Advanced Convergent Synthesis of Gefitinib Intermediates for Commercial Scale Production
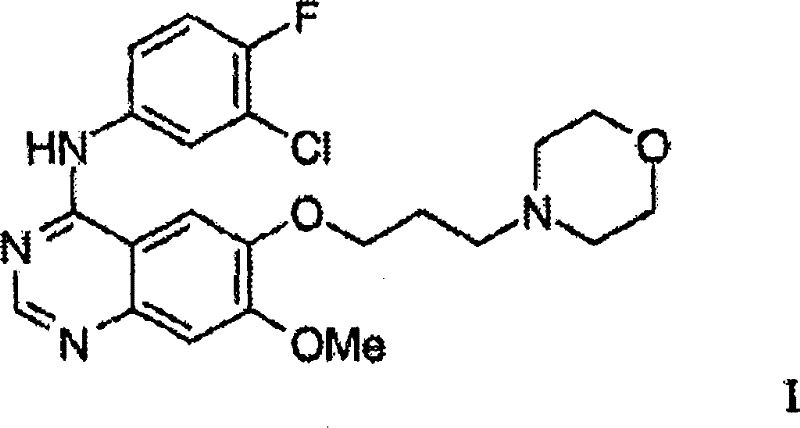
The pharmaceutical industry continuously seeks robust, scalable pathways for the production of high-value kinase inhibitors, particularly those targeting the EGFR family. Patent CN101348471B discloses a groundbreaking convergent synthesis strategy for the preparation of 4-(3'-chloro-4'-fluoroanilino)-7-methoxy-6-(3-morpholinopropoxy)quinazoline, widely known as Gefitinib. This compound, a potent tyrosine kinase inhibitor, represents a critical therapeutic agent in oncology, necessitating manufacturing processes that ensure both high purity and economic viability. The disclosed technology fundamentally shifts the paradigm from traditional linear syntheses, which are often plagued by low overall yields and reliance on resource-intensive purification techniques, to a more efficient convergent model. By introducing novel intermediates such as Formula II and Formula III, the patent outlines a route that significantly reduces the number of isolation steps and completely eliminates the need for chromatographic purification. This advancement is pivotal for reliable API intermediate suppliers aiming to meet the stringent quality and volume demands of global pharmaceutical markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex quinazoline derivatives like Gefitinib has been hindered by the inefficiencies inherent in linear synthetic routes. Traditional methodologies, such as those disclosed in earlier international applications, typically involve constructing the side chain at the very end of the synthesis or relying on intermediates that require extensive purification after every transformation. These legacy processes often mandate the use of column chromatography to separate closely related impurities, a technique that is notoriously difficult to scale beyond kilogram quantities due to solvent consumption and time constraints. Furthermore, linear approaches frequently suffer from cumulative yield losses; as the number of steps increases, the overall yield drops precipitously, driving up the cost of goods sold (COGS). The reliance on protecting groups and harsh reaction conditions in these older routes also introduces significant safety hazards and environmental burdens, making them less attractive for modern cost reduction in pharmaceutical manufacturing initiatives.
The Novel Approach
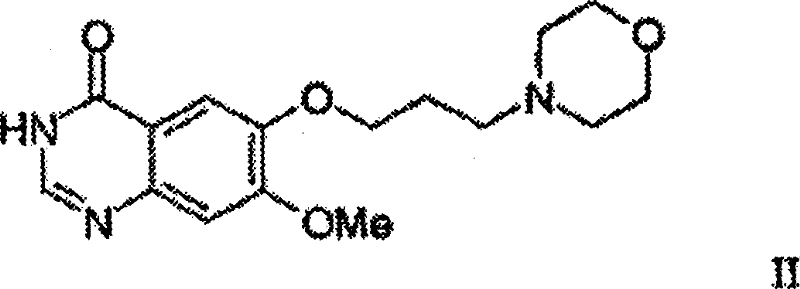
In stark contrast, the methodology presented in CN101348471B employs a convergent strategy that decouples the formation of the quinazoline core from the final aniline coupling, allowing for parallel optimization of distinct molecular fragments. A key innovation is the utilization of 7-methoxy-6-(3-morpholinopropoxy)-3,4-dihydroquinazolin-4-one (Formula II) as a pivotal intermediate. This approach allows the morpholine-containing side chain to be installed early in the sequence on a stable aromatic precursor, rather than as a late-stage modification. By synthesizing the core heterocycle first and then performing the final nucleophilic aromatic substitution with 3-chloro-4-fluoroaniline, the process achieves superior control over regioselectivity and impurity profiles. This structural modularity not only simplifies the workflow but also enhances the commercial scale-up of complex pharmaceutical intermediates by enabling the production of large batches of key building blocks that can be stockpiled and reacted on demand.
Mechanistic Insights into Reductive Cyclization and Hydration
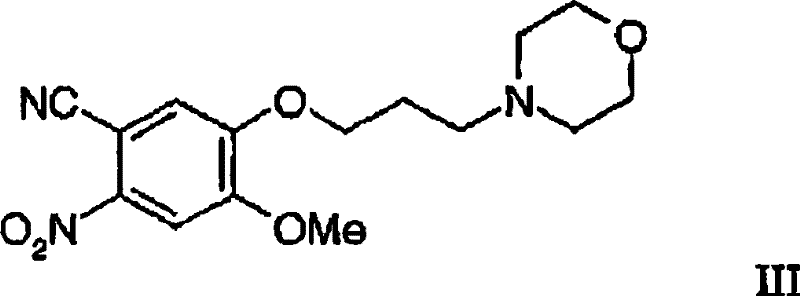
The heart of this synthetic breakthrough lies in the elegant transformation of the nitrobenzonitrile precursor (Formula III) into the quinazolinone core (Formula II) through a sequence of reduction, hydration, and cyclization. The initial step involves the selective reduction of the nitro group in Formula III to an amino group, yielding Formula IV. The patent details versatile conditions for this transformation, including catalytic hydrogenation using palladium on carbon or chemical reduction using activated metals like iron or sodium dithionite (V-Brite B). The use of V-Brite B in aqueous media at moderate temperatures (approximately 50°C) is particularly noteworthy for its operational simplicity and safety profile. Following reduction, the resulting aminobenzonitrile undergoes hydration to form the benzamide (Formula V). This step is typically conducted under alkaline conditions using potassium hydroxide in polar protic solvents like tertiary amyl alcohol, ensuring complete conversion of the nitrile functionality without degrading the sensitive ether linkages present in the molecule.
The final mechanistic cornerstone is the cyclodehydration of the benzamide (Formula V) to form the quinazolinone ring system (Formula II). This transformation is efficiently achieved using formic acid or its reactive derivatives, such as formamide, acting as both reagent and solvent. The reaction proceeds through the formation of an N-formyl intermediate which subsequently undergoes intramolecular cyclization with the loss of water. The patent specifies that this cyclization can be driven to completion at temperatures around 100°C, often in the presence of excess formamide to push the equilibrium forward. Crucially, the intermediates Formula IV and Formula V do not necessarily need to be isolated; they can be telescoped in a "one-pot" fashion directly from Formula III. This telescoping capability minimizes material handling, reduces solvent waste, and prevents the accumulation of impurities that might arise during isolation, thereby delivering high-purity quinazoline derivatives suitable for subsequent chlorination and aniline coupling steps.
How to Synthesize 7-Methoxy-6-(3-morpholinopropoxy)quinazolinone Efficiently
The practical execution of this synthesis requires precise control over reaction parameters to maximize yield and minimize byproduct formation. The process begins with the preparation of the nitro-precursor, followed by the critical reduction-cyclization sequence described above. Operators must maintain strict temperature controls during the exothermic reduction phase and ensure adequate mixing during the heterogeneous hydration step. The final cyclization requires careful removal of water to drive the equilibrium toward the product. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized protocol below which encapsulates the industrial best practices derived from the patent examples.
- Reduce 4-methoxy-5-(3-morpholinopropoxy)-2-nitrobenzonitrile (Formula III) using activated metal or catalytic hydrogenation to obtain the corresponding aminobenzonitrile (Formula IV).
- Perform hydration of the nitrile group in Formula IV under alkaline conditions to generate the benzamide derivative (Formula V).
- Execute cyclization of Formula V with formic acid or its reactive derivatives (e.g., formamide) at elevated temperatures to yield the quinazolinone core (Formula II).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates into tangible strategic benefits that extend far beyond simple chemistry. The elimination of chromatographic purification is perhaps the most significant economic driver; column chromatography is a major bottleneck in fine chemical manufacturing, consuming vast quantities of silica gel and solvents while limiting batch sizes. By replacing this with crystallization-based purifications, the process drastically reduces raw material costs and waste disposal fees. Furthermore, the ability to telescope multiple steps (reduction, hydration, cyclization) without isolating intermediates significantly shortens the production cycle time. This efficiency gain allows manufacturers to respond more agilely to market fluctuations and demand spikes, ensuring a steady flow of materials for downstream API production without the need for excessive safety stock.
- Cost Reduction in Manufacturing: The economic impact of this route is profound due to the simplification of the unit operations. By removing the need for expensive transition metal catalysts in certain variations and avoiding chromatography entirely, the direct manufacturing costs are substantially lowered. The use of commodity chemicals like formic acid, potassium hydroxide, and sodium dithionite ensures that reagent costs remain stable and predictable. Additionally, the high yields reported in the examples (e.g., 99% yield for the reduction step in Example 1) mean that less starting material is required per kilogram of final product, directly improving the margin profile for reliable API intermediate suppliers.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the intermediates involved. Formula III and Formula II are stable compounds that can be synthesized in large campaigns and stored for extended periods without significant degradation. This stability decouples the production of the core scaffold from the final coupling step, allowing for a flexible manufacturing schedule. If demand for the final API increases, manufacturers can simply draw from inventory of the advanced intermediate rather than restarting the entire synthesis from basic raw materials, thereby reducing lead time for high-purity pharmaceutical intermediates and mitigating the risk of production delays.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process aligns perfectly with green chemistry principles. The reduction in solvent usage, particularly the avoidance of large volumes of eluents required for chromatography, significantly lowers the facility's environmental footprint. The reactions are conducted in standard solvents like alcohols and esters, which are easier to recover and recycle compared to halogenated solvents often used in older routes. The scalability is evidenced by the patent examples which describe reactions on scales involving hundreds of kilograms of reagents and hundreds of liters of solvent, demonstrating that the chemistry translates seamlessly from the laboratory bench to multi-ton commercial production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific challenges associated with quinazoline chemistry and the solutions provided by the patent's innovative methodology. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this process for their own supply chains.
Q: How does this novel process improve upon conventional linear synthesis routes for Gefitinib?
A: Conventional routes often rely on linear synthesis requiring multiple isolation steps and expensive chromatographic purification, which drastically lowers overall yield. This patented convergent approach minimizes intermediate isolations and eliminates the need for column chromatography, significantly enhancing throughput and reducing production costs.
Q: What specific reagents are utilized for the critical reduction step in this methodology?
A: The process offers flexibility in reduction methods, utilizing either catalytic hydrogenation with palladium on carbon or chemical reduction using activated metals like iron. Notably, the use of water-soluble inorganic reducing agents such as sodium dithionite (V-Brite B) allows for efficient conversion at moderate temperatures (around 50°C), ensuring high purity without complex workups.
Q: Is the intermediate 4-methoxy-5-(3-morpholinopropoxy)-2-nitrobenzonitrile commercially stable?
A: Yes, Formula III is a stable, novel intermediate specifically designed for this route. It can be synthesized via a straightforward alkylation of 3-hydroxy-4-methoxybenzonitrile followed by nitration. Its stability allows it to be stored or transported as a solution, facilitating a 'one-pot' telescoping process that further streamlines manufacturing logistics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gefitinib Intermediate Supplier
The technological advancements detailed in CN101348471B represent a significant leap forward in the manufacture of EGFR inhibitors, offering a pathway that balances high quality with industrial efficiency. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to leverage these innovations for our global partners. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of intermediate meets the exacting standards required for pharmaceutical applications.
We invite potential partners to engage with us to explore how this optimized synthesis route can benefit your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how this convergent approach can lower your total cost of ownership. Please contact us today to request specific COA data for our quinazoline intermediates and to discuss route feasibility assessments for your upcoming development programs.
