Advanced Catalytic Hydrogenation for High-Purity Pregnenolone Acetate Manufacturing
The global demand for high-quality hormonal intermediates continues to surge, driven by the expanding pharmaceutical market for reproductive health and anti-inflammatory therapies. At the heart of Progesterone synthesis lies Pregnenolone Acetate, a critical intermediate whose production has historically been plagued by complex equipment requirements and inconsistent yields. A significant technological breakthrough in this domain is documented in patent CN103450306A, which outlines a refined synthetic method that addresses these longstanding industrial bottlenecks. This patent introduces a mild, highly selective catalytic hydrogenation process that utilizes a unique composite catalyst system to achieve exceptional purity levels. For R&D directors and supply chain managers, understanding this methodology is crucial for optimizing the production of high-purity pharmaceutical intermediates. The innovation lies not just in the chemical transformation but in the holistic improvement of safety, cost-efficiency, and environmental compliance, marking a pivotal shift from traditional, hazardous protocols to a modern, sustainable manufacturing paradigm.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
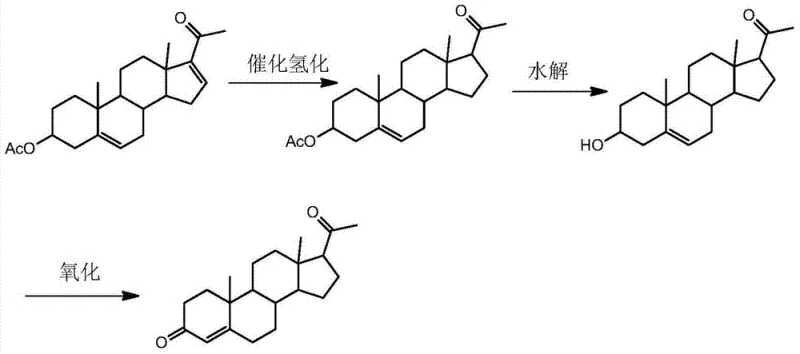
Historically, the industrial synthesis of Pregnenolone Acetate from Dehydropregnenolone Acetate has relied heavily on catalytic hydrogenation using ethanol as the primary solvent and Raney Nickel or standard Palladium on Carbon as catalysts. These conventional pathways suffer from severe drawbacks that impact both operational safety and economic viability. The use of Raney Nickel, for instance, often necessitates large catalyst loads and results in relatively low yields, creating significant waste disposal challenges due to the pyrophoric nature of the spent catalyst. Furthermore, when standard Palladium catalysts are employed in ethanol, the reaction conditions often become severe, leading to the undesirable hydrogenation of the Δ5-ethylene linkage. This side reaction generates structural impurities that are chemically similar to the target product, making purification arduous and drastically reducing the overall yield of the final active pharmaceutical ingredient. Additionally, the traditional reliance on ethanol requires massive solvent volumes—often upwards of 30 times the volume of the raw material—which inflates energy costs for solvent recovery and limits the throughput of existing reactor infrastructure.
The Novel Approach
In stark contrast, the novel approach detailed in the referenced patent revolutionizes this step by introducing a composite catalyst system comprising Palladium on Carbon and ammonium acetate, dissolved in ethyl acetate. This strategic modification fundamentally alters the reaction landscape, allowing for hydrogenation to proceed under remarkably mild conditions, specifically between 35°C and 45°C and at moderate hydrogen pressures of 0.4 MPa to 0.8 MPa. The introduction of ammonium acetate acts as a crucial modifier, forming a transient complex with the palladium that imposes steric hindrance, effectively shielding the Δ5-double bond from reduction while selectively targeting the Δ16-position. This results in a reaction profile with near-perfect selectivity, virtually eliminating the formation of over-hydrogenated byproducts. Moreover, the switch to ethyl acetate as the solvent reduces the required solvent load to merely 8 to 10 times the volume of the raw material, significantly streamlining the concentration and crystallization steps. The outcome is a robust, scalable process that delivers a white, sandy solid product with outstanding crystal habits, facilitating easy centrifugation and drying.
Mechanistic Insights into Pd/C-Ammonium Acetate Catalytic Hydrogenation
The core of this technological advancement lies in the synergistic interaction between the heterogeneous Palladium on Carbon catalyst and the homogeneous ammonium acetate additive. In a standard hydrogenation environment, the metal surface is highly active and non-discriminatory, often reducing multiple olefinic bonds indiscriminately. However, the presence of ammonium acetate introduces a sophisticated level of control through coordination chemistry. It is hypothesized that the ammonium ions interact with the reactive metal sites or the substrate itself to form a title complex that modulates the electronic density and steric accessibility of the catalyst surface. This modulation ensures that the hydrogenation is kinetically favored only at the specific Δ16-ethylene linkage, while the thermodynamically sensitive Δ5-bond remains intact. This mechanism is critical for maintaining the structural integrity required for subsequent biological activity in the final hormone product. By preventing the formation of saturated ring byproducts, the process avoids the generation of impurities that would otherwise require expensive and yield-loss-inducing chromatographic purification steps later in the synthesis train.
Furthermore, the ammonium acetate serves a dual function as a buffering agent, maintaining a stable acid-base environment throughout the reaction duration. In many hydrogenation processes, localized pH shifts can catalyze degradation pathways or promote isomerization, leading to a complex impurity profile. The buffer capacity of the ammonium salt mitigates these risks, ensuring that the reaction milieu remains neutral to slightly acidic, which is optimal for the stability of the steroid backbone. This stability is reflected in the exceptional purity metrics, where the mass purity of the product consistently exceeds 99.5%. For quality control teams, this means a much simpler impurity spectrum to monitor and validate, reducing the analytical burden and accelerating the release of batches for the next synthesis stage. The combination of high selectivity and environmental stability makes this catalytic system superior to single-component catalysts that lack such regulatory finesse.
How to Synthesize Pregnenolone Acetate Efficiently
Implementing this synthesis route requires precise adherence to the optimized parameters regarding temperature, pressure, and reagent ratios to maximize the benefits of the composite catalyst system. The process begins with the preparation of the reaction mixture in a hydrogenation vessel, followed by a controlled workup that leverages the solubility properties of the product in ethyl acetate and methanol mixtures. The following guide outlines the critical operational phases derived from the patent examples, ensuring reproducibility and high yield. For detailed standard operating procedures and safety data sheets, please refer to the technical documentation provided below.
- Perform catalytic hydrogenation of dehydropregnenolone acetate at 35-45°C and 0.4-0.8 MPa using ethyl acetate solvent and a Pd/C-ammonium acetate catalyst.
- Filter the reaction mixture, concentrate the filtrate under normal pressure, and induce crystallization by adding a methanol-ethyl acetate mixed solvent.
- Wash the crude product with warm water and methanol, then dry to obtain white sandy solid Pregnenolone Acetate with >99.5% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic methodology translates directly into tangible operational efficiencies and risk mitigation. The shift away from hazardous catalysts like Raney Nickel and the reduction in solvent volumes address two of the most significant cost drivers in fine chemical manufacturing: waste disposal and energy consumption. By utilizing a catalyst system that is easier to handle and filter, the process eliminates the need for specialized safety protocols associated with pyrophoric materials, thereby reducing insurance and compliance overheads. The simplified post-treatment workflow, which involves straightforward filtration and crystallization rather than complex extractions or distillations, shortens the batch cycle time significantly. This acceleration in production velocity enhances the overall capacity of the manufacturing plant without requiring capital expenditure on new reactors, allowing for greater responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic reduction in solvent usage and the elimination of expensive purification steps. By lowering the solvent-to-substrate ratio from the industry standard of 30-fold to less than 10-fold, the energy required for solvent recovery via distillation is substantially decreased. Furthermore, the high selectivity of the reaction means that raw material conversion is maximized with minimal loss to byproducts, effectively increasing the theoretical yield per kilogram of starting material. The removal of transition metal contaminants is also streamlined, as the Pd/C catalyst is easily filtered off, negating the need for costly scavenger resins or extensive washing protocols often required to meet heavy metal specifications in pharmaceutical grades.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of readily available and stable reagents such as ethyl acetate and ammonium acetate, which are commodity chemicals with robust global supply chains. Unlike specialized catalysts that may have long lead times or single-source dependencies, the components of this composite system are widely accessible, reducing the risk of production stoppages due to raw material shortages. The mild reaction conditions also place less stress on manufacturing equipment, extending the lifespan of reactors and hydrogenation vessels and reducing the frequency of maintenance shutdowns. This reliability ensures a steady flow of high-purity intermediates to downstream customers, securing the production schedules for finished hormonal products.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns perfectly with green chemistry principles by minimizing waste generation and utilizing safer solvents. The reduced solvent load translates to a smaller carbon footprint associated with solvent incineration or recycling. Additionally, the absence of toxic byproducts and the use of a recoverable heterogeneous catalyst simplify the treatment of effluent streams, ensuring compliance with increasingly stringent environmental regulations. The process is inherently scalable, as demonstrated by the successful translation from laboratory to pilot-scale examples in the patent, making it suitable for commercial scale-up of complex pharmaceutical intermediates without the need for process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced hydrogenation technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this route into your existing manufacturing portfolio.
Q: How does the new catalyst system improve selectivity compared to traditional Raney Nickel?
A: The novel composite catalyst system utilizing Palladium on Carbon (Pd/C) combined with ammonium acetate creates a specific steric environment. This prevents the unwanted hydrogenation of the Δ5-double bond, a common side reaction in traditional methods, thereby ensuring high selectivity for the Δ16-position and eliminating difficult-to-remove impurities.
Q: What are the solvent advantages of using ethyl acetate over ethanol in this process?
A: Ethyl acetate offers superior recovery rates and requires significantly lower volumes (8-10 times the raw material volume) compared to the large excesses of ethanol needed in conventional processes. This reduction in solvent load drastically lowers energy consumption during concentration and simplifies the downstream purification workflow.
Q: What purity levels can be achieved with this synthetic method?
A: By maintaining a stable acid-base environment through the buffer effect of ammonium acetate and avoiding harsh reaction conditions, this method consistently yields Pregnenolone Acetate with a mass purity exceeding 99.5%, meeting the stringent requirements for subsequent hormone synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pregnenolone Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN103450306A are fully realized in a commercial setting. Our facilities are equipped with state-of-the-art hydrogenation reactors capable of maintaining the precise temperature and pressure controls required for this selective process. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Pregnenolone Acetate meets the highest international standards for pharmaceutical intermediates, providing you with a secure and high-quality supply chain foundation.
We invite you to collaborate with us to leverage this technology for your specific product needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this optimized route can improve your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you streamline your hormone synthesis supply chain with confidence and precision.
