Advanced L-Threonine Derived Synthesis of Faropenem Sodium for Commercial API Production
Advanced L-Threonine Derived Synthesis of Faropenem Sodium for Commercial API Production
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective pathways for the production of critical antibiotic intermediates. Patent CN1884284A introduces a groundbreaking methodology for the preparation of Faropenem sodium, a potent oral penem antibiotic with broad-spectrum activity against both aerobic and anaerobic bacteria. This innovative process utilizes natural L-Threonine as the primary chiral source, effectively bypassing the limitations of earlier synthetic routes that relied on unstable intermediates or toxic heavy metals. By establishing a stable epoxy butyramide intermediate and employing stereospecific cyclization techniques, this technology offers a viable solution for commercial scale-up of complex beta-lactams. For R&D directors and procurement specialists, understanding this shift from classical Wittig reactions to stabilized amide-based cyclization is crucial for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent global quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Faropenem sodium has been plagued by significant technical and economic hurdles that hinder efficient large-scale manufacturing. Early approaches, such as the classical Wittig reaction pathway, suffered from inherently low reaction yields, often reaching only about 7.5% based on starting raw materials, primarily due to the difficulty in forming the necessary ylide inner salt. Furthermore, alternative routes developed to improve cyclization, such as the two-carbonyl link coupling method, introduced severe supply chain and environmental liabilities. These conventional methods frequently required expensive and scarce reagents like triphenyl thiophenol and silver nitrate, alongside toxic heavy metal lead salts used as oxidants. The reliance on such hazardous materials not only escalated production costs but also created insoluble heavy-metal residual problems, necessitating complex purification steps to meet safety regulations. Additionally, many intermediates in these traditional processes existed as unstable thick liquids, forcing manufacturers to rely on tedious column chromatography for purification, which is impractical for multi-ton production.
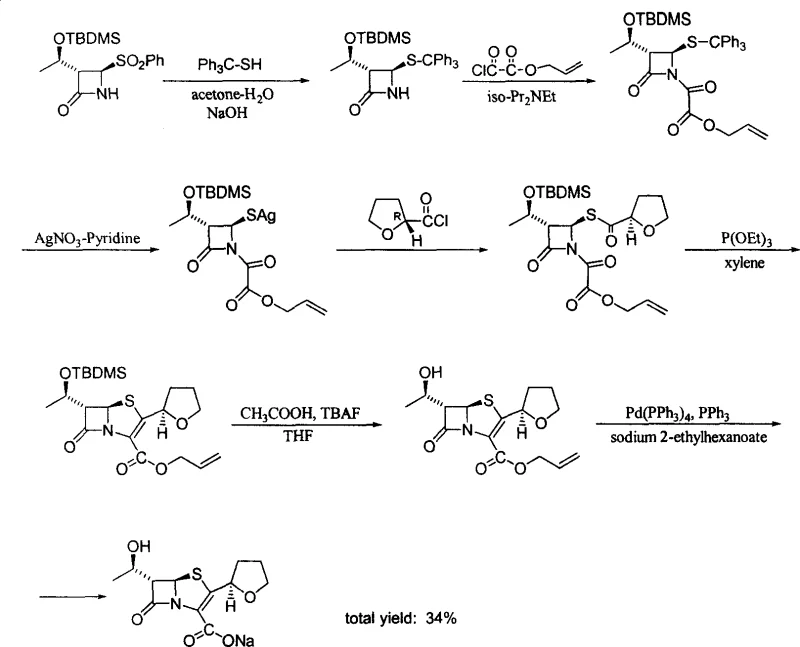
The Novel Approach
In stark contrast, the novel process outlined in the patent leverages natural L-Threonine to construct the beta-lactam nucleus with high stereospecificity and operational simplicity. This method replaces the unstable epoxy butyric acid found in older literature with a stable epoxy butyramide intermediate, which can be easily purified and handled without special atmospheric conditions. A key innovation involves the substitution of racemic DL-tetrahydrofuran thioacid with optically pure 2-(R)-tetrahydrofuran thioacid, which prevents the formation of steric isomers and eliminates the need for difficult separation processes. The reaction conditions are notably milder, utilizing mineral alkalis for cyclization and avoiding the strict anhydrous and inert gas requirements associated with expensive reagents like LiHMDS. By converting substituted benzoyl groups into acyl-oxygen bases via peroxy acid oxidation, the process ensures that most intermediates are stable solids rather than oils, facilitating easy isolation through crystallization. This streamlined approach drastically simplifies post-treatment and enhances the overall feasibility of industrial application.
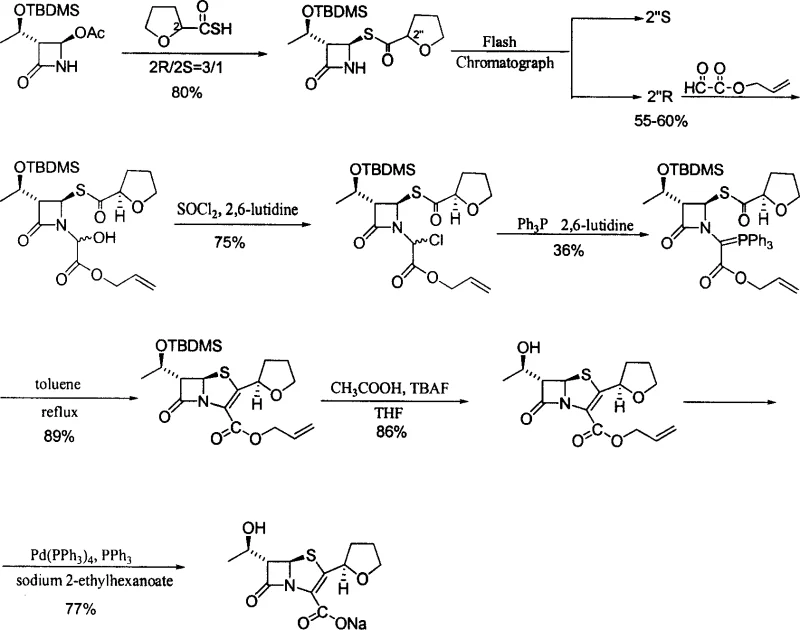
Mechanistic Insights into L-Threonine Based Beta-Lactam Cyclization
The core of this technological advancement lies in the precise stereochemical control exercised during the construction of the four-membered beta-lactam ring. The process initiates with the conversion of L-Threonine into (2S, 3R)-2-bromo-3-hydroxybutyric acid via diazotization and bromination, followed by amidation to form a stable amide. Under the influence of mineral alkali, this amide undergoes an epoxidation reaction to yield a high-purity epoxy butyramide intermediate. This intermediate is then subjected to N-alkylation and subsequent cyclization in a one-pot reaction, where the nitrogen anion attacks the carbonyl group to close the ring with trans-configuration. The use of mineral alkalis such as sodium hydroxide or potassium hydroxide allows for this cyclization to proceed at moderate temperatures (20~100 ℃), avoiding the extreme conditions that often lead to racemization or decomposition in other methods. The resulting beta-lactam nucleus is protected with tert-butyl dimethyl chlorosilane, ensuring the hydroxyl group remains intact during subsequent oxidative transformations.
Furthermore, the process incorporates specific solvent and reagent optimizations that significantly accelerate reaction kinetics without compromising purity. For instance, the formation of the phosphorus ylide, a critical step for constructing the double bond in the penem ring, is traditionally a slow process requiring reflux in tetrahydrofuran for up to 75 hours. However, this patent discloses that using dimethyl formamide (DMF) as the solvent reduces this reaction time to merely 8 hours, representing a massive gain in throughput efficiency. Additionally, the removal of trace water during the condensation of the intermediate with glyoxylic acid allyl ester is achieved using hydrolith (calcium hydride) instead of molecular sieves, which shortens the reaction time by half. These mechanistic refinements ensure that the final product, Faropenem sodium, achieves an optical purity and chemical purity of greater than or equal to 99%, meeting the rigorous specifications required for high-purity Faropenem Sodium in clinical applications.
How to Synthesize Faropenem Sodium Efficiently
The synthesis of Faropenem Sodium via this patented route involves a sequence of well-defined chemical transformations starting from readily available L-Threonine. The process is designed to maximize yield at each stage while minimizing the generation of hazardous waste and the need for complex purification equipment. Operators must strictly control reaction temperatures and stoichiometry, particularly during the diazotization and epoxidation steps, to maintain the stereochemical integrity of the chiral centers. The following guide outlines the critical phases of this synthesis, emphasizing the transition from the chiral amino acid to the final bicyclic penem structure. For detailed standard operating procedures and specific molar ratios, please refer to the technical documentation provided below.
- Convert natural L-Threonine into stable epoxy butyramide intermediates via diazotization and bromination, avoiding unstable epoxy butyric acid.
- Construct the beta-lactam nucleus stereospecifically using mineral alkali cyclization, followed by hydroxyl protection with TBDMS.
- Perform intramolecular Wittig cyclization using optically pure tetrahydrofuran thioacid, followed by deprotection and palladium-catalyzed allyl removal to yield the sodium salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this L-Threonine based synthesis route offers transformative benefits regarding cost structure and supply reliability. The elimination of expensive and scarce reagents such as silver nitrate and triphenyl thiophenol directly translates to a significant reduction in raw material expenditure. Moreover, the avoidance of heavy metal lead salts removes the need for costly重金属 removal steps and mitigates environmental compliance risks associated with toxic waste disposal. The fact that the majority of intermediates are stable solids rather than unstable oils means that the process does not rely on column chromatography, a technique that is notoriously difficult to scale and expensive to operate in a GMP environment. This shift to crystallization-based purification not only lowers capital expenditure on equipment but also reduces solvent consumption and processing time, contributing to substantial cost savings in API manufacturing.
- Cost Reduction in Manufacturing: The replacement of racemic starting materials with optically pure 2-(R)-tetrahydrofuran thioacid eliminates the generation of unwanted steric isomers, thereby preventing the loss of high-value intermediates during separation. This optimization, combined with the use of inexpensive mineral alkalis instead of costly organolithium reagents like LiHMDS, ensures a leaner cost profile. The ability to perform key cyclization steps in a one-pot manner further reduces solvent usage and labor costs, driving down the overall cost of goods sold (COGS) for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: By utilizing natural L-Threonine as the foundational chiral pool material, the process relies on a commodity chemical with a stable and abundant global supply, reducing the risk of raw material shortages. The stability of the epoxy butyramide intermediate allows for potential storage and batch consolidation, providing flexibility in production scheduling. Furthermore, the shortened reaction times achieved through solvent optimization (e.g., using DMF for ylide formation) increase plant throughput, enabling suppliers to respond more rapidly to market demand fluctuations and reducing lead time for high-purity antibiotics.
- Scalability and Environmental Compliance: The process is explicitly designed for industrialization, featuring mild reaction conditions that do not require extreme cryogenic temperatures or high-pressure equipment. The absence of heavy metal oxidants simplifies the wastewater treatment process, ensuring easier compliance with increasingly strict environmental regulations. The robustness of the solid intermediates facilitates safe handling and transport between production stages, minimizing the risk of degradation and ensuring consistent quality across large-scale batches ranging from pilot plant to commercial tonnage.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Faropenem Sodium using this advanced L-Threonine derived pathway. These answers are derived directly from the patent specifications and are intended to clarify the operational advantages and quality assurances associated with this method. Understanding these details is essential for partners evaluating the feasibility of integrating this intermediate into their existing supply chains.
Q: How does the new L-Threonine route improve upon classical Wittig synthesis?
A: The classical Wittig route suffers from low overall yields (around 7.5%) due to poor ylide inner salt formation. The new L-Threonine route utilizes stable epoxy butyramide intermediates and avoids heavy metal oxidants like lead salts, significantly improving yield and purity while simplifying post-treatment.
Q: What are the cost advantages of using optically pure 2-(R)-tetrahydrofuran thioacid?
A: Using optically pure 2-(R)-tetrahydrofuran thioacid eliminates the generation of steric isomers that occur with racemic DL-acids. This avoids complex and costly separation processes, reduces raw material loss, and streamlines the operation, leading to substantial cost reductions in the final API manufacturing.
Q: Does this process require column chromatography for purification?
A: Unlike previous methods where most intermediates were thick liquids requiring column chromatography, this novel process produces mostly solid intermediates. These solids are stable and easy to purify through crystallization or simple washing, making the technology highly suitable for industrial scale-up without complex chromatographic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Faropenem Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to ensure the consistent supply of high-quality antibiotic intermediates. Our technical team has extensively analyzed the L-Threonine based process described in CN1884284A and possesses the expertise to implement these methodologies effectively. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet stringent purity specifications. Our rigorous QC labs are equipped to verify the stereochemical integrity and impurity profiles of every batch, guaranteeing that our Faropenem Sodium intermediates are ready for immediate downstream API synthesis.
We invite global pharmaceutical partners to collaborate with us to leverage these cost-effective and environmentally friendly production technologies. By choosing NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring a seamless integration of our high-performance intermediates into your manufacturing pipeline.
