Revolutionizing Faropenem Sodium Production: A One-Pot Strategy for Industrial Scale-Up
Revolutionizing Faropenem Sodium Production: A One-Pot Strategy for Industrial Scale-Up
The pharmaceutical landscape for beta-lactam antibiotics continues to evolve, driven by the urgent need for more efficient and cost-effective manufacturing processes. Patent CN1314691C introduces a groundbreaking methodology for the preparation of Faropenem Sodium, a potent broad-spectrum penem antibiotic known for its stability against beta-lactamases. Unlike traditional synthetic routes that suffer from excessive operational complexity, this invention utilizes a streamlined "one-pot" strategy starting from the key chiral building block 4-AA. By eliminating the need for intermediate purification, the process not only enhances overall yield but also significantly reduces solvent waste and operational time. This technical insight report analyzes the mechanistic advantages and commercial implications of this novel route, positioning it as a critical asset for reliable faropenem sodium suppliers aiming to optimize their supply chains.
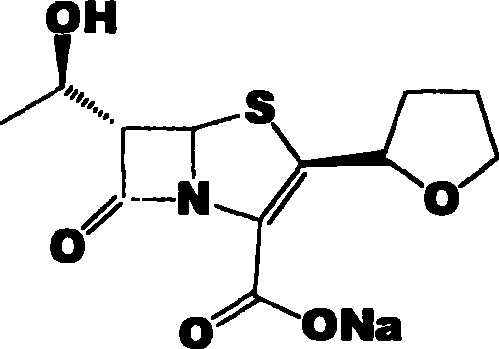
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Faropenem Sodium has been plagued by inefficiencies inherent in multi-step linear syntheses. Prior art, including patents such as US4997829 and EP410727, typically describes a lengthy 6-to-9 step sequence starting from 4-AA. In these conventional pathways, every chemical transformation necessitates a rigorous isolation and purification stage, often involving column chromatography or multiple recrystallizations. This approach creates a bottleneck in cost reduction in antibiotic manufacturing, as each isolation step incurs substantial material loss, leading to depressed overall yields. Furthermore, the accumulation of solvent usage for purification generates significant environmental burdens and increases the logistical complexity of waste management, making these older methods economically unsustainable for modern, high-volume production requirements.
The Novel Approach
The methodology disclosed in CN1314691C represents a paradigm shift by integrating multiple reaction steps into a continuous flow without intermediate isolation. The core innovation lies in the direct utilization of crude reaction mixtures; for instance, the condensation product of 4-AA and (R)-tetrahydrofuran-2-thioformic acid is concentrated and immediately subjected to acylation with allyloxy oxalyl chloride. This telescoping of reactions minimizes the exposure of sensitive intermediates to harsh workup conditions, thereby preserving stereochemical integrity. For procurement teams, this translates to a drastic simplification of the bill of materials and a reduction in processing time. The ability to bypass purification until the final crystallization step ensures that the commercial scale-up of complex pharmaceutical intermediates becomes far more feasible, offering a competitive edge in terms of both speed to market and production economics.
Mechanistic Insights into the One-Pot Cascade Synthesis
The chemical elegance of this process is rooted in the precise orchestration of condensation, cyclization, and deprotection events. The synthesis initiates with the nucleophilic attack of the nitrogen atom in 4-AA onto the thioacid derivative, forming a critical thioester linkage under mild alkaline conditions ranging from -20°C to 60°C. Subsequent acylation introduces the oxalyl moiety, setting the stage for the pivotal ring-closing event. The intramolecular Wittig reaction, facilitated by triphenylphosphine or triethyl phosphite at elevated temperatures (60°C to 180°C), drives the formation of the strained four-membered beta-lactam ring fused to the five-membered ring system. This cyclization is thermodynamically challenging but is rendered efficient by the high concentration of reactive species maintained in the one-pot environment, avoiding the dilution effects typical of stepwise additions.
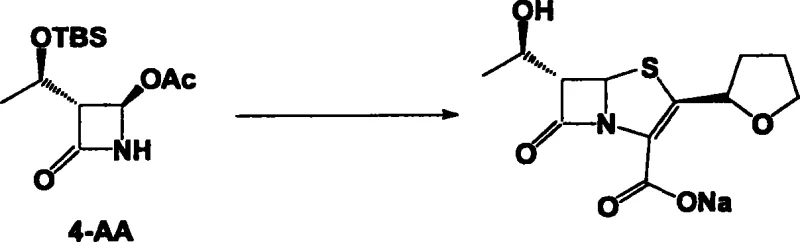
Following the construction of the bicyclic core, the protocol addresses the removal of protecting groups and the final salt formation with remarkable efficiency. The deprotection step, utilizing reagents such as hydrofluoric acid or tetrabutylammonium fluoride, cleaves the silyl ether without compromising the sensitive beta-lactam scaffold. Finally, the reaction with sodium 2-ethylhexanoate in the presence of a palladium catalyst facilitates the removal of the allyl protecting group and simultaneous salt formation. This tandem deprotection-salt formation is crucial for controlling the impurity profile, as it prevents the accumulation of free acid forms that could complicate downstream processing. Understanding these mechanistic nuances is vital for high-purity faropenem sodium manufacturers to maintain strict quality control standards while maximizing throughput.
How to Synthesize Faropenem Sodium Efficiently
Implementing this synthesis route requires careful attention to reaction parameters, particularly temperature control and stoichiometry during the Wittig cyclization phase. The process begins with the condensation of 4-AA and the thioacid, followed by acylation, where maintaining the pH between 12 and 13 is critical for optimal conversion. The subsequent cyclization step demands rigorous thermal management, often requiring reflux conditions in solvents like xylene to drive the reaction to completion over a period of up to 36 hours. While the general workflow is simplified, the specific operational details regarding solvent swaps and concentration steps are paramount for success. For a comprehensive guide on executing this protocol with precision, please refer to the standardized synthesis steps outlined below.
- Condense 4-AA with (R)-tetrahydrofuran-2-thioformic acid in the presence of a base at -20°C to 60°C.
- React the crude condensate with allyloxy oxalyl chloride using an organic base to form the acylated intermediate.
- Perform intramolecular Wittig cyclization using triphenylphosphine or triethyl phosphite, followed by deprotection and salt formation with sodium 2-ethylhexanoate.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this one-pot technology offers profound benefits that extend beyond simple yield improvements. By collapsing multiple unit operations into a single continuous process, manufacturers can significantly reduce the capital expenditure required for reactor occupancy time and separation equipment. This efficiency gain directly correlates to a lower cost of goods sold (COGS), allowing suppliers to offer more competitive pricing structures without sacrificing margin. Moreover, the reduction in solvent consumption and waste generation aligns with increasingly stringent global environmental regulations, mitigating the risk of supply disruptions caused by compliance issues. For supply chain heads, this means a more resilient and sustainable source of antibiotic intermediates that can withstand market volatility.
- Cost Reduction in Manufacturing: The elimination of intermediate purification steps removes the need for expensive chromatographic media and vast quantities of recrystallization solvents. This qualitative reduction in material usage leads to substantial cost savings, as the process avoids the yield losses typically associated with isolation procedures. Furthermore, the simplified workflow reduces labor costs and energy consumption, as fewer heating and cooling cycles are required for separate reaction vessels. These factors combine to create a highly economical production model that enhances profitability for large-scale manufacturers.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a wide range of temperatures and utilize common industrial solvents like ethyl acetate and dichloromethane, ensures consistent batch-to-batch performance. This reliability is critical for maintaining uninterrupted supply lines to downstream API producers. By minimizing the number of handling steps, the risk of human error and cross-contamination is drastically reduced, leading to higher first-pass yields and more predictable delivery schedules. This stability is essential for partners seeking a reliable faropenem sodium supplier capable of meeting long-term contractual obligations.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing reagents and conditions that are easily managed in standard stainless steel reactors found in modern CDMO facilities. The reduction in "three wastes" (waste water, waste gas, and solid waste) due to the absence of intermediate workups simplifies effluent treatment protocols. This environmental advantage not only lowers disposal costs but also future-proofs the manufacturing site against tightening ecological regulations, ensuring long-term operational continuity and social responsibility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and claims presented in CN1314691C, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing production portfolios. We encourage technical teams to review these points carefully to assess alignment with their specific quality and capacity requirements.
Q: What is the primary advantage of the one-pot method described in CN1314691C?
A: The primary advantage is the elimination of intermediate purification steps. Traditional methods require isolating and purifying intermediates after every reaction stage, which significantly reduces overall yield and increases solvent consumption. This novel approach allows the crude reaction mixture to proceed directly to the next step, drastically improving efficiency and reducing production costs.
Q: How does this process impact the purity of the final Faropenem Sodium API?
A: Despite skipping intermediate purification, the process achieves high purity levels, exemplified by the final recrystallization step which yields product with purity exceeding 99% (HPLC). The controlled reaction conditions and specific choice of reagents minimize side reactions, ensuring that impurity profiles remain within acceptable limits for pharmaceutical applications without the need for extensive intermediate workups.
Q: Is this synthesis route suitable for large-scale industrial manufacturing?
A: Yes, the process is explicitly designed for large-scale industrial production. By utilizing common solvents like ethyl acetate and dichloromethane, and avoiding complex purification techniques like column chromatography for intermediates, the method simplifies equipment requirements. The robust reaction conditions, ranging from mild temperatures to reflux, are easily manageable in standard stainless steel reactors used in CDMO facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Faropenem Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the one-pot synthesis described in CN1314691C for the global antibiotic market. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate this laboratory-scale innovation into commercial reality. Our facilities are equipped to handle complex organic syntheses with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch of Faropenem Sodium meets the highest international pharmacopoeia standards, guaranteeing safety and efficacy for the final drug product.
We invite pharmaceutical companies and procurement leaders to collaborate with us to leverage this advanced manufacturing technology. By partnering with our technical team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and detailed route feasibility assessments. Let us help you secure a sustainable, high-quality supply of Faropenem Sodium that drives your business forward.
