Advanced Synthetic Route for Aztreonam: Enhancing Purity and Scalability for Global API Production
The pharmaceutical landscape continuously demands more efficient and environmentally benign pathways for the production of critical antibiotics, and the synthesis of Aztreonam stands as a prime example of this evolution. Patent CN101514200A introduces a transformative approach to manufacturing this vital monobactam antibiotic, addressing long-standing inefficiencies in prior art. This technical disclosure outlines a robust methodology that leverages readily available intermediates, specifically (2-aminothiazole-4-yl)-2-(tert-butoxycarbonyl)-iminodiacetic acid isopropoxide acid-2-ester mercaptobenzothiazole and (3-S-trans form)-3-amino-4-methyl-2-oxo-1-sulfonic acid azetidine. By shifting away from hazardous reagents towards a mixed aqueous solution of acetic acid and hydrochloric acid for deprotection, this innovation not only streamlines the reaction sequence but also significantly enhances the safety profile and economic viability of the process. For global supply chain leaders, this represents a pivotal opportunity to secure a more reliable source of high-purity active pharmaceutical ingredients while mitigating regulatory risks associated with toxic solvent residues.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Aztreonam has been plagued by significant operational hurdles and safety concerns inherent to traditional synthetic routes. Prior art, such as that described in U.S. Patent 4775670, relies heavily on the use of dicyclohexylcarbodiimide (DCC) as a condensing agent and trifluoroacetic acid (TFA) for deprotection steps. These reagents are not only prohibitively expensive but also introduce severe toxicity challenges that complicate waste management and worker safety protocols. Furthermore, alternative methods utilizing mineral acids like sulfuric acid have demonstrated suboptimal performance, often resulting in low yields due to the formation of undesirable ring-opened byproducts. The inability to effectively recover and reuse these costly reagents creates a substantial financial burden, rendering many conventional processes economically unsustainable for large-scale commercial operations in a competitive generic drug market.
The Novel Approach
In stark contrast to these legacy methodologies, the novel approach detailed in the patent data utilizes a sophisticated yet practical strategy centered on mild reaction conditions and accessible chemistry. By employing 1,4-dioxane as a primary solvent, which exhibits excellent miscibility with water, the process eliminates the need for complex distillation apparatuses typically required for solvent switching. The introduction of N-methylmorpholine serves to optimize reaction kinetics and improve overall product purity without the necessity for additional buffering agents like ammonium acetate. Most critically, the deprotection step is revolutionized by the use of a mixed aqueous solution comprising acetic acid and hydrochloric acid. This modification allows for the efficient removal of protecting groups under controlled conditions, preventing the degradation of the sensitive beta-lactam ring and ensuring a high-yield transformation that is far more amenable to industrial scaling than previous iterations.
Mechanistic Insights into Acid-Catalyzed Deprotection and Coupling
The core of this synthetic advancement lies in the precise manipulation of reaction parameters to facilitate the coupling of the thiazole side chain with the azetidine nucleus. The mechanism involves the nucleophilic attack of the amino group on the activated ester intermediate, a process that is carefully modulated by the presence of N-methylmorpholine to maintain optimal pH levels during the initial coupling phase. Following this, the strategic application of the acetic acid and hydrochloric acid mixture triggers the cleavage of the tert-butoxycarbonyl protecting group. Unlike anhydrous acidic conditions which can be harsh and unpredictable, this aqueous acidic environment provides a buffered medium that stabilizes the transition state, thereby minimizing side reactions such as hydrolysis of the beta-lactam ring. This delicate balance ensures that the stereochemical integrity of the molecule is preserved, which is paramount for maintaining the biological activity of the final antibiotic product.
Furthermore, the control of impurities is intrinsically linked to the choice of solvents and the specific workup procedures employed in this novel route. The use of ethyl acetate for extraction allows for the efficient separation of organic impurities from the desired aqueous phase containing the product salt. Subsequent adjustment of the pH to approximately 1 using hydrochloric acid induces the precipitation of the Aztreonam product in a highly pure form. The patent data indicates that this crystallization process is highly effective at excluding structurally related impurities, as evidenced by the high purity levels reported in the experimental examples. This mechanistic understanding underscores the robustness of the process, providing R&D directors with confidence that the method can consistently deliver material that meets stringent pharmacopeial standards without requiring extensive downstream purification.
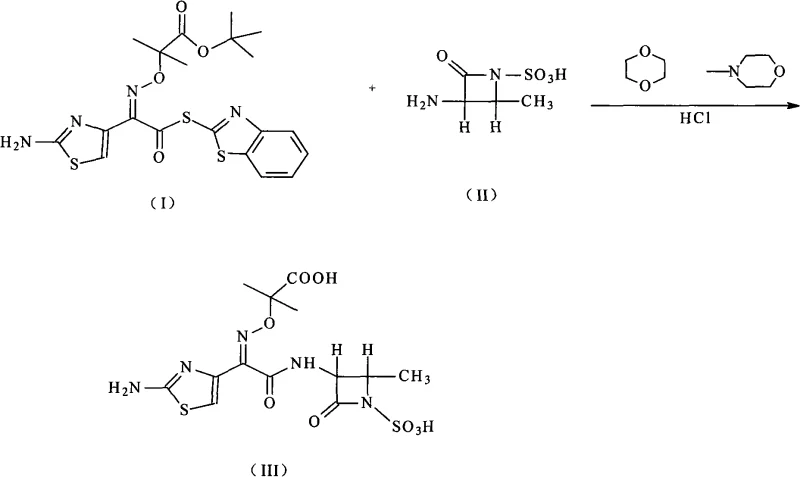
How to Synthesize Aztreonam Efficiently
Implementing this synthetic route requires strict adherence to the temperature and stoichiometric ratios defined in the patent to ensure reproducibility and safety. The process begins with the dissolution of the azetidine intermediate in 1,4-dioxane, followed by cooling to a precise temperature range to control exothermicity upon addition of the coupling partner. The subsequent steps involve careful phase separation and pH adjustment to drive the crystallization of the final product. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis protocol below which outlines the critical control points for successful manufacturing.
- Dissolve (3S-trans)-3-amino-4-methyl-2-oxo-1-azetidinyl sulfonic acid in 1,4-dioxane with N-methylmorpholine and cool to 0-10°C.
- Add the thiazole intermediate solution, stir for reaction, then add acetic acid aqueous solution and ethyl acetate for extraction.
- Adjust the aqueous phase pH to 1 using hydrochloric acid, heat to 30-50°C to induce crystallization, then filter and dry the solid product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic methodology offers profound strategic benefits that extend beyond simple cost metrics. The elimination of exotic and hazardous reagents such as DCC and TFA drastically simplifies the sourcing landscape, allowing purchasing managers to rely on a broader base of commodity chemical suppliers. This shift reduces the risk of supply disruptions caused by the limited availability of specialized reagents and mitigates the volatility associated with their pricing. Moreover, the simplified workup procedure, which avoids complex distillation and extensive solvent exchanges, translates directly into reduced processing time and lower energy consumption per kilogram of product produced. These operational efficiencies create a leaner manufacturing profile that enhances the overall agility of the supply chain, enabling faster response times to market demands.
- Cost Reduction in Manufacturing: The economic implications of replacing expensive condensing agents and deprotecting reagents with common acids and solvents are substantial. By removing the need for costly reagents that cannot be easily recovered, the direct material cost of goods sold is significantly lowered. Additionally, the ability to recover and reuse ethyl acetate further contributes to cost optimization, creating a more sustainable economic model for long-term production. This structural cost advantage allows manufacturers to offer more competitive pricing to downstream partners while maintaining healthy margins, a critical factor in the highly price-sensitive generic pharmaceutical sector.
- Enhanced Supply Chain Reliability: The reliance on standard industrial solvents like 1,4-dioxane and ethyl acetate ensures a high degree of supply security. Unlike specialized reagents that may have single-source suppliers or long lead times, these commodities are widely available globally, reducing the risk of bottlenecks. The robustness of the reaction conditions also means that the process is less susceptible to minor variations in raw material quality, further stabilizing the supply chain. This reliability is essential for maintaining continuous production schedules and meeting the rigorous delivery commitments required by major pharmaceutical clients.
- Scalability and Environmental Compliance: The environmental footprint of this process is markedly reduced compared to traditional methods, aligning with increasingly strict global regulations on chemical manufacturing. The avoidance of toxic reagents simplifies waste treatment protocols and reduces the burden on effluent treatment plants. Furthermore, the straightforward crystallization and filtration steps are inherently scalable, allowing for seamless transition from pilot plant batches to multi-ton commercial production without the need for significant re-engineering of equipment. This scalability ensures that supply can be rapidly ramped up to meet surges in demand without compromising on quality or compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Aztreonam synthesis route. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and feasibility of the technology for potential manufacturing partners and technical evaluators.
Q: What are the primary advantages of this Aztreonam synthesis method over conventional routes?
A: This method eliminates the need for toxic and expensive reagents like dicyclohexylcarbodiimide (DCC) and trifluoroacetic acid (TFA). Instead, it utilizes conventional solvents like 1,4-dioxane and a mixed aqueous solution of acetic acid and hydrochloric acid for deprotection, significantly simplifying the workflow and reducing environmental impact.
Q: How is high purity achieved in the final Aztreonam product?
A: High purity is achieved through a specific purification step involving dissolution in dehydrated alcohol, charcoal absorption to remove impurities, and controlled crystallization. The patent reports achieving purity levels exceeding 99% in experimental embodiments through this rigorous refinement process.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the route is highly suitable for scale-up. It avoids complex distillation setups for solvent recovery in certain steps and uses readily available raw materials. The simplified workup procedure, involving straightforward filtration and washing, facilitates efficient commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aztreonam Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain competitiveness in the global API market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent can be fully realized in a practical manufacturing setting. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of Aztreonam meets the highest international standards. Our commitment to technical excellence allows us to navigate the complexities of beta-lactam chemistry with precision, delivering consistent quality that supports your regulatory filings and commercial success.
We invite you to engage with our technical procurement team to discuss how this optimized synthetic route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the specific economic advantages tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your partnership with us is built on a foundation of transparency, technical capability, and shared commercial growth.
