Revolutionizing Beta-Amino Acid Production: A Scalable Ketone-Based Route for Global Pharma Supply Chains
Introduction to Advanced Beta-Amino Acid Manufacturing
The pharmaceutical industry's relentless pursuit of more stable and bioactive peptide therapeutics has placed beta-amino acids at the forefront of medicinal chemistry research. Patent CN1511826A, filed in 2004, presents a groundbreaking methodology for synthesizing these critical building blocks using ketones as the primary starting material. This innovation addresses long-standing bottlenecks in the production of unnatural amino acids, which are essential for enhancing the proteolytic stability and oral bioavailability of next-generation peptide drugs. By shifting the synthetic paradigm from limited aldehyde precursors or hazardous diazo-compounds to abundant and versatile ketones, this technology offers a transformative pathway for cost reduction in API manufacturing. The patent details three distinct yet complementary routes—the malonic acid method, the ethyl cyanoacetate method, and the cyanoacetic acid method—each providing unique advantages depending on the desired substitution pattern. For R&D directors and procurement strategists, understanding the mechanistic robustness and economic implications of this ketone-based approach is vital for securing a reliable pharmaceutical intermediate supplier capable of meeting the rigorous demands of modern drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-amino acids has been plagued by significant technical and safety hurdles that hindered large-scale adoption. The most prominent traditional method, the Arndt-Eistert synthesis, relies on the homologation of alpha-amino acids using diazomethane. As illustrated in the reaction scheme below, this process involves protecting the amino group, converting the acid to an acyl chloride, and then reacting it with highly toxic and explosive diazomethane. 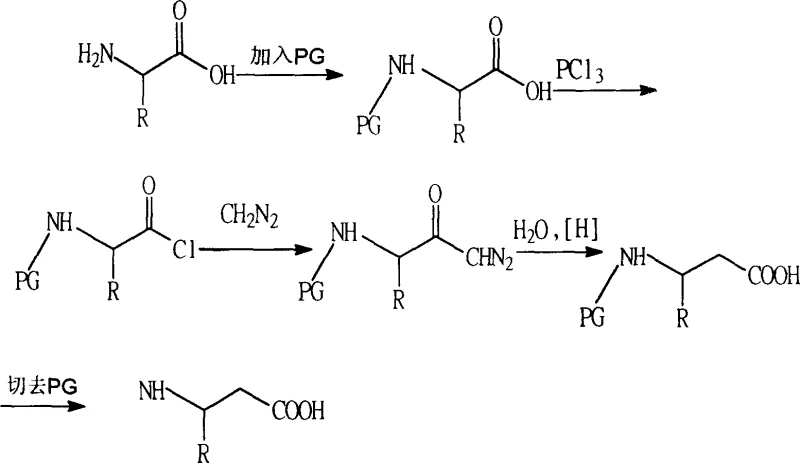 This reliance on diazomethane creates severe safety liabilities, particularly when scaling from gram to kilogram quantities, necessitating specialized equipment and rigorous hazard controls that drive up operational costs. Furthermore, the starting materials for these conventional routes are often restricted to specific aldehydes or expensive natural alpha-amino acids, limiting the structural diversity of the final products. The multi-step nature of these legacy processes, combined with difficult purification protocols and the use of unstable reagents, results in lower overall yields and inconsistent quality, making them ill-suited for the high-volume production required by the global supply chain.
This reliance on diazomethane creates severe safety liabilities, particularly when scaling from gram to kilogram quantities, necessitating specialized equipment and rigorous hazard controls that drive up operational costs. Furthermore, the starting materials for these conventional routes are often restricted to specific aldehydes or expensive natural alpha-amino acids, limiting the structural diversity of the final products. The multi-step nature of these legacy processes, combined with difficult purification protocols and the use of unstable reagents, results in lower overall yields and inconsistent quality, making them ill-suited for the high-volume production required by the global supply chain.
The Novel Approach
In stark contrast, the methodology disclosed in CN1511826A leverages the abundance and chemical versatility of ketones to construct the beta-amino acid skeleton efficiently. Instead of dangerous diazo-transfer reactions, this novel approach utilizes a condensation reaction between a ketone and an active methylene compound, such as ethyl cyanoacetate or cyanoacetic acid, to form an unsaturated intermediate. This strategy not only bypasses the safety risks associated with diazomethane but also vastly expands the library of accessible compounds, allowing for the synthesis of both alpha- and beta-substituted derivatives that were previously difficult to obtain. The subsequent steps involve catalytic hydrogenation, a well-established and scalable unit operation in the fine chemical industry, followed by a straightforward hydrolysis. This streamlined sequence reduces the number of isolation steps, minimizes solvent consumption, and utilizes commodity chemicals that are readily available from bulk suppliers. For procurement managers, this translates to a more resilient supply chain with reduced dependency on niche, high-cost reagents, effectively mitigating the risk of production delays.
Mechanistic Insights into Ketone-Based Condensation and Hydrogenation
The core of this innovative synthesis lies in the precise control of the condensation and sequential hydrogenation steps. The process begins with a Knoevenagel-type condensation where the ketone reacts with ethyl cyanoacetate in the presence of a weak base, such as diethylamine or ammonium acetate, to generate an alpha,beta-unsaturated nitrile (enester). This step is critical as it establishes the carbon framework of the beta-amino acid. Following condensation, the unsaturated intermediate undergoes a two-stage catalytic hydrogenation process. First, the carbon-carbon double bond is selectively reduced using a palladium on carbon (Pd-C) catalyst under mild hydrogen pressure (3-10 atm). This step requires careful monitoring to ensure complete saturation of the alkene without affecting the nitrile group. Subsequently, the nitrile functionality is reduced to a primary amine using Raney Nickel, a highly active catalyst for nitrile hydrogenation, typically under higher pressures (40-100 atm) and elevated temperatures. 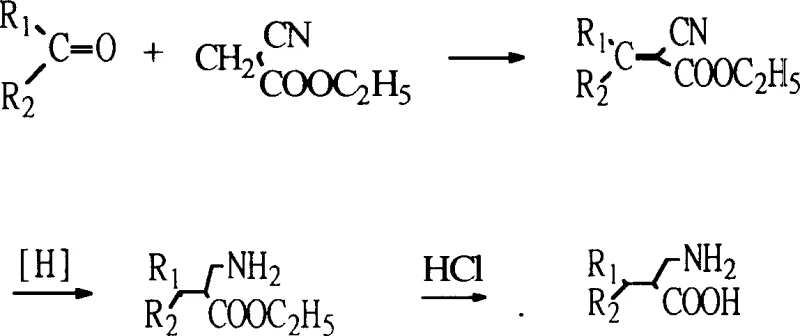 This dual-catalyst system ensures high chemoselectivity, preventing the formation of unwanted byproducts such as secondary amines or over-reduced species. The final hydrolysis step cleaves the ester group to reveal the carboxylic acid, yielding the target beta-amino acid with high purity. From a technical perspective, the ability to tune reaction conditions—such as solvent choice (methanol, ethanol, acetic acid) and base additives—allows for the optimization of impurity profiles, ensuring that the final product meets the stringent specifications required for pharmaceutical applications.
This dual-catalyst system ensures high chemoselectivity, preventing the formation of unwanted byproducts such as secondary amines or over-reduced species. The final hydrolysis step cleaves the ester group to reveal the carboxylic acid, yielding the target beta-amino acid with high purity. From a technical perspective, the ability to tune reaction conditions—such as solvent choice (methanol, ethanol, acetic acid) and base additives—allows for the optimization of impurity profiles, ensuring that the final product meets the stringent specifications required for pharmaceutical applications.
How to Synthesize Beta-Amino Acids Efficiently
Implementing this synthesis route requires a clear understanding of the operational parameters defined in the patent to ensure reproducibility and safety. The process is designed to be modular, allowing manufacturers to select the specific pathway (malonic acid, ethyl cyanoacetate, or cyanoacetic acid) that best aligns with their target molecule's substitution pattern and available infrastructure. Detailed standard operating procedures regarding molar ratios, temperature controls, and catalyst loading are essential for maximizing yield and minimizing waste.
- Condense ketone with ethyl cyanoacetate or cyanoacetic acid using a weak base catalyst to form the unsaturated intermediate (enester or enoic acid).
- Perform sequential catalytic hydrogenation: first reduce the carbon-carbon double bond using Pd-C, then reduce the cyano group using Raney Nickel under alkaline conditions.
- Hydrolyze the resulting ester or amide intermediate under acidic conditions to yield the final beta-amino acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the transition to this ketone-based synthesis represents a strategic opportunity to optimize costs and enhance supply security. The shift away from exotic or hazardous reagents towards commodity chemicals fundamentally alters the economic model of beta-amino acid production. By utilizing widely available ketones and standard catalysts, manufacturers can decouple their production schedules from the volatility of specialized reagent markets. This stability is crucial for maintaining continuous supply lines for critical drug intermediates, especially in a global market prone to logistical disruptions. Furthermore, the simplified workup procedures, which often involve basic filtration and distillation rather than complex chromatography, significantly reduce processing time and labor costs. These efficiencies compound to deliver substantial value, making the final API more cost-competitive without compromising on quality or regulatory compliance.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like diazomethane removes a major cost driver and safety burden from the production process. Additionally, the use of commodity ketones and active methylene compounds, which are produced on a massive industrial scale, ensures low raw material costs. The high atom economy of the condensation-hydrogenation sequence further contributes to cost efficiency by minimizing waste generation and reducing the load on waste treatment facilities. This holistic reduction in operational complexity allows for significant margin improvement in the final product pricing.
- Enhanced Supply Chain Reliability: Sourcing raw materials is a critical vulnerability in pharmaceutical supply chains. This method relies on reagents such as cyclohexanone, acetone, and ethyl cyanoacetate, which are produced by multiple global suppliers, thereby reducing single-source dependency. The robustness of the catalytic hydrogenation steps means that the process is less sensitive to minor variations in raw material quality compared to more delicate organic transformations. This resilience ensures consistent batch-to-batch quality and reliable delivery schedules, which are paramount for maintaining the continuity of downstream drug manufacturing operations.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing unit operations like hydrogenation and reflux that are standard in multi-ton chemical plants. The avoidance of chlorinated solvents in certain variations (using alcohols instead) and the ability to recover and recycle catalysts align with modern green chemistry principles. Reduced solvent usage and simpler purification steps lower the environmental footprint of the manufacturing process, facilitating easier regulatory approval and compliance with increasingly strict environmental regulations. This sustainability profile is becoming a key differentiator for suppliers seeking partnerships with environmentally conscious pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of using ketones over aldehydes for beta-amino acid synthesis?
A: Ketones offer significantly broader structural diversity and lower cost compared to aldehydes. Unlike aldehyde-based routes which are limited in substrate scope and often yield lower purity for aliphatic variants, the ketone-based method described in CN1511826A allows for the efficient synthesis of both alpha- and beta-substituted beta-amino acids with high versatility.
Q: How does this process improve safety compared to the Arndt-Eistert synthesis?
A: The conventional Arndt-Eistert synthesis relies on diazomethane, a highly toxic and explosive reagent that poses severe safety risks during scale-up. The novel ketone-based route eliminates the need for diazomethane entirely, utilizing stable, commercially available reagents like ethyl cyanoacetate and standard hydrogenation catalysts, thereby drastically improving operational safety.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the process is specifically designed for industrial scalability. It utilizes common solvents like ethanol and dichloromethane, employs robust heterogeneous catalysts (Pd-C and Raney Ni) that are easily filtered, and avoids complex purification steps, making it highly amenable to multi-ton manufacturing campaigns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Amino Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality beta-amino acids play in the development of advanced peptide therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from laboratory discovery to full-scale manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of beta-amino acid intermediate meets the highest international standards. Our commitment to process excellence means we can deliver complex unnatural amino acids with the consistency and reliability required for GMP environments.
We invite you to collaborate with us to leverage this advanced ketone-based synthesis for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can accelerate your development timeline and optimize your supply chain economics.
