Advanced Synthesis of Rasagiline Mesylate Intermediates for Commercial Scale-Up
Advanced Synthesis of Rasagiline Mesylate Intermediates for Commercial Scale-Up
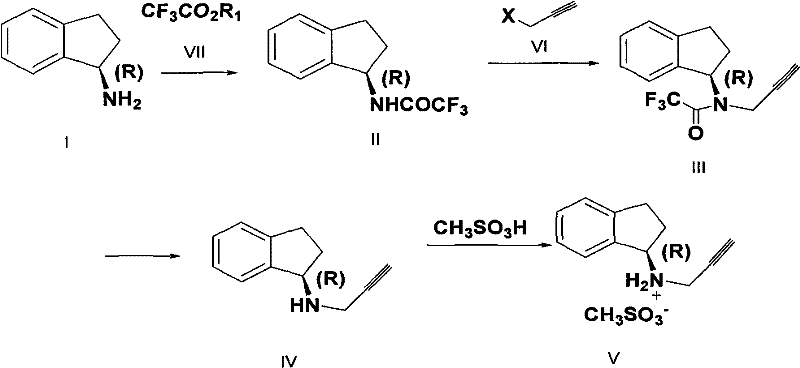
The pharmaceutical industry continuously seeks robust synthetic routes for Parkinson's disease treatments, specifically focusing on the efficient production of Rasagiline and its mesylate salt. Patent CN102464589A introduces a transformative preparation method that addresses critical bottlenecks in the existing supply chain for this vital active pharmaceutical ingredient (API). By leveraging a chiral pool strategy starting from (R)-(+)-1-indanamine, this technology bypasses the inefficient resolution steps typical of legacy processes. The innovation lies in a novel protection-alkylation-deprotection sequence that ensures high stereochemical integrity while minimizing waste generation. For procurement leaders and R&D directors, this represents a significant opportunity to optimize the manufacturing of high-purity pharmaceutical intermediates. The following analysis dissects the technical merits and commercial viability of this patented approach, highlighting its potential to redefine cost structures and supply reliability in the neurology therapeutic sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional manufacturing pathways for Rasagiline have long been plagued by inherent inefficiencies that drive up costs and complicate waste management. A predominant historical method involves the synthesis of a racemic mixture followed by optical resolution using tartaric acid or similar chiral acids. This approach is fundamentally flawed from an atom economy perspective because it inherently discards approximately half of the synthesized material—the unwanted (S)-enantiomer—as waste, drastically reducing overall yield. Furthermore, alternative routes often rely on harsh reduction conditions utilizing sodium borohydride or catalytic hydrogenation with Raney Nickel, which generate substantial quantities of boron-containing byproducts or require complex metal removal steps. Another significant challenge in prior art is the tendency for over-alkylation during the introduction of the propargyl group, leading to the formation of N,N-dipropargyl impurities that possess physical properties nearly identical to the target molecule, making purification extremely difficult and costly. Additionally, some existing methods employ 2-nitrobenzenesulfonyl protecting groups which require malodorous and toxic thiol reagents for removal, posing severe environmental and occupational health hazards that are increasingly unacceptable in modern green chemistry standards.
The Novel Approach
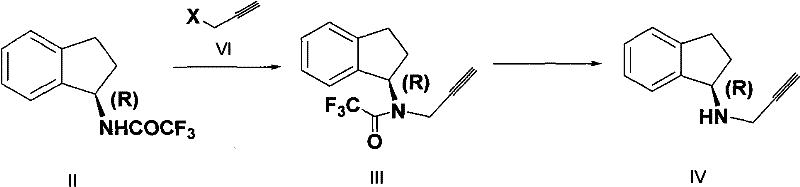
The methodology disclosed in CN102464589A offers a sophisticated solution by utilizing a chiral starting material, (R)-(+)-1-indanamine, thereby eliminating the need for resolution and maximizing theoretical yield. The core innovation involves converting the chiral amine into a trifluoroacetyl protected intermediate (Compound II) via a simple aminolysis reaction with trifluoroacetate esters. This protecting group serves a dual purpose: it maintains the stereochemical configuration and, crucially, deactivates the nitrogen sufficiently to prevent the formation of di-alkylated byproducts during the subsequent propargylation step. The process proceeds through a clean N-alkylation with halo propyne to form Compound III, followed by a mild hydrolysis to reveal the free amine (Compound IV). This sequence avoids the use of heavy metal catalysts, toxic thiols, and explosive reducing agents. The final step involves a straightforward salt formation with methanesulfonic acid to yield the stable mesylate salt. This streamlined workflow not only simplifies the operational complexity but also significantly enhances the purity profile of the final API intermediate, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks seeking to upgrade their technological capabilities.
Mechanistic Insights into Trifluoroacetyl-Mediated Selective Alkylation
The success of this synthetic route hinges on the precise electronic modulation of the amine nitrogen through trifluoroacetyl protection. In the first stage, the nucleophilic attack of (R)-(+)-1-indanamine on the carbonyl carbon of the trifluoroacetate ester results in the formation of an amide bond. The strong electron-withdrawing nature of the trifluoromethyl group significantly reduces the electron density on the nitrogen atom. This deactivation is mechanistically critical because it lowers the nucleophilicity of the nitrogen just enough to allow for mono-alkylation with the halo propyne electrophile while effectively suppressing the second alkylation event that leads to the troublesome N,N-dipropargyl impurity. Unlike unprotected amines which are highly prone to poly-alkylation, the protected intermediate reacts selectively under basic conditions using mineral alkalis such as sodium hydroxide or potassium carbonate in polar aprotic solvents like DMF or DMSO. Following the alkylation, the trifluoroacetyl group is readily cleaved under mild hydrolytic conditions using aqueous alkali in alcohol solvents. This deprotection step is highly efficient due to the lability of the trifluoroamide bond, regenerating the free amine without racemization. The preservation of optical purity throughout this cycle is paramount, with the patent data indicating optical purity levels exceeding 99% ee, demonstrating that the reaction conditions are sufficiently gentle to prevent epimerization at the chiral center.
Impurity control is further enhanced by the choice of reagents and workup procedures described in the patent. The use of specific mineral bases and controlled temperatures (ranging from 20°C to 100°C depending on the specific base and solvent system) ensures that side reactions such as elimination of the halo propyne or degradation of the indane ring are minimized. The hydrolysis step is particularly robust, tolerating a wide range of alcoholic solvents including methanol, ethanol, and trifluoroethanol, which facilitates easy processing and solvent recovery. The final isolation of the product is achieved through standard extraction and concentration techniques, avoiding the need for complex chromatographic separations that are often required to remove structural analogs in less selective synthetic routes. This mechanistic elegance translates directly into a cleaner crude product profile, reducing the burden on downstream purification units and ensuring that the final Rasagiline mesylate meets stringent regulatory specifications for genotoxic impurities and residual solvents.
How to Synthesize (R)-(+)-N-propargyl-1-indanamine Efficiently
The practical implementation of this synthesis requires careful attention to stoichiometry and reaction monitoring to maximize throughput. The process begins with the protection of the chiral amine, followed by the critical alkylation step where the propargyl group is introduced, and concludes with hydrolysis and salt formation. Detailed operational parameters regarding temperature control, molar ratios of the halo propyne, and specific base selection are critical for reproducing the high yields reported in the patent examples. Operators must ensure complete consumption of the starting amine before proceeding to workup to minimize residual starting material in the final product.
- Protect (R)-(+)-1-indanamine via aminolysis with trifluoroacetate to form the trifluoroacetyl intermediate.
- Perform selective N-alkylation using halo propyne under basic conditions to introduce the propargyl group without over-alkylation.
- Hydrolyze the trifluoroacetyl protecting group under mild alkaline conditions to yield the free amine, followed by salt formation with methanesulfonic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology offers compelling strategic advantages that extend beyond mere technical feasibility. The elimination of the optical resolution step fundamentally alters the cost basis of production by theoretically doubling the yield from the chiral starting material compared to racemic routes. This efficiency gain means that less raw material is required to produce the same amount of finished API, leading to substantial cost savings in raw material procurement. Furthermore, the avoidance of expensive and hazardous reagents such as sodium borohydride, Raney Nickel, and malodorous thiols reduces both the direct cost of goods sold and the indirect costs associated with safety compliance, waste disposal, and environmental remediation. The simplified workup procedures, which rely on standard extractions and crystallizations rather than complex chromatography or specialized filtration for metal removal, significantly reduce processing time and equipment occupancy, thereby increasing overall plant throughput and capacity utilization.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the superior atom economy and the elimination of yield-killing resolution steps. By utilizing a chiral pool starting material and a highly selective alkylation protocol, the process minimizes the formation of hard-to-remove impurities that typically require costly recrystallization or chromatographic purification. The removal of heavy metal catalysts from the process map eliminates the need for expensive scavenging resins and the associated validation testing for residual metals, which is a significant cost driver in API manufacturing. Additionally, the reagents used, such as trifluoroacetate esters and halo propynes, are commodity chemicals available from multiple global suppliers, ensuring competitive pricing and reducing the risk of single-source dependency. The overall simplification of the synthetic route reduces the number of unit operations, which directly correlates to lower labor costs, reduced energy consumption, and decreased solvent usage per kilogram of product produced.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this chemistry ensures consistent delivery schedules and reduces the risk of batch failures. The tolerance of the reaction to a variety of solvents and bases provides flexibility in sourcing; if a specific solvent like DMF faces supply constraints, alternatives like DMSO or NMP can be utilized without compromising the reaction outcome. The high purity of the intermediate generated at each step reduces the likelihood of downstream processing delays caused by off-spec material. Moreover, the stability of the intermediates allows for potential campaign manufacturing strategies where key intermediates can be stockpiled, providing a buffer against demand fluctuations. This resilience is crucial for maintaining continuity of supply for critical Parkinson's medications, where interruptions can have serious consequences for patients and liability implications for manufacturers.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing reaction conditions that are easily transferable from laboratory to pilot and commercial scale. The absence of exothermic hazards associated with hydride reductions and the avoidance of pyrophoric catalysts make the process safer to operate at large scales, reducing insurance premiums and safety infrastructure costs. Environmentally, the process aligns with green chemistry principles by reducing waste generation through higher yields and avoiding the discharge of heavy metals and sulfur-containing compounds. The solvents used are largely recoverable and recyclable, further minimizing the environmental footprint. This compliance with stringent environmental regulations facilitates faster regulatory approvals in key markets and enhances the corporate sustainability profile of the manufacturing entity, which is increasingly important for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims within the patent documentation to provide clarity on process capabilities. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer.
Q: How does this method improve optical purity compared to racemic resolution?
A: By starting directly with chiral (R)-(+)-1-indanamine, this process eliminates the need for optical resolution of a racemic mixture, thereby avoiding the inherent 50% yield loss associated with discarding the unwanted (S)-enantiomer and ensuring high optical purity from the outset.
Q: What are the primary impurities controlled in this alkylation strategy?
A: The use of the trifluoroacetyl protecting group effectively suppresses the formation of the di-propargylated byproduct, which is a common and difficult-to-remove impurity in direct alkylation methods, thus simplifying purification and enhancing final product quality.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method utilizes commercially available reagents, avoids hazardous reducing agents like sodium borohydride or malodorous thiols, and employs standard workup procedures like extraction and crystallization, making it highly scalable and environmentally compliant for ton-level manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rasagiline Intermediate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of implementing advanced synthetic methodologies like the one described in CN102464589A to deliver superior value to our global partners. As a dedicated CDMO and manufacturer, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial reality is seamless and efficient. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify optical purity and impurity profiles at every stage of production. We understand that in the competitive landscape of neurological therapeutics, consistency and reliability are non-negotiable, and our infrastructure is built to guarantee supply continuity for high-purity pharmaceutical intermediates.
We invite you to collaborate with us to leverage this cutting-edge technology for your Rasagiline supply chain. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete performance metrics. By partnering with NINGBO INNO PHARMCHEM, you secure not just a supplier, but a strategic ally committed to driving down costs while elevating the quality standards of your pharmaceutical portfolio.
