Advanced Simvastatin Manufacturing: A Technical Breakthrough for Global Supply Chains
Advanced Simvastatin Manufacturing: A Technical Breakthrough for Global Supply Chains
The global demand for HMG-CoA reductase inhibitors, particularly Simvastatin, continues to drive innovation in pharmaceutical manufacturing processes. Patent CN1226296C discloses a highly efficient and industrially viable method for producing Simvastatin from Lovastatin, addressing critical bottlenecks in traditional synthesis routes. This technical insight report analyzes the novel deacylation and selective protection strategy outlined in the patent, offering a roadmap for R&D directors and procurement leaders seeking to optimize their supply chains. By replacing harsh hydrolysis conditions with mild inorganic base treatments and utilizing robust protecting groups, this process significantly enhances yield and purity while reducing operational complexity. As a reliable simvastatin intermediate supplier, understanding these mechanistic nuances is essential for securing a competitive edge in the cardiovascular drug market.
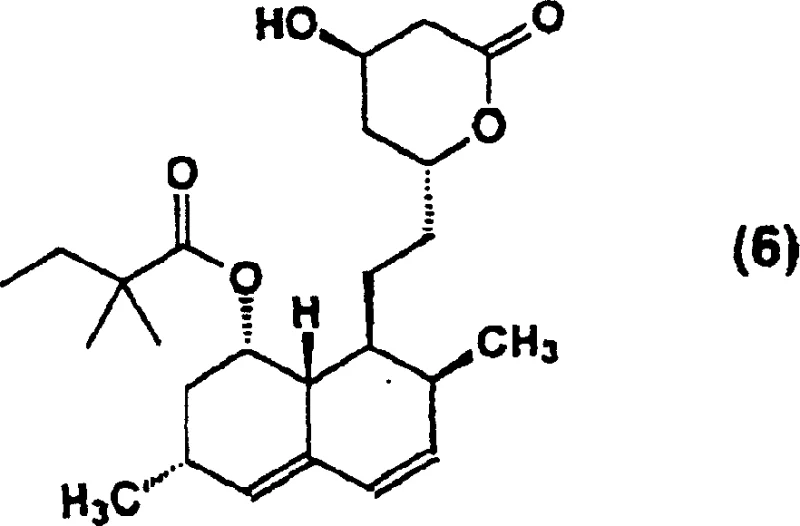
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional pathways for converting Lovastatin to Simvastatin have long been plagued by inefficiencies that hinder large-scale production. Prior art methods, such as those described in USP 4444784, typically rely on lithium hydroxide for hydrolysis, necessitating high temperatures and extended reaction times that degrade overall productivity. Furthermore, the selective silylation steps often suffer from poor reaction selectivity, leading to a mixture of by-products that require cumbersome purification protocols to isolate the desired diol lactone. Other approaches involving direct methylation of Lovastatin potassium salts or monoalkylamides frequently result in incomplete conversion of the raw material, forcing manufacturers to implement complex recycling or disposal procedures for unreacted starting materials. Additionally, certain legacy processes mandate extremely low reaction temperatures, imposing severe energy burdens and requiring specialized cryogenic equipment that escalates capital expenditure. These cumulative drawbacks create significant barriers to achieving cost-effective, high-volume manufacturing of this critical cardiovascular therapeutic.
The Novel Approach
The methodology presented in patent CN1226296C introduces a paradigm shift by employing inorganic bases in conjunction with secondary or tertiary alcohols to effectuate deacylation under remarkably mild conditions. Instead of relying on expensive organolithium reagents or extreme thermal inputs, this process utilizes accessible bases like potassium hydroxide in solvents such as tert-butanol or isopropanol at moderate temperatures ranging from 60°C to 100°C. This adjustment not only accelerates the reaction kinetics but also preserves the structural integrity of the sensitive hexahydronaphthalene ring system. Following deacylation, the resulting triol acid is seamlessly converted into a diol lactone through acidification and heating, avoiding the isolation of unstable intermediates. The subsequent use of ketal or acetal protecting groups ensures exceptional regioselectivity during the acylation phase, effectively shielding the 4-hydroxyl group while allowing precise modification at the 8-position. This strategic sequence eliminates the need for cryogenic conditions and drastically simplifies the downstream workup, positioning this route as a superior alternative for modern pharmaceutical intermediate manufacturing.
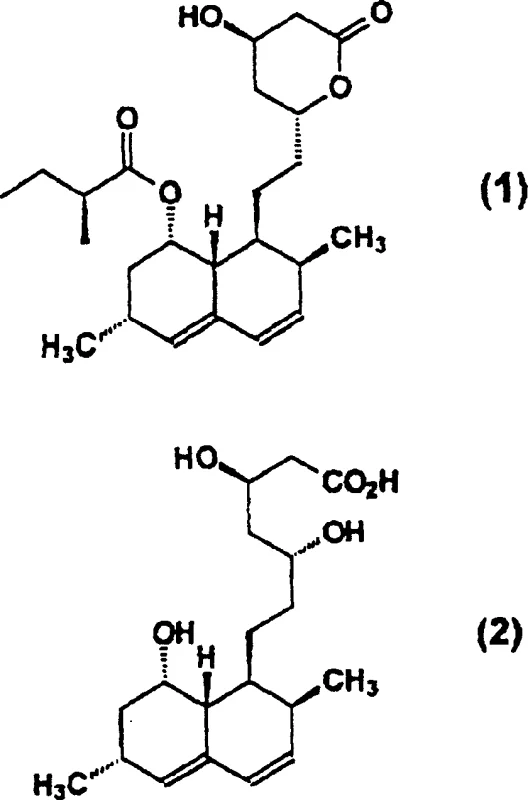
Mechanistic Insights into Deacylation and Selective Protection
The core innovation of this synthesis lies in the controlled deacylation of the Lovastatin side chain. When Lovastatin is treated with an inorganic base like KOH in a secondary alcohol solvent, the ester linkage at the 2'-position undergoes nucleophilic attack, cleaving the 2-methylbutyrate group to form a triol acid intermediate. Unlike aqueous hydrolysis which can lead to ring opening of the lactone moiety indiscriminately, the use of alcoholic solvents moderates the basicity and solvation environment, favoring the formation of the open-chain triol acid which can then be recyclized. The subsequent lactonization is driven by acid catalysis and thermal energy in organic solvents like isopropyl acetate, promoting the intramolecular esterification to yield the stable diol lactone. This two-step telescoped operation minimizes handling losses and exposure to moisture, which is critical for maintaining stereochemical purity at the chiral centers.
Following the formation of the diol lactone, the process employs a sophisticated protection strategy using dialkoxy ketals or acetals, such as 2,2-dimethoxypropane. In the presence of an acid catalyst like p-toluenesulfonic acid, the 4-hydroxyl and carboxyl groups (or the lactone oxygen in equilibrium) react to form a cyclic acetal or ketal derivative. This temporary masking of the 4-position is the linchpin of the entire synthesis; it prevents unwanted acylation at this site during the subsequent introduction of the 2,2-dimethylbutyryl side chain. The steric bulk of the protecting group combined with the electronic deactivation of the masked oxygen ensures that the acyl chloride reacts exclusively at the 8-hydroxyl position. Finally, the removal of the protecting group under acidic protic conditions regenerates the free hydroxyl and facilitates the final lactonization to yield Simvastatin. This mechanism ensures that the final product possesses the exact stereochemistry and substitution pattern required for potent HMG-CoA reductase inhibition.
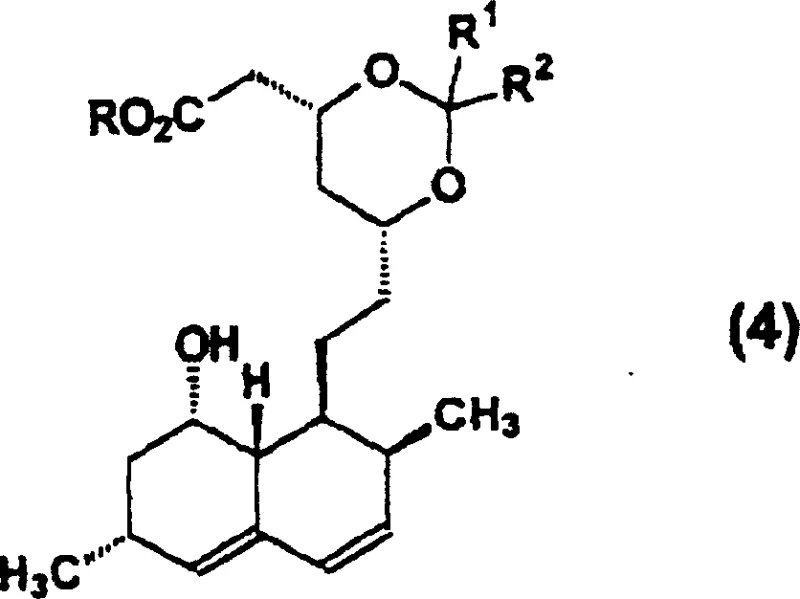
How to Synthesize Simvastatin Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize the yield of the protected intermediates and the final API. The process begins with the deacylation of Lovastatin using potassium hydroxide in tert-butanol at reflux, followed by acidification to pH 3.5 and extraction. The crude triol acid is then lactonized in isopropyl acetate with methanesulfonic acid to afford the diol lactone in high purity. Subsequent protection with 2,2-dimethoxypropane and acylation with 2,2-dimethylbutyryl chloride are conducted in organic solvents like dichloromethane or toluene, utilizing pyridine and DMAP as base catalysts. The final deprotection step utilizes dilute hydrochloric acid in acetonitrile or aqueous toluene systems to reveal the active drug substance. For detailed operational parameters, stoichiometry, and safety protocols, please refer to the standardized synthesis guide below.
- Deacylate Lovastatin using an inorganic base and secondary or tertiary alcohol to form a triol acid, followed by acidification and lactonization to yield diol lactone.
- Selectively protect the diol lactone using a ketal or acetal protecting group (e.g., 2,2-dimethoxypropane) to form a triol acid derivative.
- Acylate the protected derivative with 2,2-dimethylbutyryl chloride, followed by deprotection and final lactonization to obtain Simvastatin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates into tangible strategic benefits that extend beyond mere chemical yield. By eliminating the dependency on cryogenic cooling and expensive organometallic reagents, the manufacturing footprint is significantly simplified, leading to substantial cost reduction in pharmaceutical intermediate manufacturing. The use of commodity chemicals such as potassium hydroxide, isopropanol, and toluene ensures that raw material sourcing remains stable and resilient against market volatility, unlike specialized silylating agents which can face supply constraints. Furthermore, the high conversion rates reported in the patent examples—such as yields exceeding 90% for the protected intermediate—indicate a robust process with minimal waste generation, directly impacting the cost of goods sold (COGS). This efficiency allows suppliers to offer more competitive pricing structures while maintaining healthy margins, a critical factor for long-term contracting in the generic pharmaceutical sector.
- Cost Reduction in Manufacturing: The replacement of lithium hydroxide with cheaper inorganic bases like potassium hydroxide, combined with the elimination of energy-intensive low-temperature steps, drastically lowers utility and reagent costs. The process avoids the use of costly silyl protecting groups (like TBDS) which require expensive fluorine-based reagents for removal, substituting them with economical acetals that are easily cleaved with dilute acid. This fundamental shift in reagent selection removes the need for specialized waste treatment for heavy metals or fluorine compounds, further reducing environmental compliance costs and enhancing the overall economic viability of the production line.
- Enhanced Supply Chain Reliability: Sourcing stability is paramount for continuous API production, and this route relies heavily on bulk commodities that are widely available from multiple global vendors. The solvents employed, such as ethyl acetate, toluene, and acetonitrile, are standard industrial grades with established logistics networks, minimizing the risk of shipment delays. Additionally, the robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in temperature or reagent quality, ensuring consistent batch-to-batch performance. This reliability reduces the likelihood of production stoppages due to out-of-specification raw materials, thereby securing the continuity of supply for downstream formulation partners.
- Scalability and Environmental Compliance: The mild reaction conditions and the use of recyclable solvents make this process inherently scalable from pilot plant to multi-ton commercial production. The avoidance of hazardous reagents simplifies the safety profile of the facility, lowering insurance premiums and regulatory scrutiny. Moreover, the high selectivity of the acylation step minimizes the formation of difficult-to-remove impurities, reducing the solvent load required for chromatographic purification. This aligns with green chemistry principles by lowering the E-factor (mass of waste per mass of product), facilitating easier permitting and supporting corporate sustainability goals related to waste minimization and energy efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Simvastatin synthesis technology. These answers are derived directly from the experimental data and claims within patent CN1226296C, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of using inorganic bases over lithium hydroxide for Lovastatin deacylation?
A: Using inorganic bases like potassium hydroxide with secondary or tertiary alcohols allows the reaction to proceed under milder conditions (60-100°C) compared to the high temperatures and long reaction times required for lithium hydroxide hydrolysis, resulting in higher productivity and yield.
Q: How does this process improve the purity of the final Simvastatin product?
A: The method employs selective protection using ketal or acetal groups which prevents side reactions during the acylation step. This specificity minimizes impurity formation, leading to a final product with high purity (demonstrated at ≥93% in examples) without complex purification steps.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process is designed for industrial convenience. It avoids extremely low temperatures and expensive reagents, utilizes common solvents like isopropyl acetate and toluene, and achieves high conversion rates (e.g., ≥90% for intermediate steps), making it highly scalable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Simvastatin Supplier
At NINGBO INNO PHARMCHEM, we leverage deep technical expertise to bring complex synthetic routes like the one described in CN1226296C to commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art HPLC and NMR instrumentation to guarantee that every batch of Simvastatin meets the highest international pharmacopeial standards. Our commitment to quality assurance means that we can consistently deliver high-purity APIs that satisfy the demanding requirements of global regulatory bodies.
We invite potential partners to engage with our technical procurement team to discuss how this advanced manufacturing process can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into how switching to this methodology impacts your bottom line. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a partnership built on transparency, technical excellence, and mutual growth.
