Advanced Catalyst-Free Lactonization for High-Purity Statin Intermediates and Commercial Scale-Up
The pharmaceutical industry constantly seeks robust synthetic routes for critical cholesterol-lowering agents, specifically focusing on the lactonization processing method detailed in patent CN1425661A. This intellectual property addresses the persistent challenges in converting mevinic acid derivatives into their active lactone forms, such as lovastatin and simvastatin, which are essential pharmaceutical intermediates. Traditional synthesis often struggles with equilibrium limitations and impurity profiles, but this novel approach utilizes a mixed organic solvent system combined with a nitrogen sweep to drive the reaction efficiently without acidic catalysts. By eliminating the need for harsh chemical promoters, the process achieves superior purity levels while maintaining high yields, offering a compelling solution for manufacturers aiming to optimize their production of high-purity statin intermediates. The strategic implementation of this technology ensures that supply chains remain resilient against quality fluctuations common in older methodologies. This patent specifically targets the conversion of mevinolinic acid homologs, addressing the critical bottleneck where open-ring acids must be cyclized without degrading the sensitive lactone ring or forming dimeric impurities that compromise therapeutic efficacy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Conventional lactonization techniques frequently rely on aggressive conditions that compromise product integrity and operational efficiency. Historical methods, such as those described in U.S. Pat. No. 4,820,850, typically involve refluxing in toluene at elevated temperatures around 110°C, which inadvertently promotes the formation of unwanted heterodimer byproducts. Furthermore, alternative processes utilizing strong acid catalysts like methanesulfonic acid necessitate complex neutralization steps with large volumes of base, generating significant salt waste and complicating downstream filtration. These legacy approaches often result in heterodimer contents reaching approximately 0.4%, requiring extensive and yield-reducing recrystallization cycles to meet stringent pharmaceutical standards. The reliance on single-solvent systems or aqueous mixtures in prior art also leads to inhomogeneous crystallization and prolonged reaction times exceeding 9 to 12 hours, severely impacting throughput. Specifically, the addition of large amounts of water in prior art methods to induce crystallization often results in secondary crystallization on existing crystals, creating inhomogeneous particle sizes that are difficult to filter and dry efficiently. This not only reduces production efficiency but also traps solvent residues, necessitating extended drying cycles that further increase energy consumption and operational costs.
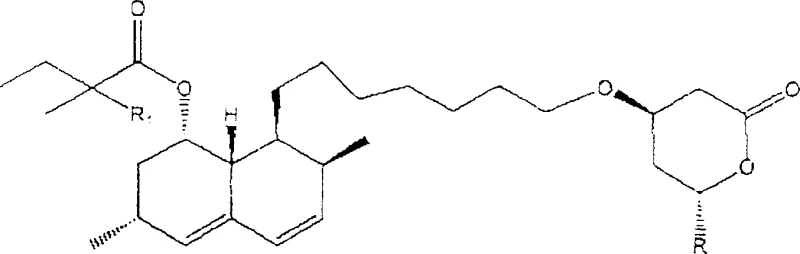
The Novel Approach
The innovative process disclosed in the patent overcomes these hurdles by employing a mixed organic solvent system, preferably a 1:1 volume ratio of toluene and acetonitrile or dichloromethane and acetonitrile, to create an optimal environment for lactonization. The continuous nitrogen purge serves a dual function: it maintains an inert atmosphere to prevent oxidation and actively removes volatile byproducts like water and ammonia from the reaction mixture. This physical removal shifts the equilibrium position decisively toward the lactone product according to Le Chatelier's principle, effectively driving the conversion of the open-ring dihydroxy acid form into the closed-ring lactone. Crucially, the absence of an acid catalyst prevents the protonation of the carbonyl group, which is the primary mechanism leading to the formation of heterodimers via esterification between the 3-hydroxyl groups of adjacent molecules. This mechanistic elegance ensures that the final product contains heterodimer impurities at levels below 0.05%, significantly lower than the 0.15% to 0.4% typical of acid-catalyzed routes. The selection of the solvent mixture is critical, as it must dissolve the starting ammonium salt effectively while allowing the lactone product to crystallize upon cooling or solvent adjustment.
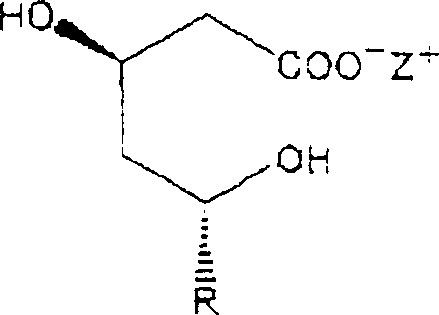
Mechanistic Insights into Nitrogen-Purge Driven Lactonization
The core innovation lies in the precise manipulation of reaction equilibrium through physical means rather than chemical catalysis. By employing a mixed organic solvent system, preferably a 1:1 volume ratio of toluene and acetonitrile or dichloromethane and acetonitrile, the process creates an optimal environment for lactonization. The continuous nitrogen purge serves a dual function: it maintains an inert atmosphere to prevent oxidation and actively removes volatile byproducts like water and ammonia from the reaction mixture. This physical removal shifts the equilibrium position decisively toward the lactone product according to Le Chatelier's principle, effectively driving the conversion of the open-ring dihydroxy acid form into the closed-ring lactone. Crucially, the absence of an acid catalyst prevents the protonation of the carbonyl group, which is the primary mechanism leading to the formation of heterodimers via esterification between the 3-hydroxyl groups of adjacent molecules. This mechanistic elegance ensures that the final product contains heterodimer impurities at levels below 0.05%, significantly lower than the 0.15% to 0.4% typical of acid-catalyzed routes. The selection of the solvent mixture is critical, as it must dissolve the starting ammonium salt effectively while allowing the lactone product to crystallize upon cooling or solvent adjustment. Preferred embodiments utilize a toluene and acetonitrile mixture or a dichloromethane and acetonitrile blend, which provide the ideal polarity balance to support the nitrogen-driven dehydration process while maintaining the stability of the sensitive hexahydronaphthalenyl moiety found in the statin structure.
Furthermore, the crystallization step is optimized to ensure high recovery and purity without the need for excessive solvent volumes. The patent describes using specific solvent mixtures for crystallization, such as ethanol and water or toluene and cyclohexane, in precise weight ratios relative to the starting material. For instance, a mixture containing 8 to 10 parts by weight of water and 8 to 10 parts by weight of ethanol per part of the dihydroxy compound allows for controlled crystal growth. If the amounts of these solvents exceed the specified range, impurities are not easily removed, and the product may fail to crystallize as a solid, which is not preferable. This precise control over the crystallization environment ensures that the lattice structure of the growing crystals excludes impurities effectively, resulting in a product that requires minimal further purification. The ability to achieve 99% HPLC purity directly from the crystallization step demonstrates the robustness of this mechanistic approach in managing impurity profiles.
How to Synthesize Simvastatin and Lovastatin Efficiently
The synthesis of these critical statin intermediates involves a streamlined sequence that begins with the reflux of the ammonium salt of mevinic acid in the chosen mixed solvent system under a nitrogen atmosphere. This initial step is crucial for driving the equilibrium toward the lactone form while simultaneously stripping away the water and ammonia generated during the cyclization. Following the reaction, the mixture is cooled, and activated carbon is often added to adsorb any colored impurities or trace organics before filtration. The filtrate is then concentrated under reduced pressure to adjust the solvent volume, preparing the solution for the final crystallization step. The detailed standardized synthetic steps, including specific temperatures, stirring times, and solvent ratios for both the reaction and crystallization phases, are outlined in the comprehensive guide below. This protocol ensures reproducibility and high yield, making it suitable for both laboratory optimization and industrial scale-up operations.
- Reflux the ammonium salt of mevinic acid in a mixed organic solvent (e.g., toluene/acetonitrile) under a nitrogen purge to remove water and ammonia.
- Cool the reaction mixture and add activated carbon to remove impurities, followed by filtration and volume reduction.
- Induce crystallization by adding a non-solvent or anti-solvent mixture, then filter and dry the high-purity lactone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement and supply chain leadership, the adoption of this catalyst-free lactonization protocol offers substantial strategic advantages in cost reduction in API manufacturing. The elimination of strong acid catalysts removes the entire unit operation dedicated to neutralization and salt removal, thereby drastically simplifying the workflow and reducing the consumption of auxiliary reagents. Since the process inherently suppresses the formation of difficult-to-remove heterodimer impurities, the reliance on multiple, yield-eroding recrystallization steps is significantly diminished, leading to a higher overall recovery of the final active pharmaceutical ingredient. From a supply continuity perspective, the use of common, commercially available solvents like toluene, acetonitrile, and cyclohexane ensures that raw material sourcing remains stable and unaffected by niche chemical shortages. Furthermore, the reduced generation of saline waste streams aligns with increasingly rigorous environmental compliance standards, lowering the burden on wastewater treatment facilities and mitigating regulatory risks associated with hazardous chemical handling. This streamlined approach facilitates the commercial scale-up of complex pharmaceutical intermediates with greater predictability and lower operational expenditure.
- Cost Reduction in Manufacturing: The removal of acid catalysts and the subsequent neutralization steps eliminates the cost of purchasing strong bases and the disposal of the resulting salt waste. Additionally, the higher purity of the crude product reduces the solvent and energy costs associated with repeated recrystallization cycles, leading to significant overall process economics improvements.
- Enhanced Supply Chain Reliability: By utilizing standard industrial solvents and avoiding specialized or hazardous reagents, the supply chain becomes more robust against disruptions. The simplified process flow also reduces the risk of batch failures due to operator error in complex neutralization or filtration steps, ensuring consistent delivery schedules.
- Scalability and Environmental Compliance: The process generates significantly less chemical waste, particularly saline effluents, which simplifies environmental permitting and waste management. The straightforward crystallization and filtration steps are easily scalable from pilot plant to multi-ton production, supporting the growing global demand for statin therapies without compromising ecological standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this lactonization technology in industrial settings. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of adopting this method for their specific production lines. The responses cover aspects of impurity control, solvent selection, and the comparative advantages over traditional acid-catalyzed routes.
Q: How does this process reduce heterodimer impurities compared to acid-catalyzed methods?
A: By avoiding acid catalysts, the process prevents the protonation of the lactone carbonyl group, which is the primary driver for heterodimer formation via esterification between 3-hydroxyl groups.
Q: What solvents are preferred for this lactonization reaction?
A: The patent specifies mixed organic solvents, preferably a 1:1 volume ratio of toluene and acetonitrile, or dichloromethane and acetonitrile, to optimize solubility and equilibrium shifting.
Q: What is the expected purity of the final statin lactone product?
A: The method yields products with HPLC purity of approximately 99% and heterodimer content reduced to below 0.05%, significantly lower than conventional methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lovastatin and Simvastatin Supplier
Partnering with NINGBO INNO PHARMCHEM provides access to this advanced lactonization technology for your reliable pharmaceutical intermediates supplier needs. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and efficient. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of lovastatin or simvastatin intermediate meets the highest global regulatory requirements. We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production volumes. By collaborating with us, you can secure specific COA data and route feasibility assessments that demonstrate how this patented method can enhance your competitive position in the global market for high-purity statin intermediates.
