Advanced Acyl Chloride Route for Aztreonam Production: Enhancing Purity and Scalability for Global Pharma Supply Chains
Introduction to Next-Generation Aztreonam Manufacturing
The global demand for high-quality monocyclic beta-lactam antibiotics continues to drive innovation in synthetic methodology, particularly for critical agents like Aztreonam. Patent CN103044415A introduces a transformative synthesis strategy that fundamentally alters the production landscape by shifting from traditional active ester methodologies to a highly efficient acyl chloride approach. This technical breakthrough addresses long-standing challenges associated with beta-lactam stability and impurity control, offering a robust pathway for the manufacture of high-purity pharmaceutical intermediates. By leveraging a triphenylphosphine oxide (TPPO) and bis(trichloromethyl) carbonate (BTC) activation system, the process achieves superior atom economy while mitigating the risks of ring degradation that plague conventional routes. For R&D directors and supply chain leaders, this patent represents a significant opportunity to optimize manufacturing protocols, ensuring consistent quality and enhanced operational efficiency in the production of this vital antibiotic.
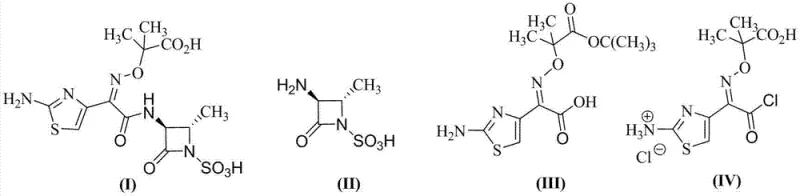
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Aztreonam has relied heavily on the MEAM method, which utilizes active esters such as mercaptobenzothiazole derivatives coupled with subsequent strong acid deprotection steps. This traditional paradigm suffers from inherent chemical vulnerabilities, primarily the exposure of the sensitive beta-lactam ring to harsh acidic conditions required for removing tert-butyl protecting groups. Processes involving heating to 60°C in the presence of excessive hydrochloric acid or trifluoroacetic acid frequently lead to the destruction of the beta-lactam nucleus, resulting in compromised yields and complex impurity profiles. Furthermore, the reliance on mercaptobenzothiazole esters introduces persistent contamination issues, as the removal of 2-mercaptobenzothiazole residues is notoriously difficult and often requires extensive downstream purification. The use of toxic and non-recyclable reagents like trifluoroacetic acid also imposes significant environmental burdens and waste disposal costs, creating friction for manufacturers aiming to meet modern green chemistry standards and regulatory compliance requirements.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN103044415A circumvents these pitfalls by employing an acyl chloride intermediate generated under mild conditions. By converting the carboxylic acid precursor directly into an acyl chloride using a TPPO-BTC system, the process eliminates the need for a separate, harsh acidic deprotection stage post-coupling. This strategic modification ensures that the beta-lactam ring is never subjected to the destructive forces of strong acids at elevated temperatures, thereby preserving the structural integrity of the molecule from the source. The direct condensation of the acyl chloride with the aztreonam parent nucleus not only streamlines the synthetic sequence but also inherently prevents the formation of mercaptobenzothiazole-related impurities. This results in a final product with purity levels consistently exceeding 99%, demonstrating a clear technological superiority that aligns with the rigorous quality expectations of international regulatory bodies and end-users.
Mechanistic Insights into TPPO-BTC Activated Acyl Chloride Formation
The core of this innovative synthesis lies in the in situ generation of dichlorotriphenylphosphine (Ph3PCl2), a potent chlorinating agent formed by the reaction of triphenylphosphine oxide with bis(trichloromethyl) carbonate. This activation system operates through a highly efficient mechanism where the oxygen of the carboxylic acid group in the starting material attacks the phosphorus center, facilitating the displacement of chloride ions and the formation of the reactive acyl chloride species. Unlike traditional coupling reagents that may leave behind difficult-to-remove urea byproducts, this system produces triphenylphosphine oxide as a benign byproduct, which, crucially, can be easily separated and recycled. The reaction kinetics are carefully managed by maintaining temperatures between -30°C and 30°C, with a preferred range of -15°C to -5°C, ensuring that the highly reactive acyl chloride intermediate remains stable prior to coupling. This precise thermal control is essential for minimizing side reactions such as racemization or hydrolysis, which could otherwise degrade the optical purity of the final antibiotic.
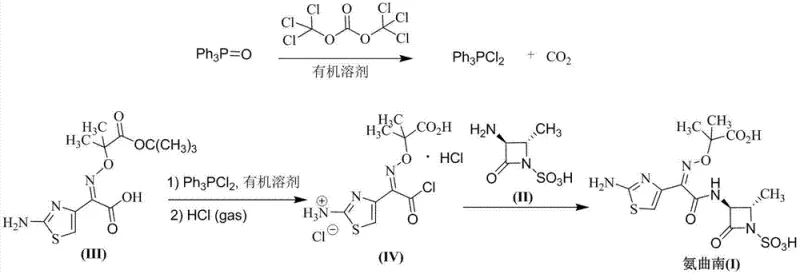
Furthermore, the impurity control mechanism is intrinsically linked to the choice of reagents and the avoidance of sulfur-containing active esters. In conventional active ester methods, the nucleophilic attack by the amine on the ester carbonyl can sometimes lead to incomplete conversion or transesterification side reactions involving the leaving group. By utilizing the acyl chloride pathway, the electrophilicity of the carbonyl carbon is maximized, driving the condensation reaction with the aztreonam parent nucleus to completion with high fidelity. The subsequent workup involves simple pH adjustment and crystallization, which effectively removes residual salts and unreacted starting materials without the need for complex chromatographic separations. This mechanistic elegance translates directly into a cleaner crude product, reducing the load on purification units and ensuring that the final API meets stringent specifications for residual solvents and related substances.
How to Synthesize Aztreonam Efficiently
The implementation of this synthesis route requires precise adherence to the stoichiometric ratios and thermal profiles outlined in the patent to maximize yield and safety. The process begins with the preparation of the chlorinating agent, followed by the low-temperature activation of the side chain acid, and concludes with the coupling step in a biphasic or mixed solvent system. Operators must ensure that the HCl gas saturation is controlled to prevent excess acidity that could prematurely hydrolyze the intermediate. The detailed standardized synthesis steps, including specific solvent volumes, addition rates, and isolation procedures, are critical for reproducibility on a commercial scale. For a comprehensive breakdown of the operational parameters and safety considerations required for laboratory or pilot plant execution, please refer to the technical guide below.
- Prepare a dichlorotriphenylphosphine solution by reacting triphenylphosphine oxide (TPPO) with bis(trichloromethyl) carbonate (BTC) in an organic solvent such as methylene dichloride at room temperature.
- React (2-amino thiazolyl-4-yl)-2-(tert-butyl oxycarbonyl)-isopropoxy iminodiacetic acid (Compound III) with the prepared dichlorotriphenylphosphine solution at low temperatures (-30 to 30°C), followed by saturation with HCl gas to generate the acyl chloride intermediate (Compound IV).
- Condense the resulting acyl chloride intermediate (Compound IV) with the aztreonam parent nucleus (Compound II) in a mixed solvent system containing an alkaline agent to directly yield high-purity Aztreonam.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this acyl chloride-based synthesis offers profound economic and logistical benefits that extend beyond simple yield improvements. The elimination of expensive and hazardous reagents like trifluoroacetic acid significantly reduces the raw material cost base, while the ability to recycle triphenylphosphine oxide creates a closed-loop system that minimizes waste generation and purchasing frequency. For supply chain heads, the robustness of this method means fewer batch failures due to beta-lactam degradation, leading to more predictable production schedules and reliable inventory levels. The simplified downstream processing also shortens the overall cycle time per batch, allowing manufacturing facilities to increase throughput without requiring additional capital investment in new equipment. These factors combine to create a more resilient supply chain capable of meeting fluctuating market demands for this critical antibiotic.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the recyclability of triphenylphosphine oxide, which serves as a catalyst carrier rather than a consumable reagent. By recovering and reusing TPPO from the mother liquor, manufacturers can drastically lower the variable costs associated with reagent consumption. Additionally, the avoidance of strong acid deprotection steps eliminates the need for specialized corrosion-resistant equipment and reduces the costs associated with neutralizing and disposing of large volumes of acidic waste. The higher atom economy of the acyl chloride route ensures that a greater proportion of raw materials are converted into the final product, further enhancing the overall cost efficiency of the manufacturing operation.
- Enhanced Supply Chain Reliability: The chemical stability of the intermediates and the mild reaction conditions contribute to a highly reliable production process with minimal risk of unexpected batch deviations. Because the method avoids the use of unstable active esters that may degrade during storage or transport, the supply chain becomes less vulnerable to raw material quality fluctuations. The consistent achievement of high purity levels reduces the likelihood of batch rejection during quality control testing, ensuring a steady flow of compliant material to downstream formulation partners. This reliability is crucial for maintaining uninterrupted supply to pharmaceutical customers who depend on just-in-time delivery models for their own production schedules.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing common organic solvents such as methylene dichloride and acetonitrile that are readily available in bulk quantities globally. The reduction in hazardous waste generation, particularly the absence of mercaptobenzothiazole residues and spent strong acids, simplifies environmental compliance and lowers the burden on wastewater treatment facilities. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturer. The ability to scale from kilogram to multi-ton production without significant process re-engineering makes this technology an attractive option for expanding capacity to meet growing global health needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Aztreonam synthesis route. These answers are derived directly from the experimental data and beneficial effects described in patent CN103044415A, providing clarity on how this method compares to existing industry standards. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for procurement professionals assessing the long-term value proposition of this manufacturing approach.
Q: How does the acyl chloride method protect the beta-lactam ring compared to traditional methods?
A: Traditional methods often require strong acid hydrolysis (e.g., trifluoroacetic acid) at elevated temperatures to remove protecting groups, which can degrade the sensitive beta-lactam nucleus. The patented acyl chloride route performs deprotection and activation simultaneously under milder conditions, avoiding strong acid exposure and preserving the structural integrity of the antibiotic core.
Q: What are the advantages regarding impurity profiles in this synthesis route?
A: This method eliminates the use of mercaptobenzothiazole active esters, thereby removing the risk of 2-mercaptobenzothiazole (M) residue in the final product. Additionally, the avoidance of harsh acidic deprotection steps reduces the formation of degradation byproducts, resulting in a cleaner crude profile and easier purification.
Q: Is the triphenylphosphine oxide (TPPO) reagent recyclable in this process?
A: Yes, a key economic and environmental advantage of this process is the recyclability of triphenylphosphine oxide. The TPPO generated as a byproduct during the activation step can be recovered from the mother liquor via crystallization and reused for the preparation of the dichlorotriphenylphosphine reagent, significantly reducing raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aztreonam Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN103044415A to maintain competitiveness in the global pharmaceutical market. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this acyl chloride method are fully realized in practical manufacturing environments. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Aztreonam intermediate meets the highest international standards for safety and efficacy. We are committed to leveraging our technical expertise to deliver cost-effective and high-quality solutions that support your drug development and commercialization goals.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic impact of switching to this greener, more efficient process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your specific volume requirements, ensuring a seamless transition to a more reliable and sustainable source of high-purity Aztreonam intermediates.
