Advanced Copper-Catalyzed Synthesis of Ortho-Alkyl Anilines for Commercial Scale-Up
Introduction to High-Efficiency Amination Technology
The pharmaceutical and agrochemical industries constantly seek robust methodologies for synthesizing sterically hindered aniline derivatives, which serve as critical building blocks for advanced active ingredients. Patent CN101072746B introduces a groundbreaking process for the preparation of ortho-alkyl-substituted primary phenylamines, specifically addressing the historical challenges associated with aminating deactivated benzene nuclei. This technology leverages a copper-catalyzed system that operates under high-pressure ammonolysis conditions to achieve high yields where traditional methods often fail. By utilizing catalytic amounts of copper-containing compounds, such as copper(I) oxide, the process enables the direct conversion of ortho-alkyl halobenzenes into valuable amine intermediates without the need for expensive palladium catalysts or complex multi-step protection strategies. 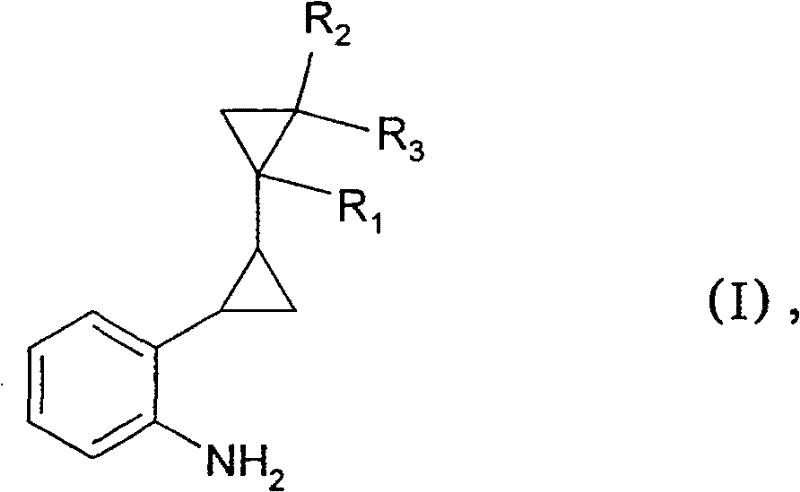 The significance of this invention lies in its ability to overcome the electronic deactivation caused by ortho-alkyl groups, which typically repel nucleophilic attack by ammonia. The disclosed method provides a scalable route to compounds like 2-dicyclopropyl-2-yl-phenylamine, a key intermediate in the synthesis of modern fungicides. For R&D directors and process chemists, this represents a viable pathway to access complex molecular architectures with improved atom economy and reduced environmental footprint compared to legacy synthetic routes.
The significance of this invention lies in its ability to overcome the electronic deactivation caused by ortho-alkyl groups, which typically repel nucleophilic attack by ammonia. The disclosed method provides a scalable route to compounds like 2-dicyclopropyl-2-yl-phenylamine, a key intermediate in the synthesis of modern fungicides. For R&D directors and process chemists, this represents a viable pathway to access complex molecular architectures with improved atom economy and reduced environmental footprint compared to legacy synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ortho-alkyl-substituted anilines has been plagued by significant technical hurdles, primarily due to the steric and electronic properties of the substrate. Conventional copper-catalyzed aminations, while well-documented for activated aryl halides or heterocycles, demonstrate poor efficacy when applied to deactivated benzene rings bearing bulky ortho-substituents. Literature precedents indicate that standard copper protocols often require stoichiometric amounts of catalyst or result in negligible conversion rates for these specific substrates. Furthermore, alternative approaches utilizing palladium catalysis, although chemically feasible, introduce substantial economic and operational burdens. These palladium-mediated routes typically necessitate a two-step sequence involving initial coupling with benzophenone imine followed by hydrolysis, which increases waste generation and extends production timelines. The reliance on precious metals also exposes the supply chain to volatile pricing and potential scarcity issues, making large-scale manufacturing economically unattractive for commodity-grade intermediates.
The Novel Approach
The novel approach detailed in the patent data circumvents these limitations by optimizing reaction parameters to facilitate direct nucleophilic substitution with ammonia. By employing a catalytic system based on inexpensive copper salts, specifically copper(I) oxide, the process achieves high conversion rates even with sterically demanding substrates like 2-(2-bromophenyl)-dicyclopropyl. The method operates at elevated temperatures ranging from 130°C to 170°C and pressures between 35 bar and 85 bar, conditions that provide the necessary activation energy to overcome the kinetic barrier of the deactivated ring. Crucially, the use of ammonia in large molar excess drives the equilibrium towards the desired primary amine, suppressing side reactions. This single-step transformation eliminates the need for protecting groups and expensive transition metals, streamlining the workflow from raw material to final intermediate. 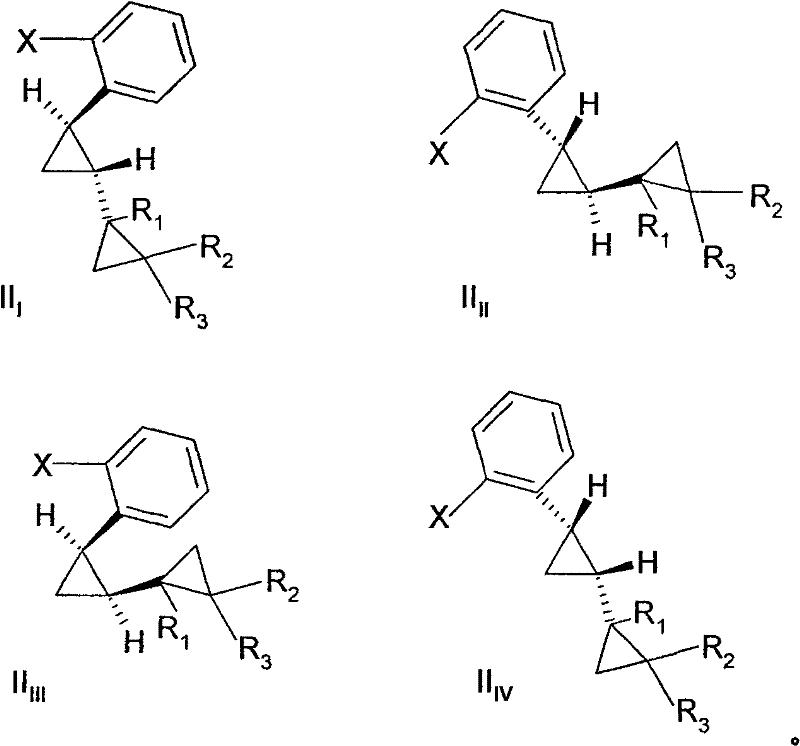 The versatility of this approach is further enhanced by its tolerance for various substituents, allowing for the synthesis of a broad library of derivatives where R1, R2, and R3 can be independently hydrogen or methyl groups, thereby offering a flexible platform for medicinal chemistry optimization.
The versatility of this approach is further enhanced by its tolerance for various substituents, allowing for the synthesis of a broad library of derivatives where R1, R2, and R3 can be independently hydrogen or methyl groups, thereby offering a flexible platform for medicinal chemistry optimization.
Mechanistic Insights into Copper-Catalyzed Nucleophilic Aromatic Substitution
The underlying mechanism of this transformation involves a copper-mediated nucleophilic aromatic substitution, where the copper catalyst likely facilitates the formation of a reactive aryl-copper intermediate or activates the aryl halide towards attack by ammonia. In the context of ortho-alkyl substituted systems, the steric bulk adjacent to the reaction center typically hinders the approach of the nucleophile; however, the high thermal energy and pressure conditions employed in this process help to surmount this steric barrier. The reaction kinetics are notably influenced by the stereochemistry of the starting material, with trans-isomers reacting significantly faster than their cis-counterparts. For instance, experimental data indicates that the trans-isomer of 2-(2-bromophenyl)-dicyclopropyl reacts approximately 1.7 times faster than the cis-isomer under identical conditions. This differential reactivity provides a mechanistic handle for controlling the stereochemical outcome of the synthesis, enabling the preferential formation of specific diastereomers through careful modulation of reaction time and temperature.
Impurity control is a critical aspect of this mechanism, particularly regarding the choice of solvent. When ethylene glycol is used as the reaction medium, there exists a competing pathway where the glycol acts as a nucleophile, potentially leading to the formation of ether byproducts. 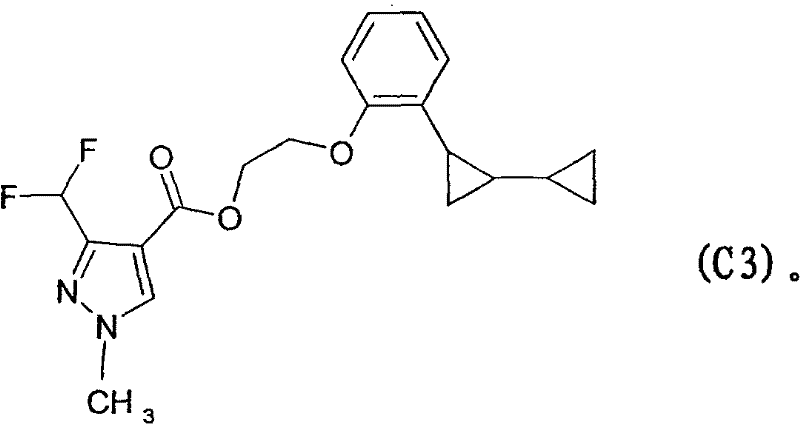 These ether impurities, such as the structure designated as C3 in the patent documentation, arise from the substitution of the halogen by the glycol moiety instead of ammonia. While these side products are formed in minor quantities, their presence necessitates robust purification strategies to ensure the high purity required for pharmaceutical applications. Understanding this competitive mechanism allows process chemists to optimize the ammonia-to-substrate ratio, ensuring that ammonia outcompetes the solvent for the electrophilic center, thereby minimizing impurity load and simplifying downstream processing.
These ether impurities, such as the structure designated as C3 in the patent documentation, arise from the substitution of the halogen by the glycol moiety instead of ammonia. While these side products are formed in minor quantities, their presence necessitates robust purification strategies to ensure the high purity required for pharmaceutical applications. Understanding this competitive mechanism allows process chemists to optimize the ammonia-to-substrate ratio, ensuring that ammonia outcompetes the solvent for the electrophilic center, thereby minimizing impurity load and simplifying downstream processing.
How to Synthesize 2-Dicyclopropyl-2-yl-phenylamine Efficiently
The synthesis of high-purity ortho-alkyl anilines requires precise control over reaction parameters to maximize yield and minimize byproduct formation. The patented procedure outlines a robust protocol involving the charging of an autoclave with the halobenzene substrate, a catalytic amount of copper(I) oxide, and a suitable solvent such as ethylene glycol. The detailed standardized synthesis steps provided below guide the operator through the critical phases of reagent addition, pressurization, and thermal management necessary to achieve reproducible results on a commercial scale. Adherence to these guidelines ensures safety and efficiency, particularly given the high-pressure nature of the ammonolysis reaction.
- Charge an autoclave with the ortho-alkyl substituted halobenzene (bromo or chloro derivative), catalytic copper(I) oxide (Cu2O), and ethylene glycol as the solvent.
- Introduce liquid or gaseous ammonia into the reactor to achieve a significant molar excess, typically ranging from 80-fold to 120-fold relative to the substrate.
- Heat the mixture to a temperature between 130°C and 170°C under elevated pressure (35-85 bar) for 6 to 24 hours, followed by standard workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this copper-catalyzed technology offers transformative advantages in terms of cost structure and material availability. The shift from precious metal catalysts to base metal catalysts fundamentally alters the cost basis of the intermediate, removing the dependency on volatile palladium markets. Furthermore, the simplification of the synthetic route from a multi-step sequence to a single direct amination step drastically reduces the consumption of solvents, reagents, and labor hours, leading to substantial overall cost savings in manufacturing operations. The use of commodity chemicals such as ammonia and simple copper salts ensures a stable and resilient supply chain, mitigating risks associated with the sourcing of specialized ligands or complex organometallic reagents.
- Cost Reduction in Manufacturing: The elimination of expensive palladium catalysts and the reduction of synthetic steps directly translate to lower production costs. By utilizing catalytic amounts of copper(I) oxide, which is significantly cheaper than palladium complexes, the process minimizes raw material expenses. Additionally, the removal of protection and deprotection steps reduces the consumption of auxiliary chemicals and waste disposal costs, enhancing the overall economic viability of producing these complex intermediates for agrochemical and pharmaceutical applications.
- Enhanced Supply Chain Reliability: The reliance on widely available raw materials such as ortho-alkyl halobenzenes and anhydrous ammonia ensures a consistent supply flow. Unlike specialized palladium catalysts which may have long lead times or limited suppliers, copper salts and ammonia are commodity chemicals produced globally in vast quantities. This abundance guarantees supply continuity and reduces the risk of production delays caused by material shortages, making the process highly reliable for meeting large-scale demand.
- Scalability and Environmental Compliance: The process is designed for scalability, utilizing standard high-pressure autoclaves common in the fine chemical industry. The simplified workflow generates less chemical waste compared to multi-step alternatives, aligning with green chemistry principles. The ability to operate without exotic reagents simplifies waste treatment protocols and reduces the environmental footprint, facilitating easier regulatory compliance and permitting for commercial-scale facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed amination technology. These insights are derived directly from the patent specifications and experimental data, providing clarity on reaction scope, safety considerations, and product quality. Understanding these aspects is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: Why is copper catalysis preferred over palladium for this specific amination?
A: While palladium catalysts are effective for deactivated halobenzenes, they often require expensive ligands and multi-step sequences involving protection/deprotection. The copper-catalyzed method described in CN101072746B allows for direct amination with ammonia in a single step, significantly reducing raw material costs and processing complexity.
Q: How does the process manage stereochemistry in dicyclopropyl derivatives?
A: The reaction kinetics favor the formation of the trans-isomer over the cis-isomer. Under optimized conditions (150°C, Cu2O catalyst), the trans-isomer reacts approximately 1.7 times faster than the cis-isomer, allowing for the enrichment of specific stereoisomers through controlled reaction times.
Q: What are the primary impurities formed during this synthesis?
A: When using ethylene glycol as a solvent, a minor side reaction can occur where the glycol acts as a nucleophile instead of ammonia, leading to ether byproducts. However, these impurities are structurally distinct and can be effectively removed via standard chromatographic or crystallization techniques.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Dicyclopropyl-2-yl-phenylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation therapeutics and crop protection agents. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of ortho-alkyl aniline meets the exacting standards required by global regulatory bodies. Our commitment to technical excellence allows us to navigate the complexities of high-pressure chemistry safely and effectively.
We invite you to collaborate with us to leverage this advanced copper-catalyzed technology for your specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and economic efficiency.
