Revolutionizing Ortho-Alkyl Aniline Production: A Cost-Effective Copper-Catalyzed Route for High-Volume Agrochemical Intermediates
Revolutionizing Ortho-Alkyl Aniline Production: A Cost-Effective Copper-Catalyzed Route for High-Volume Agrochemical Intermediates
The chemical industry constantly seeks robust methodologies to synthesize complex aromatic amines, which serve as critical building blocks for advanced agrochemicals and pharmaceuticals. A significant breakthrough in this domain is documented in patent CN101072746B, which discloses a highly efficient process for the preparation of ortho-alkyl-substituted anilines. Traditionally, the amination of deactivated benzene nuclei, such as those bearing bulky ortho-alkyl groups, has been plagued by low yields and harsh reaction conditions. This patent introduces a novel catalytic system utilizing copper compounds that overcomes these historical barriers, enabling the direct conversion of ortho-alkyl-substituted halobenzenes into primary amines with remarkable efficiency. By leveraging ammonia in the presence of catalytic copper species, this technology offers a streamlined pathway that eliminates the need for expensive palladium catalysts and complex multi-step protection strategies. For global supply chain leaders and R&D directors, this represents a pivotal shift towards more sustainable and economically viable manufacturing of key fungicide intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of primary aryl amines from deactivated aryl halides has been a formidable challenge in organic synthesis. Standard literature suggests that copper-catalyzed aminations typically require activated substrates, such as those with electron-withdrawing nitro groups, to proceed with acceptable yields. When applied to electron-rich or sterically hindered systems like ortho-alkyl-substituted halobenzenes, traditional copper methods often fail or require prohibitively long reaction times and excessive catalyst loading. Alternative approaches utilizing palladium catalysis, while effective, introduce significant economic and operational burdens. These palladium-mediated routes generally necessitate a two-step sequence involving the formation of a protected imine intermediate followed by hydrolysis. This not only increases the consumption of expensive reagents like benzophenone and hydroxylamine but also extends the production cycle, thereby inflating the overall cost of goods and complicating waste management protocols for large-scale operations.
The Novel Approach
The methodology outlined in CN101072746B fundamentally disrupts these conventional limitations by enabling a direct, one-step amination process. By employing specific copper-containing compounds, particularly Copper(I) oxide (Cu2O), the invention achieves high conversion rates even with sterically demanding substrates. The process operates under elevated temperatures ranging from 130°C to 170°C and pressures between 35 to 85 bar, conditions that are well within the capabilities of standard industrial autoclaves. Crucially, the reaction utilizes ammonia, a commodity chemical, in large excess to drive the equilibrium towards the desired amine product. This direct approach bypasses the need for protecting groups entirely, collapsing what was previously a multi-step synthesis into a single, robust transformation. The result is a dramatic simplification of the process flow, which translates directly into reduced capital expenditure and lower operational complexity for manufacturers aiming to scale up production of these valuable intermediates.
Mechanistic Insights into Copper-Catalyzed Amination
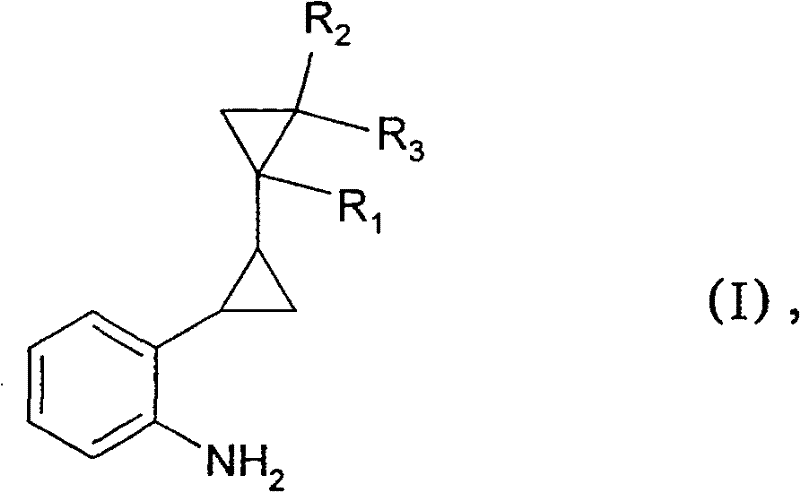
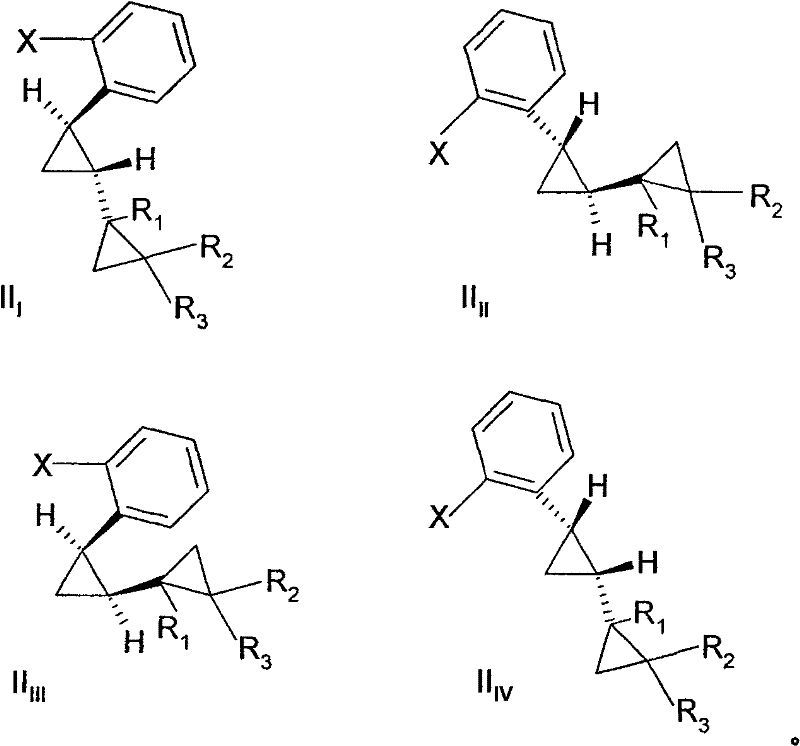
The success of this copper-catalyzed transformation lies in the unique ability of the catalyst to facilitate nucleophilic attack on the sterically hindered aryl halide. While the exact mechanistic cycle may involve oxidative addition and reductive elimination steps typical of transition metal catalysis, the specific choice of ligand-free or simple salt-based copper catalysts suggests a mechanism that tolerates significant steric bulk. The patent data indicates that the reaction kinetics are highly sensitive to the stereochemistry of the starting material. Specifically, the trans-isomers of the cyclopropyl-substituted halobenzenes exhibit a reaction rate approximately 1.7 times faster than their cis-isomers under identical conditions. This kinetic differentiation provides a powerful tool for process chemists to optimize selectivity. By controlling reaction time and temperature, it is possible to enrich the final product mixture with the desired trans-aniline isomer, which is often the biologically active configuration in downstream fungicide applications. Understanding this stereochemical nuance is critical for R&D teams aiming to maximize yield and minimize downstream purification costs.
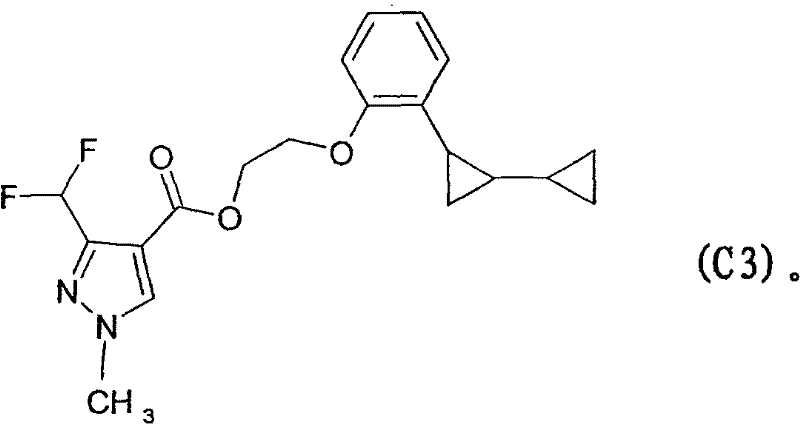
Furthermore, impurity control is a paramount concern in the synthesis of pharmaceutical and agrochemical intermediates. The patent explicitly addresses the formation of byproducts, particularly when ethylene glycol is employed as the solvent. Under the high-temperature reaction conditions, there is a competing pathway where the solvent itself acts as a nucleophile, leading to the formation of ether-linked impurities rather than the desired amine. For instance, in the synthesis of specific fungicides, this can lead to the formation of structurally related contaminants that are difficult to remove. The identification of these potential side reactions, such as the formation of Impurity C3 shown in the patent examples, allows manufacturers to implement rigorous quality control measures. By carefully selecting solvent systems or optimizing the molar ratio of ammonia to substrate, producers can suppress these side reactions, ensuring that the final API intermediate meets the stringent purity specifications required by global regulatory bodies.
How to Synthesize 2-Dicyclopropyl-2-yl-phenylamine Efficiently
The synthesis of high-purity ortho-alkyl anilines, such as 2-dicyclopropyl-2-yl-phenylamine, requires precise control over reaction parameters to ensure safety and reproducibility. The patented process provides a clear framework for executing this transformation on a commercial scale, emphasizing the importance of catalyst selection and pressure management. Operators must ensure that the copper catalyst is evenly dispersed and that the ammonia charge is sufficient to maintain the necessary partial pressure throughout the reaction duration. The following guide outlines the standardized operational procedure derived from the patent examples, serving as a foundational protocol for process development teams looking to implement this technology.
- Prepare the reaction mixture by combining the ortho-alkyl-substituted halobenzene (Formula II) with a catalytic amount of a copper-containing compound, preferably Copper(I) oxide (Cu2O).
- Introduce ammonia into the reactor, either as a liquid or gas, ensuring a significant molar excess (up to 200-fold) relative to the halobenzene substrate to drive the equilibrium forward.
- Heat the mixture to a temperature range of 130°C to 170°C under elevated pressure (35 to 85 bar) for 6 to 24 hours to achieve high conversion yields.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed technology offers substantial strategic advantages beyond mere technical feasibility. The shift from precious metal catalysts to base metal catalysts represents a significant opportunity for cost reduction in agrochemical manufacturing. Palladium prices are notoriously volatile and generally high, whereas copper salts are abundant and inexpensive commodities. By eliminating the need for palladium, companies can stabilize their raw material costs and reduce exposure to market fluctuations. Additionally, the removal of the protection-deprotection steps associated with older palladium methods drastically simplifies the supply chain. Fewer processing steps mean less equipment utilization time, lower energy consumption, and reduced labor costs, all of which contribute to a leaner and more competitive cost structure. This efficiency gain is particularly vital for high-volume products where marginal savings per kilogram translate into massive annual financial benefits.
- Cost Reduction in Manufacturing: The economic impact of switching to this copper-catalyzed route is profound, primarily driven by the elimination of expensive reagents and catalysts. Traditional methods relying on palladium require costly ligands and additional stoichiometric reagents for the protection and deprotection of the amine group. In contrast, the novel process utilizes catalytic amounts of inexpensive copper oxides and commodity ammonia. This fundamental change in the bill of materials significantly lowers the variable cost per unit. Furthermore, the reduction in synthetic steps minimizes solvent usage and waste disposal fees, which are often hidden but substantial costs in chemical production. The cumulative effect is a drastically simplified cost model that enhances profit margins without compromising product quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available raw materials. Copper catalysts and ammonia are produced on a massive global scale, ensuring consistent availability and reducing the risk of supply disruptions that can occur with specialized palladium complexes. The robustness of the reaction conditions also contributes to reliability; the process tolerates a range of operating parameters, making it less susceptible to minor variations in utility supply or equipment performance. This stability allows for more predictable production scheduling and shorter lead times for high-purity agrochemical intermediates. Manufacturers can confidently commit to delivery timelines, knowing that the underlying chemistry is robust and less prone to the failures often associated with sensitive noble metal catalysis.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process aligns well with modern green chemistry principles. The atom economy is improved by avoiding the generation of stoichiometric byproducts associated with protecting group chemistry. While the reaction requires elevated pressure, the equipment needed is standard in the fine chemical industry, facilitating easy scale-up from pilot plant to commercial production volumes. The reduction in waste streams, particularly heavy metal waste from palladium, simplifies effluent treatment and helps facilities meet increasingly strict environmental regulations. This compliance advantage reduces the administrative burden on EHS teams and minimizes the risk of regulatory penalties, ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed amination technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this process into existing manufacturing lines.
Q: Why is copper catalysis preferred over palladium for this specific amination?
A: While palladium catalysts are effective for deactivated aryl halides, they often require expensive ligands and multi-step protection/deprotection sequences (e.g., using benzophenone imine). The copper-catalyzed method described in CN101072746B allows for direct amination with ammonia in a single step, significantly reducing raw material costs and processing time.
Q: How does the process handle stereochemical selectivity?
A: The patent highlights that trans-isomers of the starting halobenzene react approximately 1.7 times faster than their cis-counterparts under optimized conditions. This kinetic difference allows manufacturers to optimize reaction times to enrich the desired trans-aniline product or separate isomers more efficiently.
Q: What are the critical impurity concerns with this synthesis route?
A: When using ethylene glycol as a solvent, there is a risk of forming ether byproducts where the glycol substitutes the halogen instead of ammonia. Careful purification or solvent selection is required to ensure the final fungicide intermediate meets strict purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Dicyclopropyl-2-yl-phenylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your final agrochemical formulations. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the identity and purity of every batch. Our facility is equipped to handle the high-pressure and high-temperature conditions required for this copper-catalyzed synthesis safely and efficiently, guaranteeing a stable supply of this essential fungicide intermediate.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our implementation of patent CN101072746B can deliver value to your organization. Let us be your partner in driving innovation and efficiency in the agrochemical sector.
