Scalable Synthesis and Commercial Viability of Novel Chiral Zinc Nitrogen Complexes for Asymmetric Catalysis
Scalable Synthesis and Commercial Viability of Novel Chiral Zinc Nitrogen Complexes for Asymmetric Catalysis
The landscape of asymmetric catalysis is undergoing a significant transformation, driven by the urgent need for cost-effective and environmentally sustainable methodologies in the production of high-value pharmaceutical intermediates. Patent CN103012445A introduces a groundbreaking chiral zinc nitrogen complex, specifically a tetranuclear zinc cluster designated as tris{2-hydroxy-N[2-(2-hydroxyl-1-phenyl-ethylamino)-1-phenyl-ethyl]-6-methylnicotinamide}Zn complex. This innovation represents a pivotal shift away from reliance on scarce and expensive precious metals towards abundant base metals without compromising catalytic efficacy. For R&D directors and procurement strategists alike, this development offers a compelling avenue for optimizing synthetic routes. The patent details a robust preparation method involving the reflux condensation of 6-methyl-2-cyano-3-hydroxypyridine with D-phenylglycinol in the presence of anhydrous zinc chloride. This approach not only simplifies the ligand design but also ensures high structural integrity, as evidenced by single-crystal X-ray diffraction data. By leveraging this technology, manufacturers can potentially achieve substantial cost reduction in pharmaceutical intermediate manufacturing while maintaining rigorous purity standards required for drug substance production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of chiral intermediates has heavily relied on complexes derived from noble metals such as Ruthenium, Rhodium, or Iridium, often coordinated with sophisticated and costly chiral ligands like BINAP or specialized oxazolines. These conventional catalytic systems, while effective, present significant logistical and economic challenges for large-scale industrial applications. The primary bottleneck lies in the exorbitant cost of the metal precursors, which are subject to volatile market pricing and geopolitical supply constraints. Furthermore, the removal of trace heavy metal residues from the final active pharmaceutical ingredient (API) is a stringent regulatory requirement that necessitates additional purification steps, such as scavenging or recrystallization, thereby increasing processing time and waste generation. Additionally, many traditional ligands require multi-step syntheses themselves, adding layers of complexity and reducing the overall atom economy of the process. These factors collectively contribute to inflated production costs and extended lead times, creating friction in the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology outlined in CN103012445A utilizes a zinc-based coordination framework that fundamentally alters the economic equation of asymmetric catalysis. The novel approach employs D-phenylglycinol, a commercially abundant and inexpensive chiral pool building block, reacting it with a simple pyridine derivative to form a robust multidentate ligand in situ. This strategy eliminates the need for pre-synthesized, high-cost chiral ligands. The use of Zinc Chloride as the metal source further drives down material costs, as zinc is orders of magnitude cheaper than platinum group metals. The reaction proceeds under relatively standard conditions—reflux in chlorobenzene for 48 hours—making it highly compatible with existing reactor infrastructure found in most fine chemical plants. 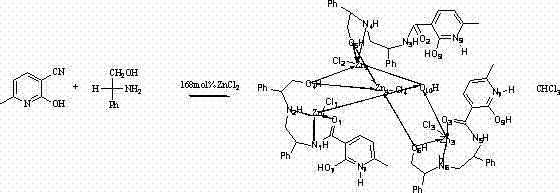 As illustrated in the reaction scheme, the formation of the complex is straightforward, yielding white crystals with a reported isolation efficiency of 75%. This simplicity translates directly into operational excellence, reducing the barrier to entry for commercial scale-up of complex pharmaceutical intermediates and offering a reliable alternative for suppliers seeking to diversify their catalytic portfolio.
As illustrated in the reaction scheme, the formation of the complex is straightforward, yielding white crystals with a reported isolation efficiency of 75%. This simplicity translates directly into operational excellence, reducing the barrier to entry for commercial scale-up of complex pharmaceutical intermediates and offering a reliable alternative for suppliers seeking to diversify their catalytic portfolio.
Mechanistic Insights into Zn-N Coordination and Catalytic Activity
Understanding the structural nuances of this chiral zinc complex is critical for R&D teams aiming to optimize its application in diverse synthetic transformations. The complex features a unique tetranuclear zinc core, where four zinc atoms are bridged by oxygen and nitrogen atoms from the organic ligands, creating a rigid chiral environment essential for enantioselective induction. 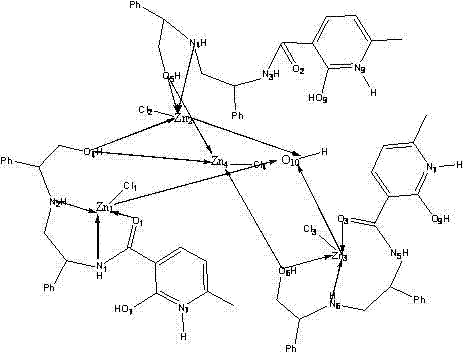 The crystallographic data reveals specific bond lengths, such as Zn-O distances ranging from 1.913 to 2.416 Angstroms and Zn-N distances around 2.128 to 2.155 Angstroms, indicating strong coordination that stabilizes the active catalytic species. This polynuclear architecture likely facilitates cooperative catalysis, where multiple metal centers work in concert to activate substrates. The chirality is imparted by the phenylglycinol moieties, which project steric bulk in a defined spatial arrangement, effectively differentiating between the prochiral faces of incoming substrates. This structural rigidity prevents non-selective background reactions, ensuring high optical purity of the products. For process chemists, this means that the catalyst can potentially be tuned by modifying the substituents on the pyridine ring or the amino alcohol component, offering a versatile platform for method development.
The crystallographic data reveals specific bond lengths, such as Zn-O distances ranging from 1.913 to 2.416 Angstroms and Zn-N distances around 2.128 to 2.155 Angstroms, indicating strong coordination that stabilizes the active catalytic species. This polynuclear architecture likely facilitates cooperative catalysis, where multiple metal centers work in concert to activate substrates. The chirality is imparted by the phenylglycinol moieties, which project steric bulk in a defined spatial arrangement, effectively differentiating between the prochiral faces of incoming substrates. This structural rigidity prevents non-selective background reactions, ensuring high optical purity of the products. For process chemists, this means that the catalyst can potentially be tuned by modifying the substituents on the pyridine ring or the amino alcohol component, offering a versatile platform for method development.
The catalytic utility of this complex extends beyond theoretical interest, demonstrating practical viability in key carbon-carbon bond-forming reactions. In the nitrile silylation of benzaldehyde, a fundamental reaction for generating alpha-hydroxy acids and amino acids, the complex exhibited remarkable performance. 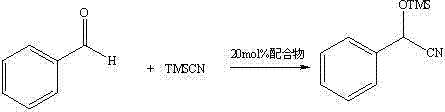 The patent data reports a transformation efficiency reaching 99%, which is indicative of a highly active Lewis acid catalyst capable of activating the carbonyl group of the aldehyde towards nucleophilic attack by trimethylsilyl cyanide. Furthermore, the complex showed activity in the Baylis-Hillman reaction, although with lower conversion rates in the initial screening, suggesting potential for optimization through solvent or temperature modulation. The ability to function effectively in these distinct mechanistic manifolds highlights the versatility of the zinc-nitrogen coordination sphere. For supply chain managers, this versatility implies that a single catalyst inventory could support multiple synthetic pathways, reducing the complexity of raw material management and minimizing the risk of stockouts for specialized reagents.
The patent data reports a transformation efficiency reaching 99%, which is indicative of a highly active Lewis acid catalyst capable of activating the carbonyl group of the aldehyde towards nucleophilic attack by trimethylsilyl cyanide. Furthermore, the complex showed activity in the Baylis-Hillman reaction, although with lower conversion rates in the initial screening, suggesting potential for optimization through solvent or temperature modulation. The ability to function effectively in these distinct mechanistic manifolds highlights the versatility of the zinc-nitrogen coordination sphere. For supply chain managers, this versatility implies that a single catalyst inventory could support multiple synthetic pathways, reducing the complexity of raw material management and minimizing the risk of stockouts for specialized reagents.
How to Synthesize Chiral Zinc Complex Efficiently
The preparation of this title complex is designed to be operationally simple, avoiding the need for glovebox techniques or ultra-dry conditions typically associated with sensitive organometallics, although anhydrous and anaerobic conditions are preferred for optimal results. The process begins with the combination of the pyridine precursor and the chiral amino alcohol in a chlorobenzene medium, followed by the addition of the zinc salt. The mixture is then subjected to prolonged heating to drive the equilibrium towards complex formation. Detailed standardized synthetic procedures, including exact stoichiometric ratios, temperature profiles, and workup protocols, are essential for reproducibility at the kilogram scale.
- Combine 6-methyl-2-cyano-3-hydroxypyridine and D-phenylglycinol with anhydrous ZnCl2 catalyst in chlorobenzene solvent under inert atmosphere.
- Maintain reflux conditions for 48 hours to ensure complete coordination and cyclization of the ligand framework around the zinc centers.
- Purify the crude mixture via solvent removal, aqueous dissolution, chloroform extraction, and final column chromatography to isolate white crystals.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this zinc-based catalytic technology offers profound advantages that extend well beyond the laboratory bench. The primary driver for adoption is the significant potential for cost reduction in manufacturing, achieved not merely through cheaper reagents but through a holistic simplification of the process flow. By eliminating the dependency on precious metals, companies can insulate themselves from the volatility of the PGM (Platinum Group Metals) market, leading to more predictable budgeting and long-term cost stability. Moreover, the reduced toxicity profile of zinc compared to heavy metals like palladium or chromium simplifies waste disposal protocols and lowers environmental compliance costs. This aligns perfectly with the growing industry mandate for greener chemistry practices, allowing manufacturers to market their intermediates as sustainably produced. The robustness of the catalyst also suggests a longer operational lifetime or the potential for recycling, further amortizing the initial investment over larger production volumes.
- Cost Reduction in Manufacturing: The substitution of expensive noble metal catalysts with abundant zinc salts results in a drastic decrease in direct material costs. Since zinc chloride is a commodity chemical available in bulk quantities at a fraction of the price of iridium or rhodium complexes, the bill of materials for the catalytic step is significantly lowered. Additionally, the ligand synthesis avoids complex multi-step organic synthesis, utilizing a one-pot condensation that maximizes atom economy. This efficiency reduces solvent consumption and energy usage per kilogram of product, contributing to substantial cost savings across the entire production lifecycle without compromising the quality of the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Sourcing high-purity chiral ligands and precious metal salts often involves long lead times and reliance on a limited number of global suppliers, creating vulnerability in the supply chain. In contrast, the precursors for this zinc complex, such as D-phenylglycinol and substituted pyridines, are widely produced by multiple chemical manufacturers globally. This abundance ensures a steady and reliable supply, mitigating the risk of production delays due to raw material shortages. For supply chain heads, this diversification of the supplier base is a critical risk mitigation strategy, ensuring continuity of supply for critical API intermediates even during periods of global logistical disruption or geopolitical tension.
- Scalability and Environmental Compliance: The synthesis protocol described in the patent utilizes chlorobenzene, a solvent with well-established handling and recovery protocols in the fine chemical industry, facilitating easy scale-up from gram to tonne quantities. The absence of air-sensitive reagents means that standard stainless steel reactors can be used without the need for specialized Hastelloy lining or rigorous exclusion of moisture, lowering capital expenditure requirements for new production lines. Furthermore, the lower toxicity of zinc residues simplifies the purification of the final product, reducing the burden on wastewater treatment facilities and ensuring easier compliance with increasingly stringent environmental regulations regarding heavy metal discharge in pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral zinc technology in industrial settings. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, offering a realistic view of the catalyst's capabilities and limitations. Understanding these nuances is vital for making informed decisions about process integration and vendor selection.
Q: What is the catalytic efficiency of this zinc complex in cyanosilylation reactions?
A: According to patent data, the complex demonstrates exceptional activity in the nitrile silylation of benzaldehyde, achieving transformation efficiencies exceeding 99% under optimized laboratory conditions.
Q: Is the synthesis of this chiral ligand scalable for industrial production?
A: Yes, the synthesis utilizes readily available starting materials like D-phenylglycinol and standard solvents like chlorobenzene, avoiding exotic reagents. The 75% isolated yield reported suggests a robust pathway amenable to scale-up.
Q: How does this zinc-based catalyst compare to traditional precious metal catalysts?
A: This complex replaces expensive transition metals like Ruthenium or Iridium with abundant Zinc. This substitution significantly lowers raw material costs and simplifies downstream heavy metal removal processes, enhancing supply chain sustainability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Zinc Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chiral zinc complex technology detailed in CN103012445A for the next generation of asymmetric synthesis. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate this patented laboratory methodology into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop to plant floor is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of chiral intermediate meets the exacting standards required by global regulatory bodies. Our commitment to quality ensures that the enantiomeric excess and chemical purity of the final product are consistently maintained, safeguarding the integrity of your downstream drug synthesis.
We invite forward-thinking pharmaceutical and agrochemical companies to collaborate with us to leverage this cost-effective catalytic solution. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific synthetic route, identifying exactly where this zinc technology can replace more expensive alternatives. We encourage you to reach out to request specific COA data from our pilot runs and comprehensive route feasibility assessments. Let us help you optimize your supply chain, reduce your carbon footprint, and accelerate your time to market with our reliable supply of high-performance chiral building blocks.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
