Advanced Industrial Synthesis of Faropenem Sodium: A Technical Breakdown for Global Procurement
The pharmaceutical landscape for oral penem antibiotics has been significantly reshaped by the innovations detailed in patent CN1884284A, which outlines a robust industrial process for the preparation of Faropenem sodium. This specific intellectual property addresses critical bottlenecks in the synthesis of this broad-spectrum antibacterial agent by leveraging natural L-threonine as a chiral pool starting material. For R&D directors and process chemists, the transition from unstable epoxy intermediates to stable epoxy butyramides represents a paradigm shift in manufacturing reliability. The patent explicitly targets the limitations of prior art, such as the low-yielding classical Wittig reactions and the environmentally hazardous use of heavy metal oxidants. By establishing a pathway that ensures high optical purity and operational simplicity, this technology offers a compelling value proposition for reliable pharmaceutical intermediates supplier networks seeking to secure the supply chain for next-generation beta-lactams.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Faropenem sodium has been plagued by inefficiencies inherent in early synthetic strategies. As illustrated in the prior art reaction schemes, classical approaches often relied on the formation of ylide inner salts which exhibited notoriously poor reproducibility and low conversion rates. Specifically, earlier United States patents reported overall yields reaching only about 7.5% based on starting raw materials, primarily due to the difficulty in forming the requisite ylide species efficiently. Furthermore, alternative routes developed to bypass these yield issues introduced severe economic and environmental liabilities, such as the reliance on triphenyl thiophenol and silver nitrate. These reagents are not only prohibitively expensive but also introduce significant pollution burdens and heavy metal residue concerns that complicate downstream purification. The use of racemic DL-tetrahydrofuran-3-carbothioic acid in older methods further exacerbated these issues by generating steric isomers that required complex and wasteful chromatographic separation.
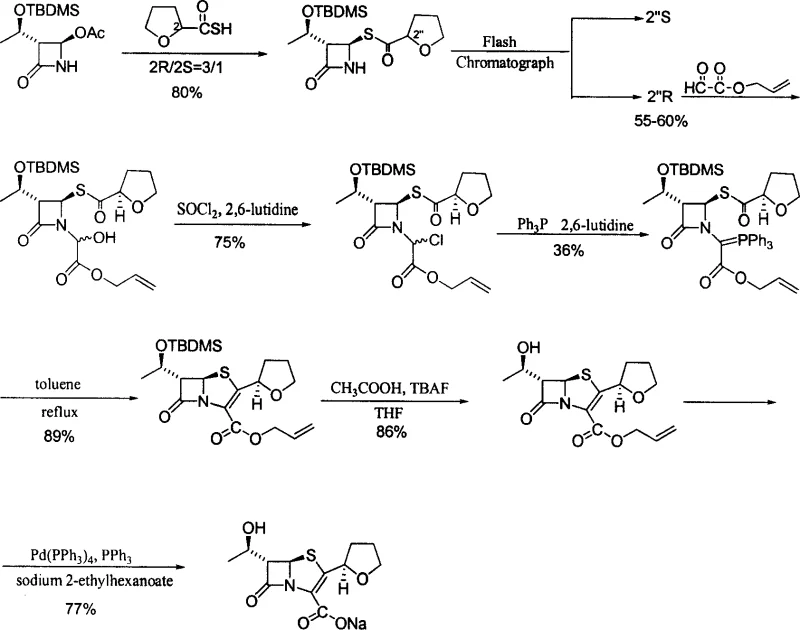
The Novel Approach
The methodology presented in CN1884284A fundamentally re-engineers the synthetic trajectory to overcome these historical deficits. By initiating the synthesis with natural L-threonine, the process capitalizes on an abundant and cost-effective chiral source to construct the beta-lactam nucleus with high stereospecificity. A key innovation is the stabilization of the epoxy intermediate; rather than isolating unstable epoxy butyric acid, the process converts it immediately into a stable epoxy butyramide (Intermediate V). This strategic modification prevents degradation and allows for one-pot alkylation and cyclization steps that dramatically streamline the workflow. Additionally, the substitution of racemic acids with optically pure 2-(R)-tetrahydrofuran-3-carbothioic acid eliminates the formation of unwanted isomers at the source. This approach not only simplifies the operation but also ensures that the final product meets stringent purity specifications without the need for extensive column chromatography, making it highly suitable for commercial scale-up of complex beta-lactams.
Mechanistic Insights into Stereospecific Beta-Lactam Cyclization
The core chemical elegance of this patent lies in its meticulous control over stereochemistry during the construction of the four-membered beta-lactam ring. The process begins with the conversion of L-threonine into (2S, 3R)-2-bromo-3-hydroxybutyric acid via diazotization, followed by amidation using a mixed anhydride method. This activation strategy is superior to acid chloride methods as it proceeds under milder conditions with fewer byproducts. The subsequent epoxidation under mineral alkali conditions yields the stable epoxy butyramide V, which serves as the pivotal scaffold. In the presence of mineral alkali, this intermediate undergoes N-alkylation with compound VIII to form compound VI, which then spontaneously cyclizes to form the trans-configured beta-lactam nucleus (Compound VII). The use of mineral alkali facilitates the formation of the nitrogen anion intermediate, driving the ring closure with high stereoselectivity. This mechanistic pathway avoids the harsh conditions and unstable intermediates associated with previous LiHMDS-mediated cyclizations, thereby enhancing the robustness of the reaction profile.
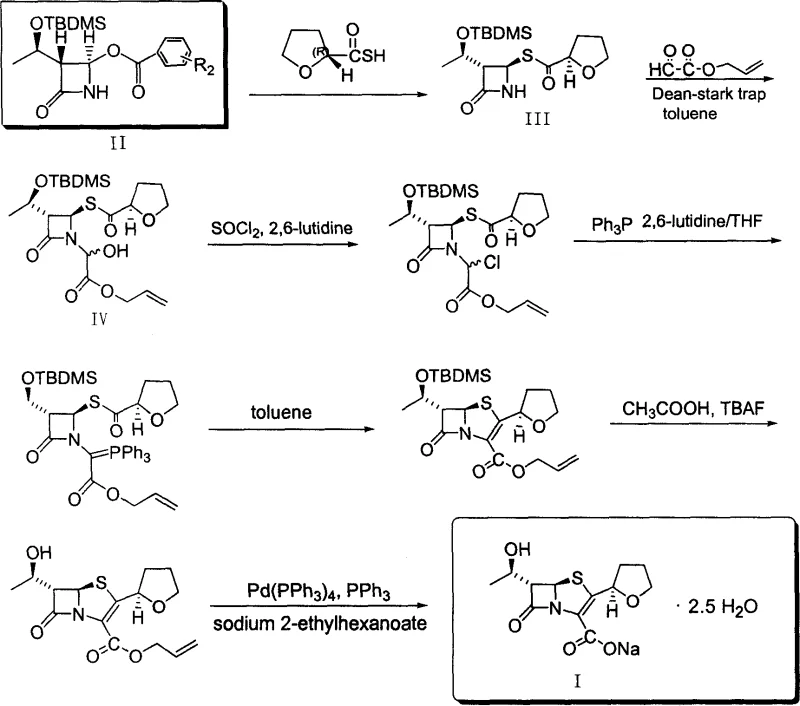
Following the construction of the nucleus, the process employs a refined intramolecular Wittig reaction to close the second ring, forming the penem structure. Unlike traditional methods that required refluxing in tetrahydrofuran for up to 75 hours, this patent optimizes the ylide formation by utilizing dimethylformamide (DMF) as a solvent, reducing the reaction time to merely 8 hours. The final deprotection steps involve the removal of the silyl protecting group and the allyl ester moiety using zero-valent palladium catalysts. Crucially, the selection of allyl acceptors, such as sodium 2-ethylhexanoate, ensures the clean formation of the sodium salt without introducing difficult-to-remove impurities. This comprehensive control over the reaction mechanism ensures that the optical purity of the product remains above 99%, addressing the critical quality attributes required for high-purity Faropenem Sodium intended for clinical applications.
How to Synthesize Faropenem Sodium Efficiently
The synthesis of Faropenem Sodium via this patented route involves a sequence of highly optimized transformations designed for industrial feasibility. The process initiates with the derivatization of L-Threonine into a stable epoxy intermediate, followed by stereospecific cyclization to build the beta-lactam core. Subsequent steps involve the introduction of the tetrahydrofuran side chain and the closure of the penem ring via an improved Wittig reaction. The final stages focus on deprotection and salt formation to yield the active pharmaceutical ingredient. While the general workflow is outlined here, precise control of reaction parameters such as temperature, stoichiometry, and solvent choice is critical for success. For detailed standard operating procedures and specific reaction conditions validated at scale, please refer to the technical guide below.
- Convert natural L-Threonine into stable epoxy butyramide intermediate V via diazotization, bromination, and mixed anhydride amidation.
- Perform N-alkylation and stereospecific cyclization under mineral alkali conditions to construct the beta-lactam nucleus (Compound VII).
- Execute intramolecular Wittig cyclization and final deprotection using palladium catalysis to yield high-purity Faropenem Sodium.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the CN1884284A process translates into tangible strategic advantages regarding cost structure and supply continuity. The shift away from exotic and expensive reagents like silver nitrate and lead salts towards commodity chemicals like L-threonine and mineral alkalis significantly de-risks the supply chain. This substitution not only lowers the direct material costs but also mitigates the regulatory and disposal costs associated with heavy metal waste management. Furthermore, the elimination of complex chromatographic purification steps, which are often bottlenecks in batch processing, allows for faster throughput and reduced solvent consumption. These factors collectively contribute to substantial cost reduction in API manufacturing, enabling more competitive pricing for the final drug product while maintaining high margins for manufacturers.
- Cost Reduction in Manufacturing: The replacement of costly chiral auxiliaries and heavy metal oxidants with inexpensive, naturally sourced L-threonine creates a fundamentally leaner cost base. By avoiding the use of lead salts and silver reagents, the process eliminates the need for expensive scavenging resins and specialized waste treatment protocols. Additionally, the ability to isolate intermediates as stable solids rather than unstable oils reduces material loss during handling and storage. These cumulative efficiencies drive down the cost of goods sold (COGS) without compromising on the quality or potency of the final antibiotic.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly simplified by the reliance on L-threonine, a bulk fermentation product with a stable global supply, rather than specialized synthetic building blocks that may face availability fluctuations. The robustness of the epoxy butyramide intermediate ensures that production campaigns can be paused and resumed without significant degradation of materials, offering greater flexibility in production scheduling. This stability is crucial for reducing lead time for high-purity antibiotics, ensuring that pharmaceutical partners can meet market demand consistently even during periods of raw material volatility.
- Scalability and Environmental Compliance: The process is inherently designed for scale, utilizing common solvents like ethyl acetate and toluene which are easily recovered and recycled in large-scale plants. The avoidance of water-sensitive reagents like LiHMDS removes the requirement for strict inert atmosphere operations, simplifying reactor requirements and lowering capital expenditure for new production lines. Moreover, the absence of heavy metal residues aligns perfectly with increasingly stringent environmental regulations, facilitating smoother regulatory approvals and minimizing the ecological footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Faropenem sodium synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer or licensing agreements.
Q: How does the L-Threonine route improve yield compared to classical Wittig methods?
A: The classical Wittig route suffers from low ylide formation yields (approx. 7.5%). The L-Threonine route utilizes stable epoxy butyramide intermediates and optimized intramolecular cyclization, significantly boosting overall productivity and optical purity.
Q: Does this process eliminate heavy metal contamination risks?
A: Yes. Unlike previous methods that utilized toxic lead salts as oxidants or expensive silver nitrate, this patented process relies on peroxy acid oxidation and avoids heavy metal reagents entirely, simplifying purification and ensuring safety.
Q: What are the scalability advantages of using optically pure 2-(R)-tetrahydrofuran-3-carbothioic acid?
A: Using the optically pure (R)-enantiomer prevents the formation of steric isomers that occur with racemic DL-acids. This eliminates the need for difficult chromatographic separations, drastically reducing solvent waste and processing time during scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Faropenem Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic pathways in the modern pharmaceutical supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN1884284A are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of Faropenem Sodium meets the highest international standards. Our commitment to technical excellence allows us to navigate the complexities of beta-lactam chemistry, delivering consistent quality that supports your regulatory filings and market launch timelines.
We invite you to engage with our technical procurement team to discuss how this advanced L-threonine route can optimize your specific manufacturing needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this greener, more efficient process. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a seamless transition to a more sustainable and cost-effective supply model.
