Advanced Copper-Catalyzed Amination for High-Purity Ortho-Alkyl Aniline Intermediates
Advanced Copper-Catalyzed Amination for High-Purity Ortho-Alkyl Aniline Intermediates
The chemical industry constantly seeks more efficient pathways to synthesize complex aromatic amines, which serve as critical building blocks for high-value agrochemicals and pharmaceuticals. Patent CN101072746B introduces a groundbreaking methodology for the preparation of ortho-alkyl-substituted primary phenylamines, specifically addressing the historical challenges associated with aminating deactivated benzene rings. This technology leverages a copper-catalyzed system to directly convert ortho-alkyl-substituted halobenzenes into their corresponding anilines using ammonia, bypassing the need for expensive palladium catalysts or multi-step protection strategies. For R&D directors and procurement specialists, this represents a significant opportunity to optimize the supply chain for key fungicide intermediates, such as 2-dicyclopropyl-2-yl-phenylamine, by adopting a more economically advantageous and operationally convenient synthetic route.
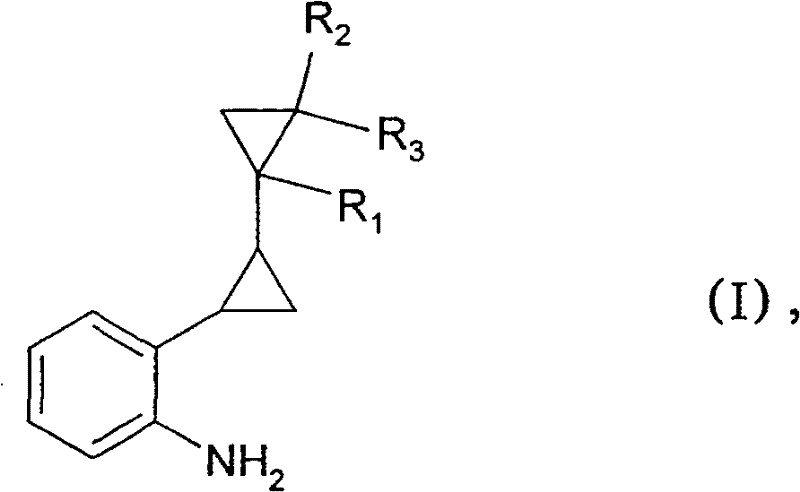
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of primary aryl amines from deactivated aryl halides, particularly those with ortho-alkyl substituents, has been a formidable challenge in organic synthesis. Conventional copper-catalyzed amination methods, while known for decades, typically suffer from low yields when applied to electron-rich or sterically hindered substrates like ortho-alkyl halobenzenes. Literature precedents often suggest that such reactions are only viable for electron-deficient nuclei, such as those activated by nitro groups, or for unsubstituted benzene cores. Furthermore, alternative modern approaches utilizing palladium catalysts, while effective, introduce significant economic and operational burdens. These palladium-mediated processes generally cannot produce primary amines directly; instead, they require a cumbersome two-step sequence involving reaction with benzophenone imine followed by hydrolysis with hydroxylamine hydrochloride. This multi-step nature not only increases the consumption of expensive precious metal catalysts but also extends production lead times and complicates waste management protocols.
The Novel Approach
The methodology disclosed in CN101072746B fundamentally shifts the paradigm by enabling the direct high-yield amination of these difficult substrates using a catalytic amount of a copper-containing compound. By reacting the ortho-alkyl-substituted halobenzene (Formula II) with ammonia under high pressure and temperature in the presence of a copper catalyst, the process achieves direct substitution of the halogen atom with an amino group. This novel approach eliminates the need for protecting groups and expensive palladium ligands, streamlining the synthesis into a single pot operation. The use of readily available copper salts, such as copper(I) oxide (Cu2O), in catalytic ratios ranging from 1:5 to 1:100 relative to the substrate drastically reduces the raw material cost profile. Additionally, the process demonstrates remarkable versatility, accommodating various stereoisomeric forms of the starting material and allowing for the tuning of reaction conditions to favor specific isomeric outcomes, thereby enhancing the overall utility for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Copper-Catalyzed Nucleophilic Aromatic Substitution
The core of this technological advancement lies in the activation of the carbon-halogen bond on the sterically hindered ortho-alkyl benzene ring by the copper catalyst. While the exact mechanistic pathway may involve a complex cycle of oxidative addition and reductive elimination typical of transition metal catalysis, the practical outcome is the facilitation of nucleophilic attack by ammonia on the aryl core. Unlike traditional Ullmann-type condensations which often require stoichiometric copper, this process operates efficiently with catalytic loads, suggesting a highly active catalytic species is generated in situ, likely involving Cu(I)/Cu(III) redox cycles stabilized by the high-temperature conditions and the solvent environment. The reaction conditions, specifically temperatures between 130°C and 170°C and pressures up to 150 bar, provide the necessary energy to overcome the activation barrier imposed by the ortho-alkyl groups, which typically deactivate the ring towards nucleophilic attack.
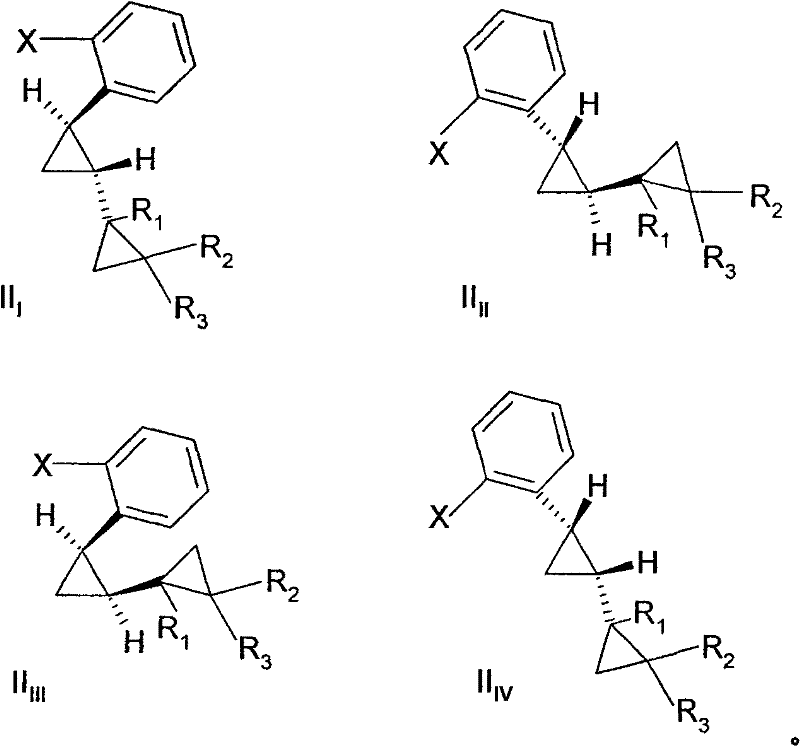
Impurity control is another critical aspect of this mechanism, particularly concerning the stereochemical integrity of the product. The patent data reveals that the reaction kinetics are stereoselective; for instance, the trans-isomer of the starting halobenzene reacts significantly faster (approximately 1.7 times) than the cis-isomer under identical conditions. This kinetic differentiation allows process chemists to manipulate the reaction time to enrich the final product mixture with the desired trans-aniline isomer, simplifying downstream purification. Furthermore, the choice of solvent plays a pivotal role in impurity profiling. While ethylene glycol is a preferred solvent for its ability to solubilize reactants at high temperatures, there is a competing pathway where the solvent itself acts as a nucleophile, leading to ether-linked byproducts. Understanding this competitive mechanism is vital for R&D teams to optimize the ammonia-to-substrate ratio, ensuring that ammonia outcompetes the solvent for the electrophilic carbon center, thus maintaining high purity standards required for reliable agrochemical intermediate supplier status.
How to Synthesize 2-Dicyclopropyl-2-yl-phenylamine Efficiently
To implement this robust synthetic route for the production of valuable aniline intermediates, operators must adhere to strict high-pressure safety protocols and precise stoichiometric controls. The process involves charging an autoclave with the specific ortho-alkyl halobenzene, a large excess of ammonia to drive the equilibrium forward, a catalytic quantity of copper(I) oxide, and ethylene glycol. The detailed standardized synthesis steps, including specific workup procedures and purification techniques to remove copper residues and solvent byproducts, are outlined below to ensure reproducibility and safety in a GMP environment.
- Charge a high-pressure autoclave with the ortho-alkyl substituted halobenzene (Formula II), liquid or gaseous ammonia (80-120 fold excess), a catalytic amount of copper(I) oxide (Cu2O), and ethylene glycol as the solvent.
- Heat the reaction mixture to a temperature range of 130°C to 170°C under a pressure of 35 to 85 bar for a duration of 6 to 24 hours to facilitate the nucleophilic substitution.
- After cooling, evaporate the excess ammonia, extract the residue with ethyl acetate, wash the organic phase with water, dry over sodium sulfate, and purify via chromatography to isolate the target aniline.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed technology offers substantial strategic benefits beyond mere technical feasibility. The shift from palladium-based systems to copper catalysis represents a fundamental decoupling from the volatile precious metal market, stabilizing raw material costs and reducing exposure to supply disruptions associated with rare earth and platinum group metals. The elimination of the second synthetic step (deprotection) inherently reduces the total processing time and labor costs, leading to a drastically simplified manufacturing workflow. This efficiency gain translates directly into improved throughput capacity, allowing manufacturers to respond more agilely to market demand fluctuations without the need for significant capital expenditure on additional reactor trains.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts and benzophenone imine reagents with inexpensive copper salts and ammonia results in a profound reduction in the bill of materials. Since the copper catalyst is used in catalytic amounts rather than stoichiometric quantities, the consumption of metal per kilogram of product is minimized, significantly lowering the cost of goods sold. Furthermore, the single-step nature of the reaction reduces utility consumption (steam, cooling water) and solvent usage compared to multi-step alternatives, contributing to substantial cost savings in the overall production budget.
- Enhanced Supply Chain Reliability: The raw materials required for this process, including ortho-alkyl halobenzenes, ammonia, and copper oxides, are commodity chemicals with well-established global supply chains. This abundance ensures high supply continuity and reduces the risk of production stoppages due to raw material shortages. By avoiding specialized ligands or sensitive organometallic reagents that often have long lead times and limited supplier bases, procurement teams can secure a more resilient and reliable supply chain for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is designed for scalability, utilizing standard high-pressure autoclave technology common in fine chemical manufacturing. The reduction in synthetic steps inherently lowers the E-factor (mass of waste per mass of product), as fewer purification stages and solvent exchanges are required. Additionally, the avoidance of heavy palladium residues simplifies wastewater treatment and metal recovery processes, aiding facilities in meeting stringent environmental regulations and sustainability goals while facilitating the commercial scale-up of complex polymer additives or agrochemical precursors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed amination technology. These insights are derived directly from the experimental data and embodiments provided in the patent literature, offering clarity on process optimization and quality control measures.
Q: Why is copper catalysis preferred over palladium for this specific amination?
A: While palladium catalysts are effective for deactivated halobenzenes, they typically require expensive ligands and a two-step process involving benzophenone imine protection and deprotection. The copper-catalyzed method described in CN101072746B allows for direct amination with ammonia in a single step, significantly reducing raw material costs and processing complexity.
Q: How does the process control stereoisomer purity in the final product?
A: The patent data indicates that the trans-isomer of the starting halobenzene reacts approximately 1.7 times faster than the cis-isomer under optimized conditions. By selecting appropriate reaction times and temperatures, manufacturers can enrich the final product with the desired trans-aniline stereoisomer, simplifying downstream purification.
Q: What are the critical impurity concerns when using ethylene glycol as a solvent?
A: When ethylene glycol is used as the solvent, there is a risk of forming ether-linked byproducts where the glycol substitutes the halogen instead of ammonia. However, these impurities can be managed through careful control of ammonia excess and reaction stoichiometry, ensuring the final fungicide intermediate meets stringent purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Dicyclopropyl-2-yl-phenylamine Supplier
As the global demand for advanced agrochemical and pharmaceutical intermediates continues to rise, partnering with a technically proficient CDMO is essential for maintaining competitive advantage. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our commitment to quality is underpinned by rigorous QC labs and stringent purity specifications, guaranteeing that every batch of ortho-alkyl aniline meets the exacting standards required for downstream fungicide synthesis.
We invite you to leverage our technical expertise to optimize your supply chain and reduce manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced copper-catalyzed processes can enhance your product portfolio and operational efficiency.
