Revolutionizing Benzimidazole Synthesis: Scalable Iron-Catalyzed Redox Coupling for Global Supply Chains
Revolutionizing Benzimidazole Synthesis: Scalable Iron-Catalyzed Redox Coupling for Global Supply Chains
The pharmaceutical and agrochemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways for synthesizing critical heterocyclic scaffolds. Patent CN111704579B introduces a groundbreaking method for synthesizing benzimidazole compounds based on an iron-catalyzed redox coupling reaction. This technology represents a significant paradigm shift from traditional condensation methods, utilizing readily available o-nitroaniline and alcohol compounds as direct raw materials. By leveraging an inexpensive iron catalyst and a proton donor, this process achieves high yields under mild conditions, effectively bypassing the need for toxic dehydrating agents and expensive precursors. For global procurement and R&D teams, this innovation offers a robust solution for securing high-purity intermediates while drastically simplifying the supply chain logistics associated with complex organic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the industrial production of benzimidazole compounds relies heavily on the condensation cyclization of o-phenylenediamine with carboxylic acids or their derivatives. This conventional pathway is fraught with significant economic and environmental inefficiencies that burden modern supply chains. The prerequisite raw material, o-phenylenediamine, is itself produced via the reduction of o-nitroaniline, adding an unnecessary upstream processing step that increases both cost and waste generation. Furthermore, the carboxylic acid components often require separate oxidation processes from alcohols, compounding the carbon footprint. The reliance on high-temperature conditions and toxic dehydrating agents not only escalates operational hazards but also complicates waste treatment protocols, leading to higher compliance costs for manufacturers striving to meet stringent environmental standards.
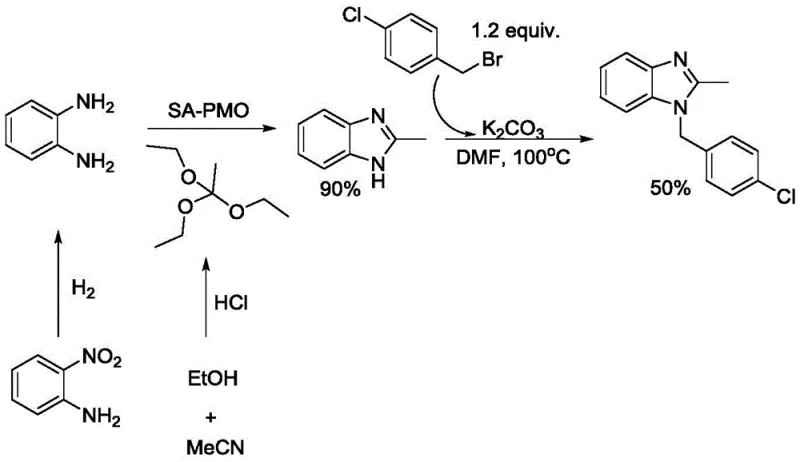
The Novel Approach
In stark contrast, the novel iron-catalyzed redox coupling reaction described in the patent streamlines the synthesis into a more direct and atom-economical process. By employing o-nitroaniline and simple alcohols like methanol or ethanol as the primary carbon and nitrogen sources, this method effectively merges the reduction and oxidation steps into a single catalytic cycle. The use of earth-abundant iron powder as a catalyst precursor eliminates the dependency on precious metals, thereby stabilizing raw material costs against market volatility. This approach not only improves the overall yield, as demonstrated by comparative data showing significant efficiency gains over traditional routes, but also drastically reduces the generation of hazardous by-products. The result is a greener, more economical manufacturing process that aligns perfectly with the sustainability goals of modern chemical enterprises.
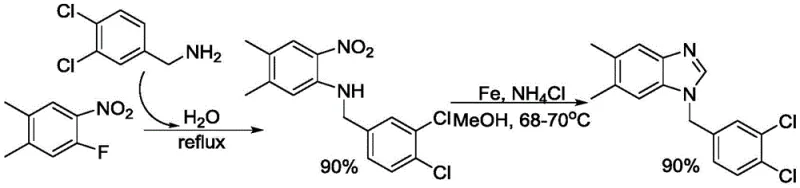
Mechanistic Insights into Iron-Catalyzed Redox Coupling
The core of this technological advancement lies in the sophisticated mechanistic interplay between the iron catalyst, the nitro group, and the alcohol solvent. The iron catalyst, generated in situ from iron powder, facilitates a dual-function redox process where the alcohol serves as both the solvent and the reducing agent. During the reaction, the alcohol is oxidized to an aldehyde intermediate, which simultaneously provides the necessary electrons to reduce the nitro group of the o-nitroaniline to an amine. This intramolecular or intermolecular coupling occurs seamlessly within the same reaction vessel, driven by the presence of a proton donor such as ammonium chloride. The mild reaction temperature of 68-70°C ensures that the delicate balance of oxidation and reduction is maintained without degrading sensitive functional groups, allowing for a broad substrate scope that includes various substituted anilines and alcohols.
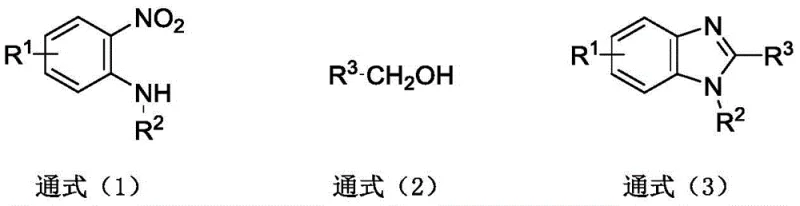
From an impurity control perspective, this mechanism offers distinct advantages over acid-catalyzed condensations. Traditional methods often suffer from side reactions involving the over-oxidation of aldehydes to carboxylic acids or the polymerization of reactive intermediates under harsh acidic conditions. The iron-catalyzed system operates under neutral to slightly acidic conditions mediated by the proton donor, which suppresses these unwanted pathways. The in situ generation of the active iron species ensures a consistent catalytic environment, minimizing the formation of metal-complexed impurities that are difficult to remove. Consequently, the crude product obtained after simple filtration and extraction exhibits high purity, reducing the burden on downstream purification units and ensuring that the final API intermediate meets rigorous quality specifications required by regulatory bodies.
How to Synthesize Benzimidazole Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize efficiency and safety. The process begins with the precise charging of o-nitroaniline, the proton donor, and the iron catalyst precursor into a reaction vessel under an inert atmosphere to prevent premature oxidation of the iron. Following the initial setup, the reaction mixture is heated to the optimal temperature range, and the alcohol is introduced to initiate the catalytic cycle. The detailed standardized synthesis steps, including specific molar ratios, stirring speeds, and quenching procedures, are critical for reproducing the high yields reported in the patent documentation. Operators must ensure strict temperature control and adequate stirring to maintain the suspension of the iron catalyst throughout the reaction period.
- Prepare the reaction mixture by adding o-nitroaniline, proton donor, and iron catalyst precursor into a reaction vessel under inert gas protection.
- Heat the oil bath to the target temperature range of 68-70°C and add the alcohol compound to initiate the redox coupling.
- Stir the mixture for 24-48 hours, then quench with saturated sodium bicarbonate, filter, extract, and concentrate to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iron-catalyzed technology translates into tangible strategic benefits beyond mere technical novelty. The shift from expensive, multi-step precursor synthesis to a direct coupling reaction fundamentally alters the cost structure of benzimidazole manufacturing. By eliminating the need for pre-synthesized o-phenylenediamine and specialized carboxylic acid derivatives, companies can source cheaper, commodity-grade raw materials that are less susceptible to supply disruptions. The simplified workflow also reduces the number of unit operations required, leading to lower energy consumption and reduced labor costs per kilogram of product. These factors collectively contribute to a more resilient and cost-competitive supply chain capable of withstanding market fluctuations.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and toxic dehydrating agents results in substantial cost savings on raw material procurement. Since the catalyst is derived from inexpensive iron powder and the solvent acts as a reactant, the overall material cost is drastically simplified. This economic efficiency allows for more competitive pricing strategies without compromising on the quality or purity of the final pharmaceutical intermediate, providing a clear margin advantage in tender negotiations.
- Enhanced Supply Chain Reliability: Relying on widely available commodity chemicals like methanol, ethanol, and iron powder mitigates the risk of supply chain bottlenecks often associated with specialized fine chemical intermediates. The robustness of the reaction conditions means that production can be scaled across multiple geographic locations without requiring highly specialized infrastructure. This flexibility ensures continuous supply continuity for downstream API manufacturers, reducing the risk of production delays due to raw material shortages.
- Scalability and Environmental Compliance: The mild operating conditions and absence of hazardous reagents make this process inherently safer and easier to scale from kilogram to tonne quantities. The reduction in three-waste pollution aligns with increasingly strict global environmental regulations, minimizing the costs associated with waste disposal and environmental compliance audits. This sustainability profile enhances the corporate reputation of suppliers and facilitates smoother regulatory approvals for new drug filings.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iron-catalyzed redox coupling technology. These insights are derived directly from the patent's experimental data and comparative analysis, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing manufacturing portfolios. The answers reflect the consensus on the method's superiority in terms of atom economy and operational simplicity compared to legacy processes.
Q: What are the primary advantages of using iron catalysts over traditional methods for benzimidazole synthesis?
A: The iron-catalyzed method eliminates the need for expensive and toxic o-phenylenediamine precursors and carboxylic acid derivatives. It utilizes cheap, earth-abundant iron powder and simple alcohols, significantly reducing raw material costs and three-waste pollution while maintaining high yields.
Q: How does this redox coupling reaction improve atom utilization compared to condensation cyclization?
A: Traditional condensation requires pre-oxidized carboxylic acids and pre-reduced diamines, involving separate energy-intensive steps. This novel approach couples the oxidation of alcohol and reduction of nitro groups in a single pot, directly forming the benzimidazole core with higher atom economy and fewer processing steps.
Q: Is this synthesis method suitable for large-scale commercial production of pharmaceutical intermediates?
A: Yes, the process operates at mild temperatures (68-70°C) and atmospheric pressure without requiring high-pressure hydrogenation or hazardous dehydrating agents. The use of stable iron powder and common solvents like methanol makes it highly scalable and compliant with strict environmental and safety regulations for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iron-catalyzed synthesis route for the global pharmaceutical market. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring such innovative technologies to fruition. Our facilities are equipped with rigorous QC labs and stringent purity specifications to ensure that every batch of benzimidazole intermediate meets the highest international standards. We are committed to leveraging this advanced chemistry to deliver high-purity products that accelerate our clients' drug development timelines while optimizing their cost structures.
We invite forward-thinking R&D and procurement leaders to collaborate with us on optimizing their supply chains through this cutting-edge technology. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of how this method can specifically benefit your project economics. Our technical procurement team is ready to provide specific COA data and route feasibility assessments tailored to your unique molecular requirements. Contact us today to explore how we can support your journey from bench-scale discovery to commercial success with reliable, high-quality chemical solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
