Advanced Simvastatin Production: Eliminating Protection Steps for Commercial Efficiency
Introduction to Next-Generation Simvastatin Synthesis
The pharmaceutical landscape for HMG-CoA reductase inhibitors has long been dominated by the transition from natural fermentation products to semi-synthetic derivatives with enhanced potency. Specifically, the evolution from Lovastatin to Simvastatin represents a critical milestone in cholesterol management therapy. The patent CN1102588C discloses a revolutionary process for manufacturing Simvastatin that fundamentally alters the economic and technical feasibility of large-scale production. Unlike legacy methods that rely on cumbersome protection-deprotection sequences, this innovation leverages a novel amide intermediate strategy to achieve direct alpha-alkylation. For R&D directors and procurement specialists, understanding this pathway is essential, as it offers a direct route to high-purity API intermediates while circumventing the yield losses inherent in traditional lactonization-hydrolysis cycles. This report analyzes the technical depth of this patent to demonstrate how it serves as a blueprint for cost-effective, scalable manufacturing of this vital cardiovascular therapeutic.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those disclosed in United States Patent 4,444,784, typically involve a tedious four-step chemical sequence that severely impacts overall process efficiency. The conventional route necessitates the initial de-esterification of the 2-methylbutyrate side chain, followed by the protection of the 4-hydroxyl group on the pyrone ring using expensive silylating reagents like tetrabutyldimethylsilyl chloride. Subsequently, the side chain must be re-esterified to form the required 2,2-dimethylbutyrate ester, and finally, the 4-hydroxyl group must be deprotected. This multi-step approach not only consumes significant quantities of specialized reagents but also introduces multiple isolation and purification stages that cumulatively degrade the total yield. Furthermore, alternative methods involving direct methylation often suffer from poor transformation efficiency and significant side reactions due to the lack of selectivity, resulting in product purity that barely meets the stringent standards required for human healthcare applications.
The Novel Approach
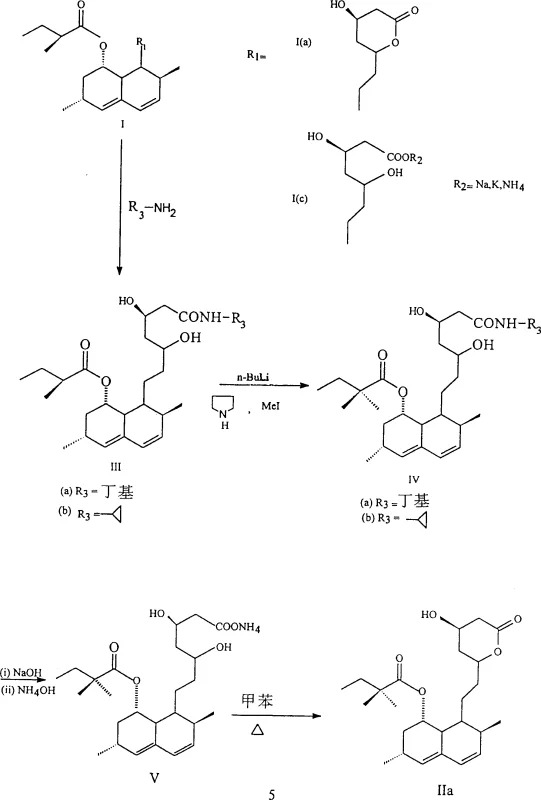
In stark contrast, the methodology outlined in CN1102588C introduces a streamlined pathway that eliminates the need for protecting the two hydroxy groups of the open pyrone ring entirely. By treating Lovastatin or Mevinolinic acid salts with specific alkylamines such as cyclopropylamine or n-butylamine, the process generates a stable amide intermediate that naturally keeps the ring open. This strategic modification allows for the direct addition of a methyl group to the 2-methylbutyrate side chain without the interference of free hydroxyl groups, effectively bypassing the need for silyl protection. The subsequent steps involve a controlled hydrolysis to form an ammonium salt intermediate, followed by a thermal lactonization to close the ring and produce Simvastatin. This approach not only reduces the number of chemical steps but also significantly lowers the consumption of reagents, time, and labor, presenting a robust solution for industrial manufacturing.
Mechanistic Insights into Direct Alpha-Alkylation via Amide Intermediates
The core chemical innovation lies in the utilization of the amide functionality to modulate the reactivity of the substrate during the critical methylation phase. When Lovastatin or Mevinolinic acid reacts with cyclopropylamine or n-butylamine, the lactone ring opens to form intermediates IIIa or IIIb. This ring-opening is crucial because it exposes the alpha-position of the side chain ester while simultaneously masking the reactivity of the hydroxyl groups through hydrogen bonding or steric hindrance within the amide structure. The subsequent treatment with a strong base, specifically lithium pyrrolidide generated in situ from n-Butyl Lithium and tetramethyleneimine in anhydrous tetrahydrofuran at temperatures between -35°C and -40°C, facilitates the selective deprotonation of the alpha-carbon. This generates a highly reactive enolate species that is perfectly positioned for nucleophilic attack on the methyl iodide electrophile.
Following the methylation, the process employs a sophisticated hydrolysis and recrystallization protocol to ensure exceptional purity. The methylated intermediate is treated with sodium hydroxide in methanol to hydrolyze the amide back to the carboxylic acid form, which is then precipitated as an ammonium salt (Intermediate V). This salt formation acts as a powerful purification step, allowing impurities to remain in the mother liquor while the desired product crystallizes out. The final lactonization is achieved by heating the ammonium salt in a hydrocarbon solvent like toluene at approximately 105°C. This thermal treatment drives the equilibrium towards the closed lactone ring, yielding Simvastatin with a purity exceeding 99%. The mechanistic elegance of avoiding external protecting groups means fewer impurities are introduced from reagent degradation, simplifying the downstream purification burden significantly.
How to Synthesize Simvastatin Efficiently
The synthesis of Simvastatin via this patented route requires precise control over reaction conditions, particularly temperature and stoichiometry, to maximize yield and minimize byproduct formation. The process begins with the amidation of the starting material, followed by the critical low-temperature lithiation and methylation sequence. Operators must ensure anhydrous conditions are maintained throughout the alkylation phase to prevent quenching of the organolithium species. Following the methylation, the hydrolysis step must be carefully monitored to ensure complete conversion to the ammonium salt before proceeding to the final thermal cyclization. For detailed operational parameters, stoichiometry ratios, and specific workup procedures, please refer to the standardized synthesis guide below.
- React Lovastatin or Mevinolinic acid salt with cyclopropylamine or n-butylamine to form the open-ring amide intermediate (III).
- Perform alpha-methylation using n-Butyl Lithium and methyl iodide in THF at low temperature (-35°C to -40°C) to generate the dimethyl side chain.
- Hydrolyze the amide to the ammonium salt (V) using NaOH, followed by acidification and extraction.
- Execute thermal lactonization in toluene at 105°C to close the pyranone ring and crystallize pure Simvastatin.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this protection-free synthesis route offers substantial advantages for supply chain stability and cost management in the production of pharmaceutical intermediates. By eliminating the requirement for expensive silylating agents and the associated solvents needed for their removal, the raw material cost profile is drastically improved. Furthermore, the reduction in the total number of unit operations translates directly into lower utility consumption and reduced labor hours per kilogram of finished product. This streamlined workflow enhances the overall throughput capacity of manufacturing facilities, allowing for faster turnaround times and more reliable delivery schedules for downstream API manufacturers who depend on consistent supply chains.
- Cost Reduction in Manufacturing: The most significant economic driver of this process is the complete avoidance of hydroxyl protection and deprotection steps. In traditional synthesis, silyl chlorides are costly reagents that generate stoichiometric amounts of salt waste, requiring extensive washing and disposal protocols. By removing these steps, the process achieves a substantial reduction in reagent expenditure and waste treatment costs. Additionally, the ability to use Mevinolinic acid salts directly as starting materials bypasses the yield loss associated with the natural lactonization of the fermentation broth, effectively increasing the mass balance efficiency of the entire production line without requiring additional capital investment.
- Enhanced Supply Chain Reliability: The simplified reaction sequence reduces the dependency on a complex array of specialized chemicals, many of which may be subject to market volatility or supply constraints. Relying on commodity chemicals like amines, methyl iodide, and standard hydrocarbon solvents creates a more resilient supply chain that is less susceptible to disruptions. The robustness of the ammonium salt precipitation step also ensures that intermediate quality is maintained even if minor variations occur in upstream fermentation batches, providing a buffer that stabilizes the final output quality and ensures continuous availability for global pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing common industrial solvents like toluene and tetrahydrofuran which are easily recovered and recycled in standard distillation units. The elimination of silicon-based waste streams significantly lowers the environmental footprint of the manufacturing process, aligning with increasingly stringent global regulations regarding hazardous waste disposal. This green chemistry advantage not only reduces compliance costs but also positions the manufacturer as a sustainable partner, which is a critical factor for multinational corporations aiming to reduce the carbon intensity of their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis route, focusing on the specific advantages of the amide intermediate strategy and the operational parameters required for successful execution. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the technology's applicability to your specific manufacturing context.
Q: Why is the amide intermediate strategy superior to silyl protection?
A: Traditional methods require expensive silylating agents to protect hydroxyl groups, adding two extra steps (protection and deprotection). The amide strategy utilizes the amine to naturally open the lactone ring, allowing direct methylation without auxiliary protecting groups, significantly reducing reagent costs and waste.
Q: What are the critical temperature controls for the methylation step?
A: Precision is vital. The reaction with n-Butyl Lithium must be maintained between -35°C and -40°C to prevent side reactions. Subsequent addition of methyl iodide requires careful warming to -10°C to ensure complete conversion while maintaining selectivity.
Q: Can Mevinolinic acid salts be used directly instead of Lovastatin?
A: Yes, the process explicitly supports using Mevinolinic acid salts (such as ammonium salts) as starting materials. This bypasses the natural fermentation loss associated with converting the acid to the lactone (Lovastatin) first, thereby improving overall material efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Simvastatin Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of advanced synthesis routes like the one described in CN1102588C are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Simvastatin intermediate meets the highest global pharmacopeial standards, providing you with the confidence needed for regulatory filings.
We invite you to engage with our technical procurement team to discuss how this optimized manufacturing process can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic impact of switching to this protection-free methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a seamless partnership that drives value and efficiency in your statin production portfolio.
