Advanced Synthetic Route for Gefitinib Intermediates: Enhancing Purity and Scalability
Advanced Synthetic Route for Gefitinib Intermediates: Enhancing Purity and Scalability
The pharmaceutical landscape for oncology treatments continues to evolve, with Gefitinib standing as a cornerstone therapy for non-small cell lung cancer. As a potent EGFR tyrosine kinase inhibitor, the demand for high-purity Gefitinib and its key intermediates remains robust across global markets. Patent CN102120731A introduces a transformative manufacturing methodology that addresses longstanding inefficiencies in the synthesis of 4-(3-chloro-4-fluorophenylamino)-7-methoxyl-6-(3-morpholinepropoxy)quinazoline. This technical disclosure outlines a pathway that not only streamlines the reaction sequence but also drastically improves the purity profile of the final active pharmaceutical ingredient (API). For R&D directors and procurement specialists, understanding the nuances of this novel route is essential for optimizing supply chains and reducing the cost of goods sold (COGS) in competitive generic and proprietary drug markets.
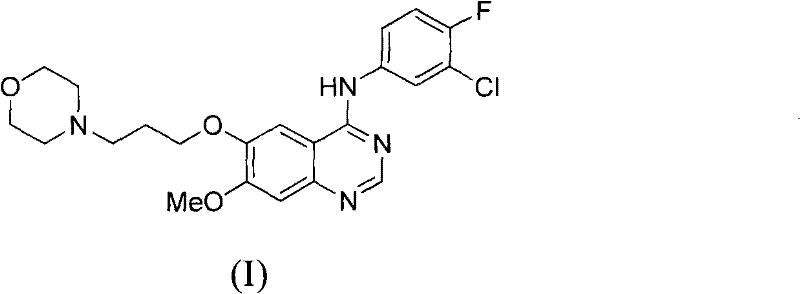
The structural complexity of Gefitinib requires precise control over substitution patterns on the quinazoline ring. Traditional methods often struggle with regioselectivity and the removal of trace impurities that can compromise biological efficacy. The approach detailed in CN102120731A leverages a strategic assembly of the side chain prior to ring closure, ensuring that the critical methoxy and morpholine-propoxy groups are installed with high fidelity before the final heterocyclic formation. This forward-thinking design minimizes the risk of side reactions that typically plague late-stage functionalization, thereby offering a more reliable source of high-purity pharmaceutical intermediates for downstream processing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthesis routes for Gefitinib, such as those described in early patents like WO9633980, rely heavily on the selective demethylation of 6,7-dimethoxyquinazoline-4(3H)-one. This conventional pathway is fraught with significant technical hurdles that impede efficient commercial scale-up. The selectivity demethylation step typically requires harsh reagents like methanesulfonic acid and L-methionine, which are not only difficult to recover but also generate substantial chemical waste, posing severe environmental compliance challenges. Furthermore, the linear nature of these older routes results in low overall yields, necessitating extensive purification protocols.
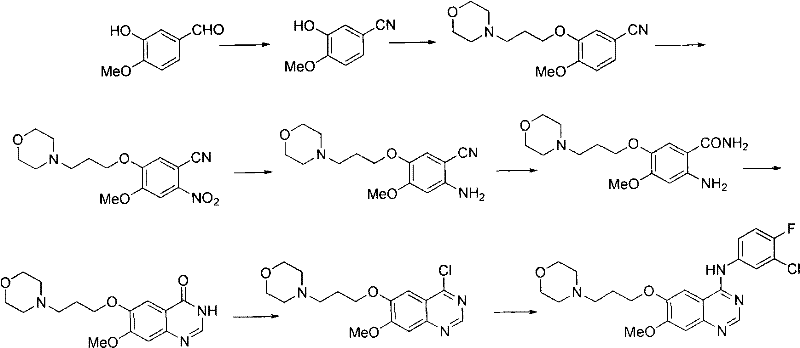
A critical bottleneck in these legacy methods is the reliance on column chromatography for purifying key intermediates. In an industrial setting, column chromatography is economically unviable due to high solvent consumption, low throughput, and difficulties in automation. Additionally, the final etherification step in conventional routes often suffers from N-alkylation side reactions, further complicating the isolation of the target molecule. These factors collectively drive up production costs and extend lead times, making it difficult for manufacturers to meet the rigorous price and volume demands of the global supply chain without compromising on quality standards.
The Novel Approach
In stark contrast, the methodology presented in CN102120731A circumvents these pitfalls by employing a convergent strategy centered around a nitrile intermediate. By starting with readily available 3-hydroxy-4-methoxybenzaldehyde, the process builds the molecular complexity through a series of high-yielding transformations including etherification, nitration, and a unique dual functional group conversion. This route effectively bypasses the problematic selective demethylation step entirely. Instead, it utilizes a Dimroth rearrangement reaction for the final ring closure, which proceeds under much milder conditions than the high-temperature rearrangements seen in other prior art like WO2005023783.
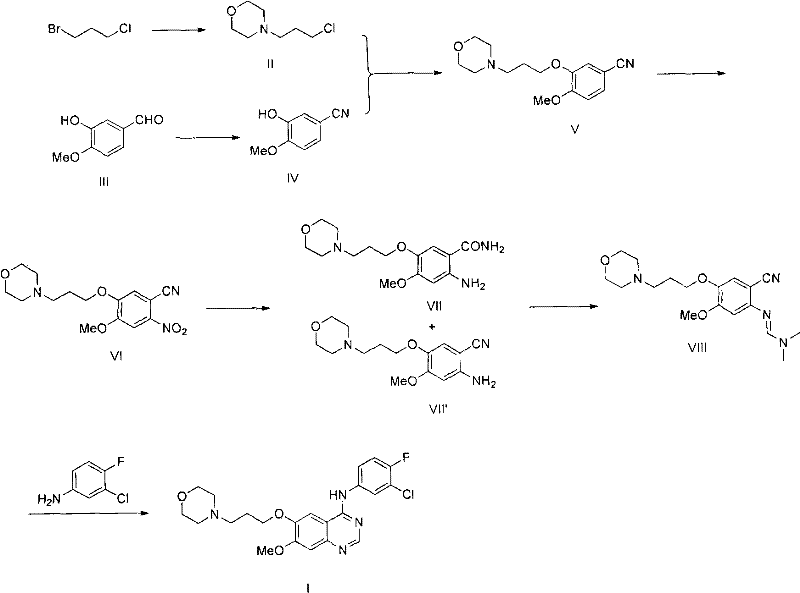
The elegance of this new approach lies in its operational simplicity and robustness. The intermediates generated along this pathway, such as the nitro-benzonitrile derivative and the subsequent amino-amide mixture, are stable and easily purified through standard crystallization or extraction techniques, completely eliminating the need for column chromatography. This shift from chromatographic purification to crystallization-based isolation represents a paradigm shift for process chemistry, enabling true continuous manufacturing potential. The result is a streamlined process that delivers the target quinazoline with exceptional purity, directly addressing the needs of a reliable API intermediate supplier seeking to optimize their manufacturing footprint.
Mechanistic Insights into the Dimroth Rearrangement and Cyclization
The core innovation of this patent lies in the mechanistic pathway leading to the quinazoline ring formation. The process begins with the conversion of a nitro-benzonitrile intermediate into a mixture of an amino-benzamide and an amino-benzonitrile via catalytic hydrogenation. This mixture is then subjected to a second functional group transformation using chlorinating agents such as phosphorus pentachloride or thionyl chloride in the presence of dimethylformamide (DMF). This step generates a reactive 2-(N,N-dimethylformimidoyl) intermediate, which serves as the electrophilic partner for the subsequent cyclization.
The final ring closure involves the reaction of this imidate intermediate with 3-chloro-4-fluoroaniline. Under acidic conditions and elevated temperatures, a Dimroth rearrangement occurs. This mechanism involves the nucleophilic attack of the aniline nitrogen on the imidate carbon, followed by a rearrangement that establishes the pyrimidine ring of the quinazoline core. Unlike traditional methods that might require extreme thermal energy leading to decomposition, this specific rearrangement is optimized to proceed efficiently at moderate temperatures (e.g., 100°C in acetic acid). The mechanistic precision ensures that the 4-anilino substituent is installed regioselectively, minimizing the formation of isomeric impurities that are notoriously difficult to separate in later stages.
How to Synthesize Gefitinib Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. It emphasizes the use of common solvents like toluene, DMF, and acetic acid, alongside standard reagents such as potassium carbonate and palladium on carbon. The procedure is designed to be telescoped where possible, reducing the number of isolation steps and maximizing material throughput. For detailed operational parameters, stoichiometry, and specific workup procedures required to replicate these results in a GMP environment, please refer to the standardized synthesis guide below.
- Preparation of 3-morpholine propyl chloride from 1-bromo-3-chloropropane and morpholine.
- Etherification of 2-hydroxy-3-methoxybenzonitrile with the chloro-intermediate to form the nitrile precursor.
- Nitration followed by hydrogenation to generate an amino/amide mixture, conversion to an imidate, and final cyclization with 3-chloro-4-fluoroaniline.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers compelling economic and logistical benefits. The primary driver for cost reduction is the elimination of expensive and time-consuming purification steps. By removing the dependency on column chromatography, manufacturers can significantly reduce solvent usage and waste disposal costs, which are major components of the total manufacturing expense. Furthermore, the use of commodity chemicals like 3-hydroxy-4-methoxybenzaldehyde and morpholine ensures a stable and cost-effective raw material supply base, insulating the production process from volatility in specialty chemical markets.
- Cost Reduction in Manufacturing: The streamlined nature of this synthesis directly translates to lower operational expenditures. The high yields reported in the patent examples, often exceeding 90% for intermediate steps, mean that less raw material is wasted per kilogram of final product. Additionally, the avoidance of harsh demethylation reagents reduces the burden on waste treatment facilities and lowers the cost associated with hazardous material handling. This efficiency allows for a more competitive pricing structure for the final API, providing a distinct market advantage.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to greater batch-to-batch consistency. Traditional routes that rely on sensitive selective demethylation are prone to variability, which can lead to batch failures and supply disruptions. In contrast, the nitration and hydrogenation steps in this new route are well-understood unit operations that can be tightly controlled. This reliability ensures a steady flow of high-purity intermediates, reducing the risk of stockouts and enabling manufacturers to meet strict delivery schedules for their pharmaceutical clients.
- Scalability and Environmental Compliance: From a sustainability perspective, this method aligns with green chemistry principles by minimizing waste generation. The ability to purify intermediates via crystallization rather than chromatography significantly reduces the volume of organic solvents required, simplifying solvent recovery and recycling processes. This makes the process inherently more scalable, allowing for seamless transition from pilot batches to multi-ton commercial production without the engineering bottlenecks associated with large-scale chromatographic columns. Such scalability is crucial for meeting the growing global demand for oncology therapeutics.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this specific Gefitinib synthesis pathway. These insights are derived directly from the comparative data and experimental results presented in the patent documentation, aimed at clarifying the operational advantages for technical stakeholders.
Q: What are the primary advantages of this new synthetic route over conventional methods?
A: The novel method avoids harsh demethylation conditions and eliminates the need for column chromatography purification, significantly reducing production costs and environmental impact while improving overall yield.
Q: How does the process handle the critical cyclization step?
A: The process utilizes a Dimroth rearrangement reaction between a specific imidate intermediate and 3-chloro-4-fluoroaniline, which proceeds under milder conditions compared to traditional high-temperature rearrangements.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method employs readily available raw materials, avoids complex purification techniques like column chromatography, and demonstrates high yields in each step, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gefitinib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex oncology drugs depends on a robust and efficient supply chain. Our team of expert chemists has extensively evaluated the synthetic route described in CN102120731A and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Gefitinib intermediate meets the highest international regulatory standards.
We invite pharmaceutical partners to collaborate with us to leverage this advanced technology for their next-generation formulations. By optimizing the synthesis of key building blocks, we can help you achieve significant efficiencies in your manufacturing process. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and commercialization goals, ensuring a seamless integration of our high-quality intermediates into your supply chain.
