Advanced Manufacturing of Cinacalcet Hydrochloride: A Technical Breakthrough for Global Supply Chains
Advanced Manufacturing of Cinacalcet Hydrochloride: A Technical Breakthrough for Global Supply Chains
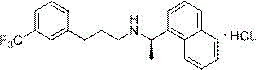
The pharmaceutical industry continuously seeks robust synthetic routes that balance high purity with economic viability, particularly for complex active pharmaceutical ingredients like Cinacalcet Hydrochloride. A recent technological advancement disclosed in patent CN112830879A presents a novel preparation method that fundamentally shifts the production paradigm by utilizing m-trifluoromethyl benzaldehyde as a primary feedstock. This approach addresses critical bottlenecks associated with traditional synthesis, such as the instability of imine intermediates and the high cost of chiral starting materials. By integrating a one-pot reductive amination strategy with efficient catalytic hydrogenation, this method offers a streamlined pathway that enhances both yield and optical purity. For R&D directors and procurement specialists, understanding the mechanistic nuances of this patent is essential for evaluating its potential to reduce manufacturing costs and secure a reliable supply of this critical calcimimetic agent.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cinacalcet has been plagued by significant technical and economic challenges that hinder efficient commercial scale-up. Conventional routes frequently rely on expensive and difficult-to-obtain starting materials such as trifluoromethylamphetamine, trifluoromethylphenylpropionic acid, or their alcohol derivatives, which drastically inflate the raw material cost base. Furthermore, these traditional pathways often necessitate the isolation of imine intermediates, which are inherently unstable and prone to hydrolysis under acidic or alkaline conditions, reverting to cinnamaldehyde and naphthylethylamine. This instability not only complicates purification but also leads to the generation of diverse impurities that are difficult to remove, ultimately compromising the optical purity of the final product. Additionally, many existing methods employ metal catalysts that require stringent reaction conditions and sophisticated removal processes to meet regulatory limits for residual metals in APIs.
The Novel Approach
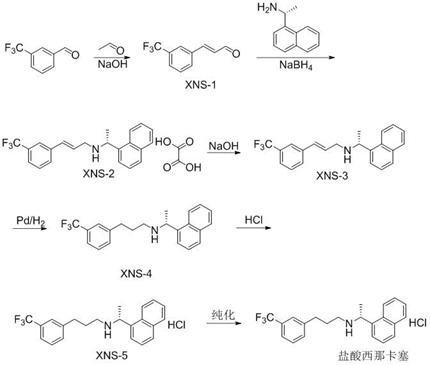
In stark contrast, the methodology outlined in CN112830879A introduces a transformative strategy that circumvents these historical pitfalls through intelligent process design. The core innovation lies in the direct conversion of m-trifluoromethyl cinnamaldehyde into a stable oxalate intermediate via a one-pot reaction with R-1-(1-naphthyl)ethylamine and sodium borohydride. By avoiding the isolation of the unstable free imine and instead immediately forming a salt, the process effectively locks the molecular structure, preventing hydrolysis and minimizing side reactions. This approach not only simplifies the operational workflow but also significantly enhances the purity profile of the intermediate, facilitating easier downstream purification. The subsequent use of Pd/C catalytic hydrogenation at room temperature further exemplifies the process's efficiency, offering a greener and safer alternative to high-pressure or high-temperature hydrogenation techniques commonly found in legacy protocols.
Mechanistic Insights into Pd/C Catalytic Hydrogenation and Chiral Preservation
The success of this synthetic route hinges on the precise control of stereochemistry during the reductive amination and hydrogenation steps. The initial condensation of m-trifluoromethyl benzaldehyde with acetaldehyde to form the cinnamaldehyde derivative sets the stage for the introduction of the chiral amine. Crucially, the use of sodium borohydride in the presence of the chiral amine facilitates a diastereoselective reduction that favors the formation of the desired (R)-enantiomer. The immediate salification with oxalic acid serves a dual purpose: it stabilizes the newly formed amine against racemization and allows for the crystallization of the intermediate, which acts as a powerful purification step to remove unreacted starting materials and by-products before the final reduction. This strategic intervention ensures that the optical integrity of the molecule is maintained throughout the synthesis, a critical parameter for the biological efficacy of Cinacalcet.
Furthermore, the catalytic hydrogenation step utilizing Pd/C represents a masterclass in process optimization for industrial applications. Unlike homogeneous catalysts that are difficult to separate, the heterogeneous Pd/C catalyst can be easily filtered and recycled, reducing both material costs and environmental waste. The reaction proceeds efficiently at room temperature and moderate hydrogen pressure, eliminating the need for energy-intensive heating or specialized high-pressure equipment. This mild condition is particularly beneficial for preserving the stereochemical configuration of the sensitive naphthyl-ethylamine moiety, preventing thermal degradation or epimerization. The result is a high-purity Cinacalcet base that requires minimal downstream processing to meet the stringent quality standards required for pharmaceutical-grade active ingredients.
How to Synthesize Cinacalcet Hydrochloride Efficiently
Implementing this novel synthesis requires strict adherence to the optimized reaction parameters defined in the patent to ensure maximum yield and purity. The process begins with the careful control of temperature during the initial aldol condensation, followed by the precise stoichiometric addition of reagents in the one-pot reductive amination. Operators must maintain low temperatures during the salification and deacidification steps to prevent thermal stress on the intermediates. The detailed standardized synthesis steps below outline the critical control points necessary for successful replication of this high-efficiency route in a GMP environment.
- Condense m-trifluoromethyl benzaldehyde with acetaldehyde under alkaline conditions at low temperature to form m-trifluoromethyl cinnamaldehyde (XNS-1).
- Perform a one-pot reaction with R-1-(1-naphthyl)ethylamine and sodium borohydride, followed by salification with oxalic acid to isolate the stable oxalate intermediate (XNS-2).
- Deacidify the oxalate intermediate using an alkaline solvent to obtain the free amine intermediate (XNS-3), then subject it to Pd/C catalytic hydrogenation to yield Cinacalcet.
- React the resulting Cinacalcet with hydrochloric acid at low temperature to form the final hydrochloride salt, followed by recrystallization for high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented methodology offers profound strategic advantages that extend beyond mere technical feasibility. The shift to m-trifluoromethyl benzaldehyde as a starting material leverages a widely available commodity chemical, thereby insulating the supply chain from the volatility and scarcity often associated with specialized chiral building blocks. This fundamental change in raw material sourcing translates directly into a more resilient and cost-effective supply network, reducing the risk of production delays caused by material shortages. Moreover, the simplification of the synthetic sequence reduces the overall number of unit operations, which lowers labor costs and minimizes the consumption of solvents and utilities, contributing to a leaner manufacturing footprint.
- Cost Reduction in Manufacturing: The elimination of expensive starting materials like trifluoromethylamphetamine significantly lowers the direct material cost per kilogram of the final API. Additionally, the ability to recycle the Pd/C catalyst and the avoidance of complex metal scavenging steps further drive down operational expenditures. The one-pot nature of the key intermediate formation reduces solvent usage and waste disposal costs, creating a substantially more economical production model compared to legacy multi-step processes.
- Enhanced Supply Chain Reliability: By relying on commercially abundant feedstocks, manufacturers can secure long-term supply contracts with multiple vendors, mitigating the risk of single-source dependency. The robustness of the synthetic route, characterized by stable intermediates and mild reaction conditions, ensures consistent batch-to-batch quality and high throughput. This reliability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients without compromise.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard reactor equipment and avoiding hazardous reagents or extreme conditions. The use of aqueous workups and recyclable catalysts aligns with modern green chemistry principles, simplifying regulatory compliance regarding environmental emissions and waste management. This eco-friendly profile not only reduces the environmental impact but also streamlines the permitting process for new manufacturing facilities, accelerating time-to-market for generic or branded versions of the drug.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the process capabilities. Stakeholders are encouraged to review these points to assess the fit of this technology within their existing manufacturing frameworks.
Q: How does this new method improve impurity control compared to traditional routes?
A: Traditional routes often involve isolating unstable imine intermediates which hydrolyze easily, leading to high impurity levels. This patent utilizes a one-pot reductive amination followed by immediate salification to form a stable oxalate intermediate, effectively locking the stereochemistry and preventing hydrolysis-related impurities.
Q: What are the cost advantages of using m-trifluoromethyl benzaldehyde as a starting material?
A: Unlike conventional methods that rely on expensive and hard-to-source materials like trifluoromethylamphetamine or trifluoromethylphenylpropanol, m-trifluoromethyl benzaldehyde is a commodity chemical that is economically viable and readily available in bulk quantities, significantly lowering the raw material cost basis.
Q: Is the catalytic hydrogenation step scalable for industrial production?
A: Yes, the process employs Pd/C catalytic hydrogenation which can be conducted at room temperature and moderate pressure. The catalyst is recyclable, reducing waste and operational costs, making the process highly suitable for large-scale commercial manufacturing with enhanced safety profiles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cinacalcet Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative patents like CN112830879A can be seamlessly translated into industrial reality. We are committed to delivering high-purity Cinacalcet Hydrochloride that meets stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our infrastructure is designed to handle complex chiral syntheses with the utmost precision, guaranteeing product consistency and regulatory compliance for our partners.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cost-effective and robust manufacturing route. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, allowing you to make informed decisions that optimize your supply chain and enhance your product portfolio's profitability.
