Advanced Gefitinib Manufacturing: A Safer, Scalable Route for Global Supply Chains
Advanced Gefitinib Manufacturing: A Safer, Scalable Route for Global Supply Chains
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical oncology therapeutics, and the synthesis of Gefitinib, a potent EGFR tyrosine kinase inhibitor, remains a focal point for process optimization. Patent CN103304491A introduces a transformative preparation method that addresses longstanding inefficiencies in the production of this vital anticancer agent. By re-engineering the synthetic sequence, this technology offers a streamlined approach that bypasses hazardous chlorination steps and high-pressure hydrogenation, thereby enhancing both operational safety and environmental compliance. For global supply chain leaders, this innovation represents a significant opportunity to secure a more reliable source of high-purity pharmaceutical intermediates while mitigating the risks associated with complex, multi-step legacy processes. The following analysis details the technical merits and commercial viability of this advanced synthesis route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Gefitinib, such as those documented in earlier literature and patents like WO2004024703, are characterized by excessive complexity and significant environmental burdens. As illustrated in the reaction scheme below, these legacy methods typically involve a cumbersome sequence of peroxidation, hydrocarbylation, nitration, reduction, cyclization, and critically, chlorination steps. The reliance on chlorinating agents such as thionyl chloride or phosphorus oxychloride introduces severe safety hazards, including the generation of corrosive acidic gases and toxic halogenated waste streams that require costly disposal protocols. Furthermore, the extended reaction sequence inherently accumulates impurities at each stage, necessitating rigorous and expensive purification procedures to meet stringent pharmacopeial standards. The cumulative effect of these factors is a manufacturing process that is not only economically inefficient due to low overall yields but also operationally fragile, posing substantial challenges for consistent commercial scale-up.
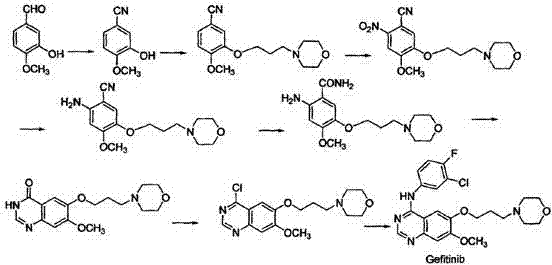
The Novel Approach
In stark contrast to the convoluted pathways of the past, the methodology disclosed in patent CN103304491A presents a concise and elegant solution that radically simplifies the production landscape. The new route initiates with the etherification of 3-hydroxy-4-methoxybenzonitrile and proceeds through a streamlined series of nitration, reduction, and cyclization reactions, completely eliminating the need for dangerous chlorination steps. As depicted in the schematic below, the process utilizes 3-chloro-4-fluoroaniline and DMF dimethylacetal to form a key imine intermediate, which then undergoes cyclization with the amino-benzonitrile derivative to yield the final product. This strategic redesign not only shortens the overall synthetic timeline but also operates under ambient pressure conditions, removing the requirement for specialized high-pressure reactors. The result is a process that is inherently safer, easier to operate, and significantly more conducive to green chemistry principles, offering a compelling alternative for manufacturers seeking to optimize their production capabilities.
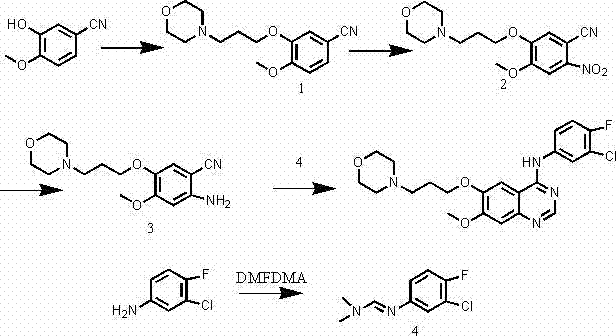
Mechanistic Insights into Acid-Catalyzed Cyclization
The cornerstone of this innovative synthesis lies in the final cyclization step, where the amino-benzonitrile intermediate reacts with the pre-formed imine derivative to construct the quinazoline core of Gefitinib. This transformation is facilitated by the presence of acidic catalysts, such as acetic acid or hydrochloric acid, often in conjunction with phase transfer catalysts like tetrabutylammonium chloride. The mechanism involves the nucleophilic attack of the amino group on the electrophilic carbon of the imine, followed by intramolecular cyclization and subsequent aromatization to form the stable heterocyclic ring system. The use of mild acidic conditions allows for precise control over the reaction kinetics, minimizing the formation of side products and ensuring high conversion rates. Furthermore, the selection of solvents such as dimethyl sulfoxide or toluene plays a crucial role in solubilizing the reactants and stabilizing the transition states, thereby driving the equilibrium towards the desired product. This mechanistic understanding is vital for R&D teams aiming to replicate the process with high fidelity and consistency.
From an impurity control perspective, the elimination of halogenating reagents in the earlier stages of the synthesis profoundly impacts the final impurity profile of the API. Traditional routes often leave traces of organochlorine byproducts that are difficult to remove and can pose genotoxicity risks. By avoiding these reagents entirely, the new process inherently reduces the burden on downstream purification, leading to a cleaner crude product. Additionally, the reduction step utilizes common reducing agents like iron powder or hydrazine hydrate under atmospheric pressure, which avoids the introduction of metal catalysts that might require complex scavenging procedures. The combination of these factors results in a superior impurity spectrum, facilitating easier compliance with regulatory requirements for residual solvents and heavy metals. This level of chemical cleanliness is a critical differentiator for suppliers targeting regulated markets.
How to Synthesize Gefitinib Efficiently
The implementation of this synthesis route requires careful attention to reaction parameters such as temperature, stoichiometry, and solvent selection to maximize yield and purity. The process is divided into five distinct operational stages, beginning with the etherification of the starting nitrile and concluding with the final cyclization and isolation of Gefitinib. Each step has been optimized to balance reaction rate with selectivity, ensuring that the process remains robust even when scaled to industrial volumes. Operators should monitor reaction progress using thin-layer chromatography (TLC) to determine optimal endpoints, particularly during the nitration and reduction phases where over-reaction can lead to degradation. The detailed standardized synthesis steps, including specific mass ratios and temperature ranges for each stage, are outlined in the guide below to assist technical teams in process validation.
- Etherification of 3-hydroxy-4-methoxybenzonitrile with 4-(3-chloropropyl)morpholine to form the nitrile intermediate.
- Nitration of the nitrile intermediate followed by reduction to obtain the amino-benzonitrile derivative.
- Formation of the imine intermediate from 3-chloro-4-fluoroaniline and DMF dimethylacetal.
- Final cyclization of the amino-benzonitrile and imine intermediate under acidic conditions to yield Gefitinib.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis method offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the substantial simplification of the supply chain for raw materials; by eliminating the need for hazardous chlorinating agents and high-pressure hydrogen gas, the process relies on widely available and stable commodity chemicals. This shift significantly reduces the logistical complexities and regulatory hurdles associated with transporting and storing dangerous goods, thereby enhancing the overall reliability of the supply network. Moreover, the shortened reaction sequence directly translates to reduced manufacturing cycle times, allowing for faster turnover and improved responsiveness to market demand fluctuations. These operational efficiencies collectively contribute to a more resilient and cost-effective production model.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive and hazardous reagents, such as thionyl chloride, which not only carry high purchase prices but also incur significant costs for safe handling and waste disposal. By removing the chlorination step and utilizing ambient pressure conditions, the capital expenditure required for specialized corrosion-resistant equipment and high-pressure reactors is drastically minimized. Furthermore, the simplified workup procedure, which often requires only washing and drying rather than multiple solvent extractions, leads to significant savings in solvent consumption and energy usage. These cumulative factors result in a markedly lower cost of goods sold, providing a competitive edge in pricing strategies for the final API.
- Enhanced Supply Chain Reliability: The reliance on readily accessible raw materials like 3-hydroxy-4-methoxybenzonitrile and 4-(3-chloropropyl)morpholine ensures a stable supply base that is less susceptible to geopolitical or logistical disruptions. Unlike processes dependent on specialized catalysts or gases that may face supply bottlenecks, this route utilizes common organic solvents and reagents that are produced at scale globally. The robustness of the reaction conditions, which tolerate a range of temperatures and do not require inert atmospheres for every step, further enhances operational continuity. This reliability is crucial for maintaining consistent inventory levels and meeting the rigorous delivery schedules demanded by downstream pharmaceutical partners.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is facilitated by the absence of exothermic hazards associated with chlorination and the lack of high-pressure requirements. The reaction can be performed in standard glass-lined or stainless steel reactors, simplifying the technology transfer process. Environmentally, the reduction in toxic waste generation aligns with increasingly stringent global regulations on industrial emissions and effluent discharge. The ability to recycle solvents and the minimization of halogenated byproducts make this route a sustainable choice for manufacturers committed to green chemistry initiatives, thereby future-proofing the production facility against evolving environmental compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear overview of the process capabilities. Understanding these aspects is essential for stakeholders evaluating the feasibility of adopting this method for their own manufacturing operations or sourcing strategies. The responses highlight the safety, efficiency, and quality advantages that distinguish this approach from conventional alternatives.
Q: How does this new synthesis route improve operational safety compared to traditional methods?
A: The novel process eliminates the use of high-risk halogenating reagents like thionyl chloride and phosphorus oxychloride, and avoids high-pressure hydrogenation, significantly reducing explosion hazards and toxic waste generation.
Q: What are the primary cost drivers reduced in this manufacturing method?
A: Costs are lowered by shortening the reaction sequence, removing the need for expensive chlorination steps, and simplifying post-processing which traditionally requires multiple solvent extractions.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the reaction operates at ambient pressure with readily available solvents and reagents, and the workup involves simple filtration and washing, making it highly amenable to multi-ton scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gefitinib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis technologies to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex chemistries like the one described in CN103304491A can be successfully translated into robust manufacturing operations. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs which employ state-of-the-art analytical techniques to verify product identity and quality. Our infrastructure is designed to handle the specific requirements of heterocyclic synthesis, providing a secure and compliant environment for the production of oncology intermediates.
We invite potential partners to engage with our technical procurement team to discuss how this innovative route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits tailored to your volume requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on comprehensive technical and commercial evidence. Together, we can drive efficiency and reliability in the production of life-saving medicines.
