Advanced Synthesis of Obeticholic Acid Intermediates via Regioselective Epoxidation for Commercial Scale-Up
Advanced Synthesis of Obeticholic Acid Intermediates via Regioselective Epoxidation for Commercial Scale-Up
The pharmaceutical industry's demand for high-purity bile acid derivatives has surged following the approval of Obeticholic acid (OCA) as a potent Farnesol X Receptor (FXR) agonist for treating primary biliary cholangitis. However, the commercial viability of this life-saving medication has long been hindered by the complexities of its synthetic route. Patent CN112341516A introduces a transformative methodology for synthesizing critical 5,6-epoxy steroid intermediates, addressing the longstanding bottlenecks of regioselectivity and yield that have plagued previous generations of synthesis. This technical insight report analyzes the novel vanadyl-catalyzed epoxidation strategy, demonstrating how it offers a robust pathway for reliable API intermediate suppliers to secure the global supply chain. By shifting away from non-selective alkylation and problematic diene epoxidation, this technology enables the production of high-purity steroid intermediates with significantly reduced impurity profiles, directly impacting the cost of goods and manufacturing efficiency for downstream drug producers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the critical 6-alpha-ethyl group in Obeticholic acid has been a formidable challenge for process chemists. Early approaches, such as the route disclosed in WO2002072598, relied on the direct alkylation of 7-ketolithocholic acid derivatives using ethyl iodide under strong alkaline conditions. This brute-force chemical approach is fundamentally flawed due to its abysmal selectivity; the reaction generates a multitude of byproducts, resulting in a one-step ethylation yield of merely 12 percent. Such inefficiency renders the process economically unviable for large-scale manufacturing, as the sheer volume of waste and the difficulty in purifying the desired isomer create prohibitive costs. Furthermore, alternative strategies attempting to utilize 4,6-diene intermediates, as seen in various PCT applications by companies like NZP, encounter severe regioselectivity hurdles. In these poly-unsaturated systems, the presence of multiple double bonds at the 4,5 and 22,23 positions creates a chaotic reaction landscape where oxidation can occur at multiple sites and faces.
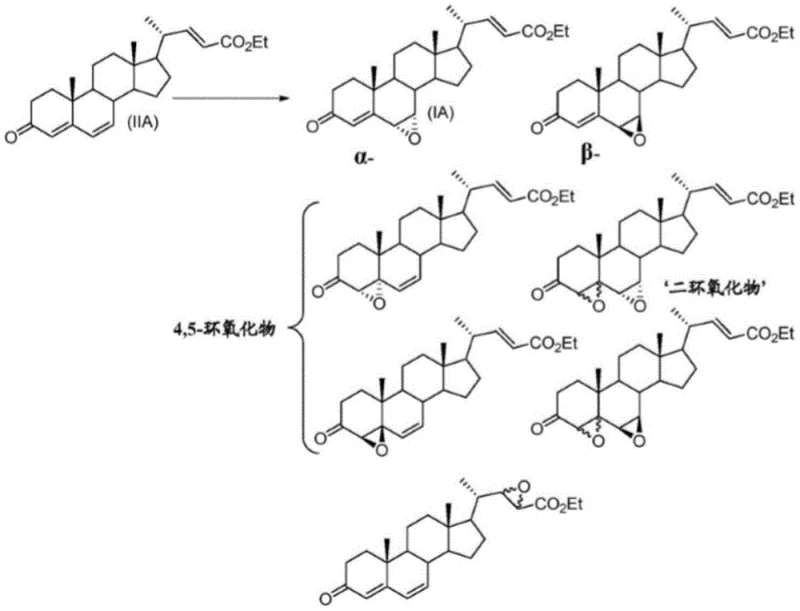
As illustrated in the analysis of prior art, the oxidation of 4,6-diene compounds often results in a complex mixture of mono-epoxides, di-epoxides, and various stereoisomers, collectively referred to as 'dioxide' impurities. Isolating the specific 6,7-alpha epoxy compound from this soup of structural analogs requires extensive and costly chromatographic purification, which drastically reduces the overall throughput of the manufacturing line. These conventional methods not only suffer from low yields but also generate significant hazardous waste, posing environmental compliance challenges that modern green chemistry initiatives seek to eliminate. The inability to control the stereochemistry at the 5 and 6 positions effectively means that a substantial portion of the starting material is lost to useless isomers, creating a supply chain bottleneck that limits the availability of the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast to these inefficient legacy methods, the technology disclosed in CN112341516A presents a paradigm shift by utilizing a highly selective vanadyl-catalyzed epoxidation on a specifically engineered precursor. Instead of battling the reactivity of multiple double bonds, this novel approach constructs the steroid skeleton with a single, targeted olefinic bond at the 5,6-position prior to the oxidation step. The process employs vanadyl acetylacetonate as a specialized catalyst in conjunction with tert-butyl hydroperoxide as the oxidant. This catalytic system is uniquely tuned to recognize the electronic and steric environment of the 5,6-double bond, facilitating the formation of the 5,6-epoxy intermediate with exceptional regioselectivity and stereoselectivity. By avoiding the formation of the problematic 4,6-diene system entirely, the process sidesteps the generation of the complex impurity profile associated with prior art. The result is a streamlined synthesis where the desired beta-configuration epoxy is formed predominantly, allowing for simpler purification protocols such as crystallization rather than exhaustive column chromatography. This methodological leap transforms the synthesis from a low-yielding struggle into a high-efficiency operation suitable for industrial mass production, ensuring a consistent supply of high-quality intermediates.
Mechanistic Insights into Vanadyl-Catalyzed Regioselective Epoxidation
The core innovation of this patent lies in the precise mechanistic control exerted by the vanadyl catalyst during the oxygen transfer event. Vanadyl acetylacetonate acts as a Lewis acid catalyst that coordinates with the hydroperoxide oxidant to form a reactive metal-peroxo species. This active species is sterically and electronically guided to attack the electron-rich 5,6-double bond of the steroid substrate (Compound D) while leaving other potential reactive sites untouched. The reaction is conducted in dichloromethane at a mild temperature range of 40-45°C, conditions that are sufficiently energetic to drive the reaction forward but controlled enough to prevent thermal degradation or non-selective background oxidation. The transition state favors the formation of the 5-beta, 6-beta-epoxide due to the specific conformational constraints imposed by the steroid nucleus and the directing influence of the adjacent 7-hydroxyl group (protected or free depending on the specific embodiment). This level of control is critical because it ensures that the subsequent ring-opening steps to introduce the ethyl group will proceed with the correct stereochemistry, ultimately yielding the biologically active 6-alpha-ethyl configuration.
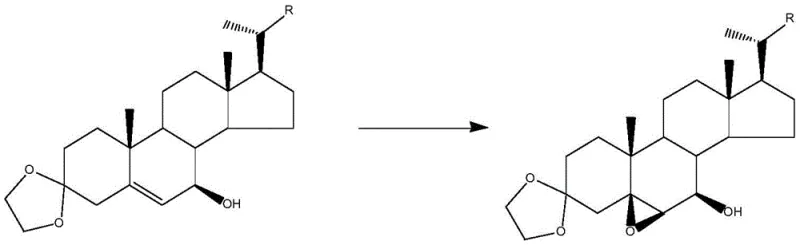
Furthermore, the impurity control mechanism is inherently built into the choice of substrate and catalyst. By preparing the precursor Compound D through a sequence of ketal protection, allylic oxidation, and reduction, the molecule is pre-organized to present only the 5,6-olefin as a viable target for epoxidation. The 3-position is protected as an ethylene ketal, preventing unwanted oxidation at the A-ring, while the side chain remains inert under these specific catalytic conditions. This orthogonal reactivity ensures that the oxidant is consumed almost exclusively by the desired transformation, leading to a reaction yield of approximately 60-70% for the epoxidation step itself, with the potential for optimization even higher. The reduction in side reactions means that the crude product contains significantly fewer structurally related impurities, simplifying the downstream processing. For R&D directors, this mechanistic clarity offers confidence in the robustness of the process, as the chemical logic dictates a clean transformation rather than a statistical distribution of products.
How to Synthesize 5,6-Epoxy Steroid Intermediate Efficiently
The synthesis of the target intermediate involves a logical sequence of functional group manipulations designed to set the stage for the critical epoxidation. The process begins with the protection of the 3-keto group of the starting steroid using ethylene glycol and trimethyl orthoacetate, followed by a highly selective allylic oxidation to install the 7-hydroxyl functionality. This is subsequently reduced to the alcohol to yield Compound D, the direct precursor for the epoxidation. The detailed operational parameters, including specific mass ratios of catalysts and solvents, are critical for reproducing the high selectivity reported in the patent. For a comprehensive guide on executing these steps with precision, please refer to the standardized protocol below.
- Protect the 3-keto group of the starting steroid using ethylene glycol and trimethyl orthoacetate with p-toluenesulfonic acid catalysis to form the ketal intermediate.
- Perform allylic oxidation using N-hydroxyphthalimide and cobalt acetate under air flow to introduce the 7-hydroxyl functionality.
- Reduce the resulting ketone to the corresponding alcohol using potassium borohydride to prepare the epoxidation substrate.
- Execute the critical regioselective epoxidation at the 5,6-position using tert-butyl hydroperoxide and vanadyl acetylacetonate catalyst at 40-45°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route translates into tangible strategic advantages that go beyond mere technical elegance. The primary value proposition lies in the drastic simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures and enhanced supply security. By eliminating the need for complex separation of multiple epoxide isomers, the process reduces the consumption of expensive chromatography media and solvents, leading to substantial cost savings in raw material usage. Furthermore, the mild reaction conditions (40-45°C) and the use of common solvents like dichloromethane mean that the process can be executed in standard glass-lined or stainless steel reactors without the need for specialized cryogenic or high-pressure equipment. This compatibility with existing infrastructure lowers the barrier to entry for contract manufacturing organizations (CMOs) and allows for rapid technology transfer.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the significant improvement in yield and the reduction in purification steps. Unlike the legacy alkylation routes which suffer from yields as low as 12%, this method achieves high conversion rates, meaning less starting material is wasted. The elimination of transition metal catalysts that require rigorous removal steps (often needed in other cross-coupling strategies) further streamlines the workflow. Consequently, the cost reduction in pharmaceutical manufacturing is realized through higher throughput per batch and lower waste disposal costs, creating a more competitive pricing structure for the final API intermediate.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by processes that rely on exotic reagents or produce inconsistent quality. This method utilizes widely available commodity chemicals such as ethylene glycol, tert-butyl hydroperoxide, and vanadyl acetylacetonate, reducing the risk of raw material shortages. The robustness of the reaction against minor variations in conditions ensures consistent batch-to-batch quality, which is essential for maintaining regulatory compliance and avoiding production delays. By securing a reliable API intermediate supplier who utilizes this stable technology, pharmaceutical companies can mitigate the risk of stockouts and ensure a steady flow of materials for clinical and commercial needs.
- Scalability and Environmental Compliance: The process is explicitly designed with commercial scale-up in mind, featuring simple workup procedures like filtration and crystallization that scale linearly from the laboratory to the plant. The reduced generation of hazardous byproducts and the avoidance of heavy metal contaminants align with increasingly stringent environmental regulations. This facilitates easier permitting and reduces the environmental footprint of the manufacturing site. The ability to perform commercial scale-up of complex bile acid derivatives using this greener chemistry approach positions manufacturers as leaders in sustainable pharmaceutical production, appealing to environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical aspects of adopting this route.
Q: Why is the vanadyl-catalyzed epoxidation superior to traditional alkylation methods for Obeticholic Acid?
A: Traditional alkylation methods, such as those described in WO2002072598, suffer from extremely poor selectivity and low yields (often around 12%) due to competing reactions. The vanadyl-catalyzed route described in CN112341516A achieves high regioselectivity for the 5,6-epoxide, minimizing impurities and significantly improving overall yield without requiring complex purification steps.
Q: What are the specific challenges with prior art epoxidation routes involving 4,6-diene compounds?
A: Prior art routes utilizing 4,6-diene compounds face severe regioselectivity issues because the molecule contains multiple double bonds (at positions 4,5 and 22,23) that are all susceptible to oxidation. This leads to a complex mixture of mono-epoxides, di-epoxides, and stereoisomers, making isolation of the desired 6,7-alpha epoxide difficult and costly. The new method avoids these poly-unsaturated precursors.
Q: Is this synthesis process suitable for large-scale industrial production?
A: Yes, the process is explicitly designed for industrial scalability. It utilizes mild reaction conditions (40-45°C), common organic solvents like dichloromethane, and readily available raw materials. The absence of extreme temperatures or pressures, combined with simple workup procedures like filtration and crystallization, makes it highly amenable to commercial scale-up in standard chemical reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Obeticholic Acid Intermediate Supplier
The technological breakthroughs detailed in CN112341516A represent a significant leap forward in the efficient production of Obeticholic acid precursors, yet translating patent claims into commercial reality requires deep process engineering expertise. NINGBO INNO PHARMCHEM stands at the forefront of this translation, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with the advanced reactor systems necessary to handle the specific thermal and mixing requirements of the vanadyl-catalyzed epoxidation, ensuring that the high selectivity observed in the lab is maintained at the tonnage scale. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of high-purity steroid intermediates meets the exacting standards required for FDA and EMA filings.
We invite global pharmaceutical partners to collaborate with us to optimize this route for your specific supply chain needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to this novel synthesis method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most robust and cost-effective chemistry available in the market.
