Advanced Manufacturing of Obeticholic Acid: A Novel Silane-Protection Strategy for High-Purity API Intermediates
Introduction to Patent CN112898369A
The pharmaceutical landscape for treating Primary Biliary Cirrhosis (PBC) and Non-Alcoholic Steatohepatitis (NASH) has been significantly shaped by the approval of Obeticholic Acid (OCA), a potent farnesoid X receptor agonist. As global demand for this critical active pharmaceutical ingredient surges, the industry faces persistent challenges in manufacturing high-purity OCA efficiently. Patent CN112898369A introduces a groundbreaking synthetic methodology that fundamentally restructures the production pathway to address long-standing quality issues. By implementing a strategic 'quality by design' approach, this invention utilizes a brand-new intermediate and reaction sequence that drastically simplifies operations while enhancing the purity profile of the final drug substance. The core innovation lies in the early-stage protection of the reactive 3-alpha-hydroxyl group, a modification that serves as a shield against oxidative degradation and dimerization throughout the synthesis.
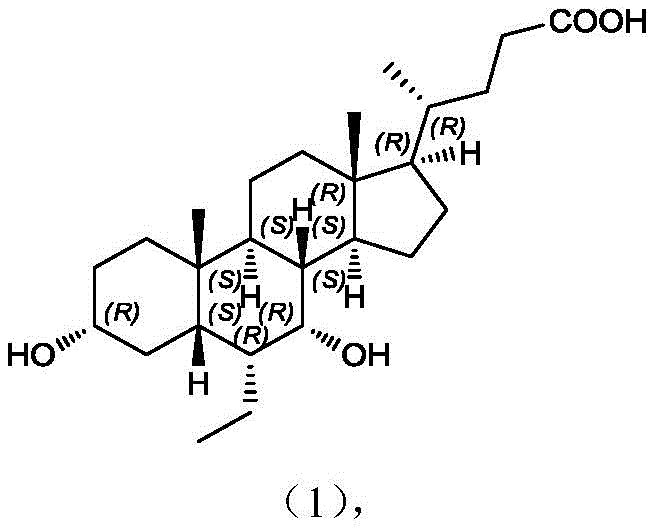
This technical advancement is particularly relevant for reliable API intermediate suppliers seeking to differentiate their offerings through superior impurity control. The patent details a robust route that not only meets stringent registration quality requirements but also ensures clinical medication safety by minimizing genotoxic or difficult-to-remove byproducts. For procurement and supply chain leaders, understanding this shift from conventional oxidation-prone routes to this protected intermediate strategy is essential for securing a stable, high-quality supply of high-purity Obeticholic Acid. The following analysis dissects the mechanistic advantages and commercial implications of this novel process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the majority of existing synthetic routes for Obeticholic Acid have relied on 3-alpha-hydroxy-6-ethylidene-7-ketone-5-beta-cholic acid (Formula II) as the primary starting material. While this compound is commercially available, its utilization introduces significant vulnerabilities in the manufacturing process. As disclosed in prior art such as WO02/072598, the conventional pathway involves a palladium-on-carbon catalytic reduction followed by conformational transformation. However, this approach suffers from critical defects: approximately 10% of the functional groups are oxidized to carbonyl groups, generating Impurity III, due to the sensitivity of the free 3-alpha-hydroxyl group under high-temperature hydrogenation conditions.
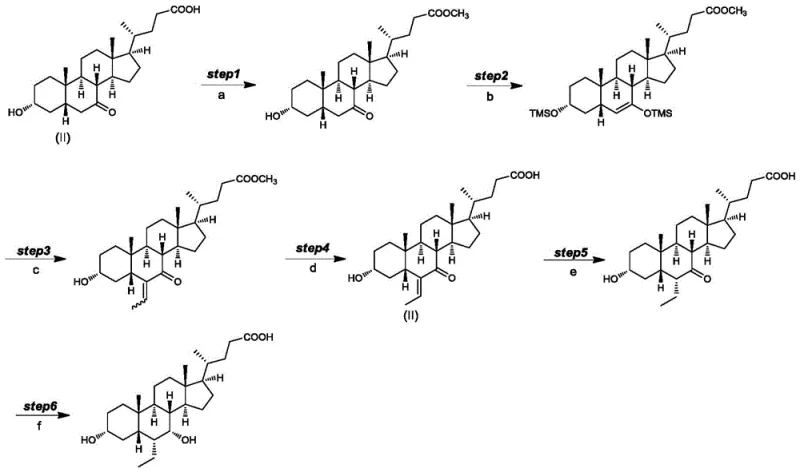
Furthermore, the conventional method is prone to forming intermediate dimeric impurities, known as Impurity IV, particularly during acid-catalyzed work-up procedures. These side reactions not only severely impact the overall yield of the reaction but also complicate downstream purification, often requiring extensive chromatography or recrystallization efforts that drive up costs. Additionally, the necessity for configuration conversion in a sodium hydroxide aqueous solution at high temperatures results in excessive energy consumption and creates a challenging environment for controlling raw material quality. These cumulative inefficiencies make the traditional route suboptimal for cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast, the novel approach detailed in Patent CN112898369A circumvents these pitfalls by introducing a silane protecting group at the very beginning of the synthesis. The process begins with the esterification of the starting material, followed immediately by the protection of the 3-alpha-hydroxyl group using a halosilane reagent such as tert-butyldimethylsilyl chloride (TBSCl). This strategic modification effectively 'masks' the vulnerable hydroxyl functionality, rendering it inert to the oxidative conditions encountered during subsequent hydrogenation steps. By preventing the formation of the 3-ketone species, the generation of Impurity III is virtually eliminated at the source.
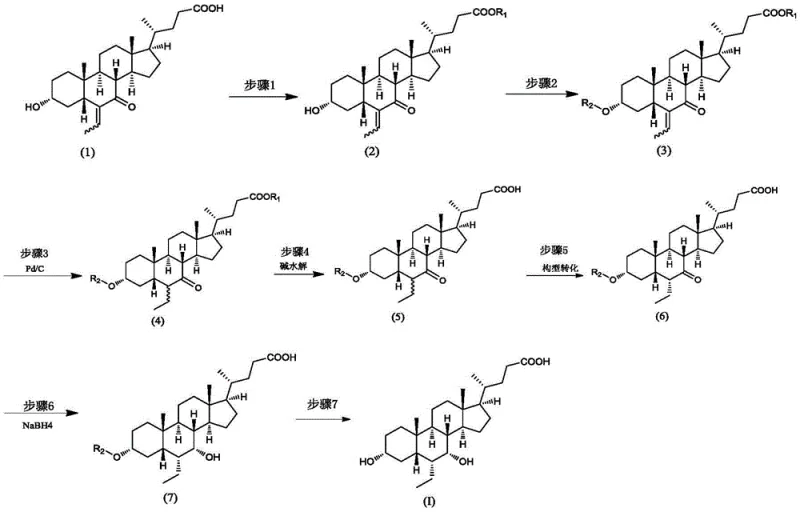
Moreover, the new route streamlines the configuration transformation and reduction steps, allowing them to proceed with higher selectivity and yield. The final deprotection step is mild and efficient, utilizing acid or tetrabutylammonium fluoride to reveal the target molecule without inducing the dimerization seen in older methods. This comprehensive redesign of the synthetic logic ensures that the commercial scale-up of complex steroid intermediates can be achieved with a significantly cleaner impurity profile. The result is a process that is not only chemically superior but also operationally simpler, aligning perfectly with modern green chemistry principles and regulatory expectations for drug substance manufacturing.
Mechanistic Insights into Silane-Protection and Stereocontrol
The cornerstone of this improved synthesis is the mechanistic role of the silyl ether protecting group. In organic synthesis, the 3-alpha-hydroxyl group on the cholanic acid skeleton is notoriously susceptible to oxidation, especially in the presence of transition metal catalysts like palladium. By converting this hydroxyl group into a silyl ether (e.g., TBS-ether), the electron density and steric environment around the oxygen atom are altered. This bulky protecting group physically blocks the approach of oxidizing agents and stabilizes the molecule against the harsh conditions of catalytic hydrogenation. Consequently, the reduction of the 6-ethylidene double bond proceeds cleanly to the 6-ethyl configuration without compromising the integrity of the A-ring functionality.
Another critical mechanistic feature is the base-catalyzed configuration transformation at the 7-position. In the protected intermediate state, the molecule undergoes epimerization under alkaline conditions (using NaOH or KOH) to establish the thermodynamically favorable 7-beta stereochemistry required for biological activity. The presence of the protecting group ensures that this equilibrium is not disturbed by competing side reactions at the 3-position. Following this, the reduction of the 7-ketone with sodium borohydride is highly stereoselective, delivering the desired 7-alpha-hydroxyl group. This precise control over stereochemistry is vital for meeting the rigorous specifications of high-purity Obeticholic Acid, ensuring that the final product possesses the exact spatial arrangement necessary for receptor binding.
How to Synthesize Obeticholic Acid Efficiently
The synthesis of Obeticholic Acid via this novel route involves a logical sequence of protection, reduction, transformation, and deprotection. The process begins with the esterification of the carboxylic acid tail, followed by the installation of the silane protecting group. Subsequent hydrogenation reduces the exocyclic double bond, and hydrolysis removes the ester. The core structural rearrangement occurs via base-mediated epimerization, followed by ketone reduction. Finally, acidic deprotection yields the target molecule. This streamlined workflow minimizes unit operations and maximizes throughput. For detailed laboratory protocols and specific reaction conditions, please refer to the standardized synthesis guide below.
- Esterification of the starting cholic acid derivative followed by protection of the 3-alpha-hydroxyl group using a halosilane reagent.
- Catalytic hydrogenation of the ethylidene double bond using palladium on carbon, followed by hydrolysis to remove the ester protecting group.
- Base-catalyzed configuration transformation at the 7-position, reduction of the 7-ketone, and final deprotection to yield pure Obeticholic Acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the purification train. By eliminating the formation of hard-to-remove impurities like the dimeric Impurity IV and the oxidized Impurity III, manufacturers can bypass complex and yield-losing chromatographic separations. This directly translates to substantial cost savings in raw material consumption and solvent usage, as fewer processing steps are required to achieve pharmacopeial purity. The robustness of the silane protection strategy also means that batch-to-batch variability is significantly reduced, providing a more predictable supply stream for downstream formulation teams.
- Cost Reduction in Manufacturing: The elimination of expensive heavy metal scavengers and extensive recrystallization cycles leads to a leaner cost structure. Since the process avoids the high-energy consumption associated with the high-temperature configuration conversions of the prior art, utility costs are also optimized. Furthermore, the higher overall yield resulting from reduced side reactions means that less starting material is required per kilogram of finished API, driving down the cost of goods sold (COGS) significantly.
- Enhanced Supply Chain Reliability: The reagents utilized in this process, such as palladium on carbon, sodium borohydride, and common silane chlorides, are commodity chemicals with stable global supply chains. Unlike specialized catalysts that may face sourcing bottlenecks, these materials are readily available from multiple vendors. This diversity in sourcing options mitigates the risk of supply disruption, ensuring reducing lead time for high-purity pharmaceutical intermediates and maintaining continuity of supply even during market fluctuations.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reactor types and conditions that are easily transferable from pilot plant to multi-ton commercial production. The reduction in hazardous waste generation, owing to higher selectivity and fewer purification steps, aligns with increasingly strict environmental regulations. This 'green' profile facilitates faster regulatory approvals and reduces the environmental footprint of the manufacturing site, a key consideration for sustainable supply chain management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented process. These insights are derived directly from the experimental data and claims within Patent CN112898369A, providing a clear picture of the technology's capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their own production lines or for procurement teams negotiating supply agreements.
Q: How does the new process reduce Impurity III and IV?
A: The novel process introduces a silane protecting group at the 3-alpha position early in the synthesis. This prevents the oxidation of the hydroxyl group to a ketone (Impurity III) during the palladium-catalyzed hydrogenation step and inhibits the acid-catalyzed dimerization that leads to Impurity IV.
Q: What are the key reagents used in the configuration transformation step?
A: The configuration transformation is achieved under alkaline conditions, typically using sodium hydroxide in a methanol/water solvent system at elevated temperatures (60°C to 90°C) to ensure the correct stereochemistry at the 7-position.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes common industrial reagents such as palladium on carbon, sodium borohydride, and standard silane chlorides. The elimination of complex purification steps for difficult impurities makes it highly scalable and cost-effective for metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Obeticholic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust infrastructure. Our team has extensively analyzed the pathways described in Patent CN112898369A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art rigorous QC labs capable of detecting and quantifying trace impurities to meet stringent purity specifications required by global regulatory bodies. Our commitment to quality ensures that every batch of Obeticholic Acid we produce adheres to the highest standards of safety and efficacy.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to request specific COA data and route feasibility assessments to verify the superiority of our manufacturing process. Let us help you secure a reliable, high-quality supply of Obeticholic Acid that drives your pharmaceutical projects forward with confidence and efficiency.
