Optimizing Etoposide Phosphate Production: A Novel Glycosylation Strategy for Commercial Scale
Optimizing Etoposide Phosphate Production: A Novel Glycosylation Strategy for Commercial Scale
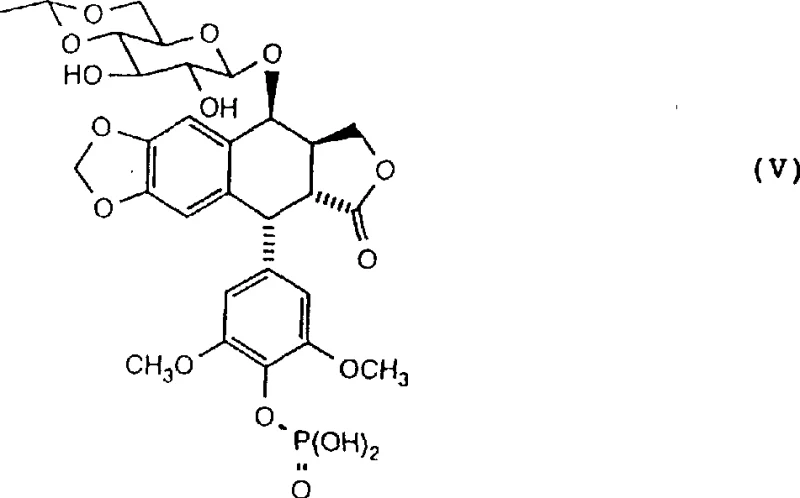
The development of water-soluble prodrugs for poorly soluble anticancer agents remains a critical challenge in pharmaceutical manufacturing, particularly for the podophyllotoxin class of compounds. Patent CN1046290C discloses a highly efficient methodology for the preparation of Etoposide phosphate, a water-soluble prodrug of the potent antineoplastic agent Etoposide. This technology addresses the longstanding limitations of solubility and formulation associated with the parent compound by introducing a phosphate moiety at the 4'-position. The core innovation lies in a convergent synthesis strategy that couples a specifically protected glucopyranose derivative with a phosphorylated aglycone precursor. By utilizing a unified benzyl protection strategy, the process enables the simultaneous removal of protecting groups in a final hydrogenation step, thereby streamlining the production workflow. For a reliable etoposide phosphate supplier, mastering this specific glycosylation and crystallization protocol is essential to delivering high-purity intermediates that meet stringent regulatory standards for oncology drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for podophyllotoxin glucosides often suffer from excessive step counts and complex protection-deprotection sequences that erode overall yield. Historically, methods required distinct protecting groups for the phenolic hydroxyl, the sugar hydroxyls, and the phosphate functionality, necessitating multiple orthogonal cleavage conditions. These harsh acidic or basic conditions frequently led to the degradation of the sensitive ethylidene bridge on the sugar ring or epimerization at the anomeric center. Furthermore, separating the desired beta-anomer from the alpha-anomer typically required laborious chromatographic purification, which is economically unviable and technically difficult to implement on a multi-kilogram or ton scale. The cumulative effect of these inefficiencies results in high manufacturing costs and inconsistent supply chains for high-purity etoposide phosphate.
The Novel Approach
The methodology described in the patent revolutionizes this landscape by employing a unified benzyl protection strategy for both the sugar and the phosphate components. By reacting 4'-demethyl-4-epipodophyllotoxin-4'-dibenzyl phosphate with 2,3-di-O-benzyl-4,6-O-ethylene-glucopyranose, the synthesis creates a fully protected intermediate where all removable groups are benzyl ethers or esters. This strategic alignment allows for a single catalytic hydrogenation step to reveal the final active pharmaceutical ingredient, eliminating the need for sequential deprotection. Crucially, the process leverages the specific solubility characteristics of the beta-anomer in methanol or acetonitrile mixtures. This allows the desired C-1' beta isomer to crystallize directly from the reaction matrix or upon simple solvent adjustment, bypassing the need for chromatography entirely and ensuring a robust, scalable purification method suitable for industrial commercial scale-up of complex glycosides.
Mechanistic Insights into Lewis Acid-Catalyzed Stereoselective Glycosylation
The heart of this synthetic route is the glycosylation reaction, which is meticulously controlled to favor the formation of the therapeutically active beta-anomer. The reaction proceeds via the activation of the protected glucopyranose donor by a Lewis acid, specifically boron trifluoride etherate, in a polar aprotic solvent like acetonitrile. The choice of solvent is not merely incidental; acetonitrile possesses a high dielectric constant that stabilizes the oxocarbenium ion intermediate and influences the stereochemical outcome of the nucleophilic attack by the phosphorylated aglycone. Experimental data within the patent indicates that conducting the reaction at reduced temperatures, specifically between -20°C and -40°C, significantly retards anomerization and kinetically favors the formation of the beta-linkage. In contrast, reactions performed in halogenated solvents like dichloroethane show lower beta-selectivity, highlighting the critical role of solvent-solute interactions in directing the stereochemistry.
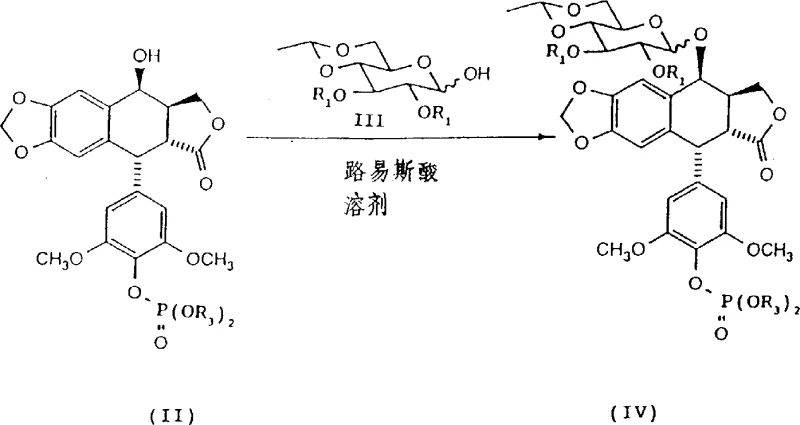
Following the coupling event, the mechanism of purification is driven by thermodynamic solubility differences between the anomers. The protected beta-anomer exhibits significantly lower solubility in methanol compared to its alpha-counterpart. Upon cooling the reaction mixture or adding methanol as an anti-solvent, the beta-anomer precipitates out of the solution in a highly purified form, often with purity exceeding 99% as determined by HPLC. This crystallization-induced diastereoselection is a powerful tool for impurity control, effectively removing the alpha-anomer and other side products without the need for silica gel columns. This mechanistic understanding allows process chemists to fine-tune parameters such as cooling rates and solvent ratios to maximize recovery, ensuring that the final etoposide phosphate product meets rigorous impurity profiles required for clinical applications.
How to Synthesize Etoposide Phosphate Efficiently
The synthesis of this critical oncology intermediate follows a logical progression from phosphorylation to glycosylation and finally to global deprotection. The process begins with the phosphorylation of the aglycone using dibenzyl phosphite and carbon tetrachloride, followed by the critical Lewis acid-mediated coupling described above. The isolation of the pure beta-anomer via crystallization serves as the pivotal quality control point before the final reduction. Detailed operational parameters, including specific molar ratios, temperature ramps, and workup procedures, are essential for reproducing the high yields reported in the literature. For a comprehensive guide on executing this synthesis with maximum efficiency and safety, please refer to the standardized protocol below.
- Phosphorylate 4'-Demethylepipodophyllotoxin using dibenzyl phosphite, carbon tetrachloride, and a tertiary amine base in acetonitrile at -10°C to form the protected phosphate ester.
- Couple the protected phosphate with 2,3-di-O-benzyl-4,6-O-ethylene-glucopyranose using boron trifluoride etherate in acetonitrile at -20°C to -40°C, followed by direct crystallization to isolate the pure C-1' beta anomer.
- Perform simultaneous deprotection of the benzyl groups on the sugar and phosphate moieties via catalytic hydrogenation using Pd/C to yield high-purity Etoposide phosphate.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this patented synthesis route offers transformative advantages in terms of cost structure and operational reliability. The elimination of chromatographic purification steps represents a significant reduction in consumable costs, such as silica gel and solvents, while simultaneously increasing batch throughput. The ability to crystallize the key intermediate directly from the reaction mixture simplifies the equipment requirements, allowing for the use of standard glass-lined reactors rather than specialized purification columns. This simplification translates directly into cost reduction in anticancer API manufacturing, making the final drug substance more accessible for healthcare systems. Furthermore, the robustness of the crystallization step ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed purification attempts.
- Cost Reduction in Manufacturing: The unified benzyl protection strategy allows for a single hydrogenation step to remove all protecting groups simultaneously. This consolidation of steps eliminates the need for multiple reagent purchases, waste disposal streams, and unit operations associated with orthogonal deprotection. By avoiding the use of expensive and hazardous reagents required for selective deprotection in older methods, the overall material cost per kilogram is substantially lowered. Additionally, the high yield of the phosphorylation step (reported around 90%) and the efficient recovery of the coupled product ensure that raw material utilization is maximized, further driving down the cost of goods sold.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as acetonitrile, methanol, and palladium on carbon ensures that the supply chain is not vulnerable to shortages of exotic reagents. The process operates under mild conditions, primarily low temperatures and ambient pressure during coupling, which reduces the energy burden and equipment stress compared to high-pressure or high-temperature alternatives. This operational simplicity enhances the predictability of production schedules, reducing lead time for high-purity oncology intermediates and ensuring a steady flow of material to downstream formulation teams. The stability of the intermediates also allows for flexible inventory management if needed.
- Scalability and Environmental Compliance: The transition from laboratory to commercial scale is facilitated by the absence of chromatography, which is notoriously difficult to scale linearly. Crystallization and filtration are unit operations that scale predictably, allowing for seamless technology transfer from pilot plant to multi-ton production. Moreover, the process minimizes the generation of hazardous waste associated with silica disposal and large volumes of chromatographic solvents. The final enzymatic conversion of Etoposide phosphate to Etoposide uses aqueous buffers and biocatalysts, aligning with green chemistry principles and simplifying wastewater treatment protocols, which is a critical factor for modern environmental compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the synthesis, stability, and application of Etoposide phosphate and its precursors. These insights are derived directly from the experimental data and claims found in the underlying patent literature, providing a factual basis for technical decision-making. Understanding these nuances is vital for R&D teams evaluating this route for in-house production or for procurement specialists assessing supplier capabilities.
Q: How does this method improve the purity of the beta-anomer compared to traditional glycosylation?
A: The patented method utilizes acetonitrile as a solvent which, combined with low temperatures (-20°C to -40°C), significantly favors the formation of the beta-anomer over the alpha-anomer. Furthermore, the unique solubility profile allows the beta-anomer to crystallize directly from the reaction mixture or upon methanol addition, effectively purifying it without chromatography.
Q: Can Etoposide phosphate be converted back to Etoposide efficiently?
A: Yes, the process includes an enzymatic dephosphorylation step using alkaline phosphatase in a buffered aqueous solution (pH 8.4-8.8). This mild condition ensures high conversion yields while preventing the degradation of the sensitive ethylidene group on the sugar moiety.
Q: What are the advantages of using benzyl protecting groups for both the sugar and phosphate?
A: Using identical benzyl protecting groups for both the hydroxyls on the sugar and the phosphate ester allows for a single, simultaneous deprotection step via hydrogenation. This eliminates the need for multiple, orthogonal deprotection sequences, drastically reducing processing time and potential product degradation associated with acidic or basic hydrolysis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Etoposide Phosphate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the complexity of synthesizing podophyllotoxin derivatives requires a partner with deep technical expertise and proven scalability. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We employ stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify the anomeric purity and phosphate content of every batch. Our commitment to quality assurance means that we can deliver high-purity etoposide phosphate that complies with international pharmacopeial standards, ready for immediate use in formulation development or clinical trials.
We invite you to collaborate with us to optimize your supply chain for this critical anticancer intermediate. Our technical procurement team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. By leveraging our optimized synthesis route, we can help you achieve significant efficiencies in your manufacturing budget. Please contact us today to request specific COA data, discuss route feasibility assessments, or initiate a dialogue on how we can support your long-term strategic goals in oncology drug development.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
