not only costly to procure but also limit the structural diversity of the final products. These conventional pathways often suffer from low atom economy, requiring stoichiometric amounts of oxidants and generating large volumes of toxic byproducts, thereby creating bottlenecks for sustainable commercial scale-up and increasing the overall cost reduction challenges in pharmaceutical intermediate manufacturing.
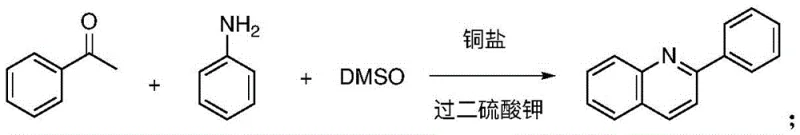
The Novel Approach
In stark contrast to these legacy processes, the technology disclosed in CN114031552A introduces a streamlined, metal-free protocol that utilizes ubiquitous and inexpensive reagents to construct the quinoline core with high efficiency. The core innovation lies in the dual role of dimethyl sulfoxide (DMSO), which functions simultaneously as a high-boiling polar solvent to enhance reaction kinetics and as a unique C2 synthon that donates two carbon atoms to the forming pyridine ring. This approach eliminates the dependency on precious metal catalysts and exotic substrates, replacing them with common aromatic aldehydes, aromatic amines, potassium peroxydisulfate, and alkali bases like potassium tert-butoxide. The reaction proceeds smoothly under an air-containing atmosphere at moderate temperatures, typically around 150°C, facilitating a one-pot transformation that minimizes unit operations and solvent exchanges. By adopting this novel synthetic route, manufacturers can achieve high yields of polysubstituted quinolines while drastically simplifying the workup procedure, thus offering a reliable pharmaceutical intermediate supplier pathway that is both economically viable and environmentally compliant.
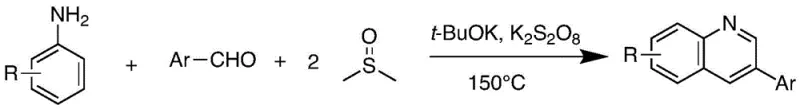
Mechanistic Insights into DMSO-Mediated Oxidative Cyclization
The mechanistic elegance of this transformation is rooted in a radical-mediated oxidative cyclization process that fundamentally redefines the utility of DMSO in organic synthesis. Detailed investigations, including deuterium labeling experiments, have confirmed that both the C2 and C4 positions of the resulting quinoline pyridine ring are derived directly from the dimethyl sulfoxide molecule, a finding that overturns previous assumptions about DMSO's inertness in such cyclizations. The proposed mechanism initiates with the base-promoted nucleophilic addition of DMSO to the aromatic aldehyde, generating an unsaturated sulfoxide intermediate which is subsequently oxidized by peroxydisulfate to form a reactive unsaturated sulfone species. Concurrently, the aromatic amine undergoes oxidation to generate a transient nitrene intermediate, which then engages in a [4+2] cycloaddition with the activated sulfone species. This cascade culminates in the formation of a stable dihydroquinoline intermediate that undergoes spontaneous elimination to yield the final aromatic quinoline product, demonstrating a sophisticated interplay of radical chemistry and pericyclic reactions that ensures high regioselectivity and product purity.
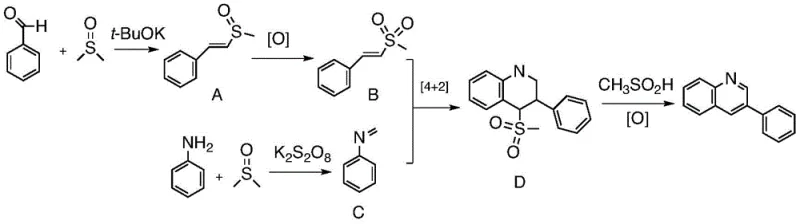
From an impurity control perspective, this metal-free mechanism offers distinct advantages by avoiding the formation of metal-complexed side products that are notoriously difficult to separate. The use of potassium peroxydisulfate as a terminal oxidant ensures that the reaction environment remains sufficiently oxidative to drive the cyclization to completion without over-oxidizing sensitive functional groups on the aromatic rings. The absence of transition metals means that the impurity profile is dominated by organic byproducts that can be effectively managed through standard crystallization or chromatographic techniques, rather than requiring specialized scavenging resins or complex extraction protocols. Furthermore, the mild basic conditions provided by alkali tert-butoxides help to suppress acid-catalyzed polymerization or decomposition pathways that often plague traditional acid-mediated quinoline syntheses. This results in a cleaner crude reaction mixture, reducing the burden on quality control laboratories and ensuring that the final high-purity pharmaceutical intermediates meet stringent regulatory specifications with minimal processing effort.
How to Synthesize Substituted Quinoline Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and thermal management to maximize yield and safety. The process is designed to be operationally simple, utilizing a one-pot configuration where all reagents are combined in a single vessel, thereby reducing the risk of material loss and exposure to hazardous intermediates. Operators should ensure that the reaction is conducted under an air-containing atmosphere, as oxygen plays a subtle but important role in the radical propagation steps, although the primary oxidant is the peroxydisulfate salt. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and workup procedures, are outlined below to guide technical teams in replicating this high-efficiency protocol.
- Dissolve aromatic aldehyde and aromatic amine in dimethyl sulfoxide (DMSO) within a reaction vessel under an air-containing atmosphere.
- Add potassium peroxydisulfate (oxidant) and potassium tert-butoxide (base) to the mixture to initiate the oxidative cyclization reaction.
- Heat the reaction mixture to 150°C for approximately 2 hours, then cool, extract with ethyl acetate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this DMSO-participation technology translates into tangible strategic benefits that extend far beyond simple yield improvements. The most significant advantage lies in the drastic simplification of the raw material supply chain, as the process relies on commodity chemicals like benzaldehyde, aniline derivatives, and DMSO, all of which are produced on a massive global scale with stable pricing and abundant availability. This contrasts sharply with traditional methods that may require custom-synthesized alkynes or unstable imines, which are subject to long lead times and volatile market pricing. By shifting to this robust platform, companies can mitigate supply chain risks associated with specialty reagents and ensure continuous production schedules even during periods of market disruption, thereby enhancing the overall resilience of the manufacturing operation.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts such as copper, iron, or rare earth elements removes a major cost center from the bill of materials, while simultaneously eradicating the downstream expenses associated with metal removal and validation. Without the need for specialized metal scavengers or extensive purification steps to meet residual metal limits, the overall processing time and solvent consumption are significantly reduced, leading to substantial cost savings in utilities and waste disposal. Furthermore, the high atom economy of using DMSO as a carbon source means that less mass is wasted as byproducts, optimizing the input-to-output ratio and driving down the cost per kilogram of the final active pharmaceutical ingredient intermediate.
- Enhanced Supply Chain Reliability: Utilizing widely available bulk chemicals ensures that production is not bottlenecked by the scarcity of niche catalysts or ligands, allowing for more flexible sourcing strategies and better negotiation leverage with vendors. The robustness of the reaction conditions, which tolerate air and moisture better than many sensitive organometallic processes, reduces the likelihood of batch failures due to environmental factors, thereby improving on-time delivery performance. This reliability is crucial for maintaining just-in-time inventory levels and meeting the rigorous delivery schedules demanded by downstream pharmaceutical clients, ultimately strengthening the supplier-client relationship through consistent performance.
- Scalability and Environmental Compliance: The one-pot nature of this synthesis minimizes the number of unit operations, making it inherently easier to scale from gram-scale laboratory experiments to multi-ton commercial production without complex engineering modifications. The avoidance of concentrated mineral acids and heavy metals aligns perfectly with modern green chemistry principles and increasingly stringent environmental regulations, reducing the regulatory burden and potential liability associated with hazardous waste management. This environmental compatibility not only lowers compliance costs but also enhances the corporate sustainability profile, which is becoming an increasingly important criterion for selection by major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this innovative synthesis method, providing clarity on its practical implementation and benefits. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, ensuring that stakeholders have accurate information for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into existing manufacturing portfolios.
Q: What is the primary advantage of using DMSO in this quinoline synthesis method?
A: Unlike traditional methods where DMSO acts solely as a solvent, this patent demonstrates that DMSO serves as a critical reaction substrate, providing two carbon atoms (C2 and C4) to the pyridine ring, thereby eliminating the need for expensive alkyne or ketone substrates.
Q: Does this process require expensive transition metal catalysts?
A: No, the method is entirely metal-free. It utilizes common inorganic oxidants like potassium peroxydisulfate and alkali bases like potassium tert-butoxide, which significantly reduces raw material costs and eliminates the need for complex heavy metal removal steps.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process operates under mild conditions (air atmosphere, 150°C) using widely available commercial reagents. The one-pot nature of the reaction simplifies operational procedures and waste treatment, making it highly favorable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Quinoline Supplier
As the global demand for complex heterocyclic building blocks continues to rise, partnering with a technically proficient CDMO partner is essential for translating innovative patent technologies into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of substituted quinoline derivative meets the highest international standards for pharmaceutical applications. We are committed to leveraging advanced synthetic methodologies, such as the DMSO-participation route, to deliver superior value to our clients through enhanced quality and operational excellence.
We invite forward-thinking pharmaceutical companies and research institutions to collaborate with us to explore the full potential of this metal-free quinoline synthesis platform. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific project needs, along with specific COA data and route feasibility assessments. Let us help you optimize your supply chain and accelerate your drug development timelines with our reliable Substituted Quinoline solutions, ensuring that your critical projects proceed without interruption or compromise on quality.
