Scalable Production of 2-Trifluoromethyl Benzimidazoles Using Recyclable Copper Catalysts
Scalable Production of 2-Trifluoromethyl Benzimidazoles Using Recyclable Copper Catalysts
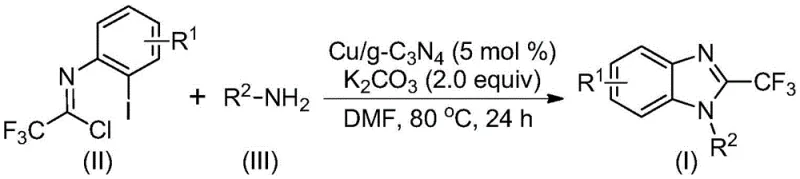
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable, and environmentally benign synthetic routes for nitrogen-containing heterocycles, particularly benzimidazoles, which serve as critical scaffolds in numerous bioactive molecules. A significant technological advancement in this domain is detailed in patent CN113429349A, which discloses a novel preparation method for 2-trifluoromethyl substituted benzimidazole compounds utilizing a heterogeneous copper-doped carbon nitride catalyst. This innovation addresses long-standing challenges in heterocycle synthesis by replacing traditional homogeneous catalytic systems with a recyclable, solid-state alternative that operates under remarkably mild conditions. For R&D directors and process chemists, this represents a paradigm shift towards more sustainable manufacturing, offering a pathway to high-purity intermediates with simplified downstream processing. The introduction of the trifluoromethyl group is particularly strategic, as it enhances the metabolic stability, lipophilicity, and bioavailability of the final active pharmaceutical ingredients (APIs), making this methodology highly relevant for the development of next-generation therapeutics and crop protection agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the benzimidazole core, especially when functionalized with electron-withdrawing groups like trifluoromethyl, has relied on methodologies that are often fraught with operational difficulties and economic inefficiencies. Traditional routes frequently involve the condensation of o-phenylenediamines with trifluoroacetic acid derivatives or reductive cyclizations that require harsh reaction conditions, strong acids, or expensive oxidants. Furthermore, earlier copper-catalyzed approaches typically employed homogeneous catalysts, which, while effective, pose significant challenges in product isolation and catalyst recovery. The presence of residual copper in the final product is a major regulatory concern for pharmaceutical applications, necessitating costly and time-consuming purification steps to meet stringent heavy metal limits. Additionally, many conventional methods suffer from narrow substrate scope, failing to tolerate diverse functional groups, which limits their utility in the rapid synthesis of compound libraries required for modern drug discovery programs.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN113429349A utilizes a copper-doped graphitic carbon nitride (Cu/g-C3N4) heterogeneous catalyst to drive the cascade cyclization of trifluoroacetimide chlorides and amines. This approach fundamentally alters the process economics and environmental profile of the synthesis. By employing a solid catalyst, the reaction facilitates easy separation via simple filtration, allowing the catalyst to be recovered and reused multiple times without significant loss of activity, thereby drastically reducing the cost per kilogram of the produced intermediate. The reaction proceeds efficiently in polar aprotic solvents like DMF at moderate temperatures ranging from 70°C to 90°C, avoiding the thermal stress and safety hazards associated with high-temperature protocols. This mildness not only preserves sensitive functional groups on the substrate but also minimizes the formation of thermal degradation byproducts, resulting in cleaner reaction profiles and higher crude purity.
Mechanistic Insights into Cu/g-C3N4 Catalyzed Cyclization
The mechanistic pathway of this transformation is a sophisticated interplay of nucleophilic substitution and transition metal-mediated C-N bond formation, orchestrated by the unique electronic properties of the copper-doped carbon nitride surface. Initially, the amine nucleophile attacks the electrophilic carbon of the trifluoroacetimide chloride, undergoing a nucleophilic addition-elimination sequence to generate an amidine intermediate in situ. Subsequently, the copper species embedded within the carbon nitride matrix activates the aryl carbon-iodine bond of the intermediate, forming a transient organocopper complex. This activation is critical, as it lowers the energy barrier for the subsequent intramolecular cyclization. Under the basic conditions provided by potassium carbonate, a molecule of hydrogen iodide is eliminated, facilitating the formation of a bivalent or trivalent cyclic copper intermediate. The final step involves a reductive elimination that releases the desired 2-trifluoromethyl benzimidazole product and regenerates the active copper catalytic site, completing the cycle. This mechanism ensures high atom economy and selectivity, as the heterogeneous support helps stabilize the reactive copper species, preventing aggregation and deactivation.
From an impurity control perspective, this mechanism offers distinct advantages over solution-phase homogeneous catalysis. The confined environment of the carbon nitride support can impose steric constraints that favor the desired cyclization pathway over competing side reactions, such as polymerization or hydrolysis of the imidoyl chloride. Furthermore, the use of mild bases like potassium carbonate, rather than strong alkoxides or hydrides, minimizes base-sensitive decomposition pathways. For the R&D director, this translates to a more predictable impurity profile, simplifying the analytical characterization and validation of the process. The ability to tune the electronic density of the catalyst by varying the copper loading or the carbon nitride precursor provides an additional handle for optimizing selectivity, ensuring that even complex substrates with multiple reactive sites can be converted cleanly into the target benzimidazole scaffold with minimal byproduct formation.
How to Synthesize 2-Trifluoromethyl Benzimidazoles Efficiently
The practical implementation of this synthesis is designed for ease of operation, requiring standard laboratory equipment and commercially available reagents, making it highly accessible for both pilot-scale studies and full commercial production. The protocol involves a straightforward one-pot procedure where the catalyst, base, and substrates are combined in a solvent, eliminating the need for inert atmosphere techniques or specialized pressure vessels. This simplicity reduces the training burden on operators and minimizes the risk of batch-to-batch variability caused by complex handling procedures. The following section outlines the standardized steps derived from the patent examples to ensure reproducibility and optimal yield.
- Prepare the reaction mixture by adding copper-doped carbon nitride (Cu/g-C3N4), potassium carbonate, trifluoroethylimidoyl chloride, and the chosen amine into an organic solvent such as DMF.
- Heat the reaction mixture to a temperature between 70°C and 90°C and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture to recover the heterogeneous catalyst, mix the filtrate with silica gel, and purify via column chromatography to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this heterogeneous catalytic process offers compelling strategic benefits that extend beyond mere chemical yield. The shift from homogeneous to heterogeneous catalysis fundamentally reshapes the cost structure of manufacturing by eliminating the need for expensive ligand systems and complex metal scavenging resins that are typically required to purge homogeneous copper residues. This simplification of the downstream processing train directly correlates to reduced operational expenditures (OpEx) and shorter cycle times, allowing facilities to increase throughput without capital investment in new equipment. Moreover, the robustness of the catalyst means that supply chain disruptions related to specialized reagent availability are mitigated, as the catalyst can be synthesized in-house from abundant precursors like urea and copper salts, ensuring long-term supply continuity for critical intermediates.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the recyclability of the Cu/g-C3N4 catalyst, which can be recovered via simple filtration and reused for multiple cycles with maintained efficiency. This eliminates the recurring cost of purchasing fresh catalyst for every batch, a common expense in homogeneous systems. Additionally, the use of inexpensive inorganic bases like potassium carbonate and common solvents like DMF further drives down the raw material costs. The high conversion rates observed, often exceeding 90% for a broad range of substrates, mean that less starting material is wasted, maximizing the return on investment for every kilogram of reagent purchased and reducing the volume of waste solvent that requires disposal.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, specifically trifluoroacetimide chlorides and various primary amines, are commodity chemicals with well-established global supply chains, reducing the risk of sourcing bottlenecks. Unlike proprietary catalysts that may be sourced from a single vendor, the components of the Cu/g-C3N4 catalyst are widely available, granting procurement teams greater negotiating power and flexibility. The mild reaction conditions also reduce the strain on utility infrastructure, such as heating and cooling systems, making the process less susceptible to disruptions caused by facility maintenance or energy supply fluctuations, thereby ensuring a more reliable delivery schedule to downstream customers.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is inherently safer and more manageable due to the absence of pyrophoric reagents or extreme pressures. The heterogeneous nature of the catalyst simplifies the transition to continuous flow chemistry, a key enabler for modern green manufacturing that offers superior heat and mass transfer. From an environmental compliance standpoint, the ability to recycle the catalyst significantly reduces the heavy metal load in the effluent, easing the burden on wastewater treatment facilities and helping the organization meet increasingly stringent environmental regulations. This green chemistry profile not only avoids potential fines but also enhances the corporate sustainability image, which is increasingly valued by end-users in the pharmaceutical and agrochemical sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this heterogeneous catalytic technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities and limitations for potential adopters.
Q: What are the primary advantages of using Cu/g-C3N4 over homogeneous copper catalysts?
A: The use of copper-doped carbon nitride (Cu/g-C3N4) offers significant operational advantages, primarily due to its heterogeneous nature which allows for easy filtration and recycling. Unlike homogeneous catalysts that require complex removal processes and generate heavy metal waste, this solid catalyst can be recovered and reused multiple times with only partial loss of efficiency, drastically simplifying post-treatment and reducing environmental impact.
Q: What is the substrate scope for this benzimidazole synthesis method?
A: This methodology demonstrates excellent substrate tolerance, accommodating a wide range of amines including aliphatic chains (such as n-butyl, n-pentyl, and cyclohexyl) and aromatic groups (like benzyl and phenyl). Furthermore, the aryl ring of the imidoyl chloride can bear various substituents such as halogens (Cl, Br), methyl, or trifluoromethyl groups, allowing for the diverse functionalization required in modern drug discovery.
Q: How does this process improve cost efficiency in manufacturing?
A: Cost efficiency is achieved through the use of cheap and readily available starting materials like trifluoroethylimidoyl chloride and common amines, coupled with the recyclability of the catalyst. The mild reaction conditions (70-90°C) reduce energy consumption compared to traditional high-temperature methods, and the simplified workup procedure minimizes solvent usage and labor time, leading to substantial overall cost reductions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Benzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating the time-to-market for new therapeutic agents. Our technical team has extensively evaluated the Cu/g-C3N4 catalytic system described in CN113429349A and validated its potential for large-scale application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl benzimidazole intermediate meets the highest quality standards required by global regulatory bodies.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this greener route can optimize your budget. We encourage you to contact us today to discuss your specific COA data requirements and to receive comprehensive route feasibility assessments that will empower your R&D and supply chain strategies with confidence and precision.
