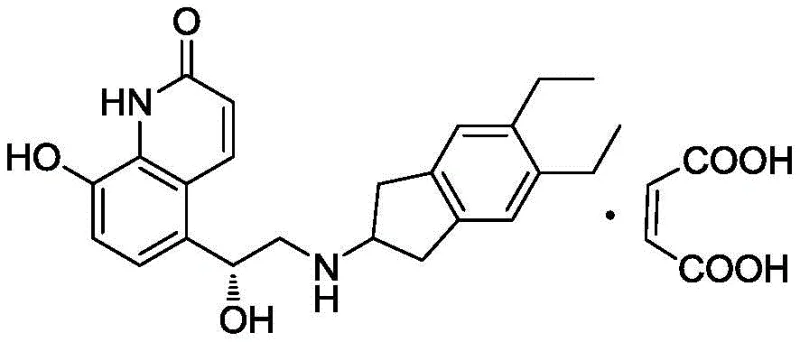
Optimizing Indacaterol Maleate Production: A Technical Breakthrough in Impurity Control
The respiratory pharmaceutical market continues to demand highly efficient bronchodilators, with Indacaterol Maleate standing out as a pivotal long-acting beta-agonist for treating Chronic Obstructive Pulmonary Disease (COPD) and asthma. However, the commercial viability of this Active Pharmaceutical Ingredient (API) hinges critically on the ability to synthesize it with exceptional purity, specifically regarding the control of structurally related impurities. Patent CN108101841B introduces a transformative approach to this challenge, detailing a refined catalytic hydrogenation process that effectively mitigates the formation of "Impurity A," a persistent byproduct in conventional synthesis routes. By leveraging a diluted hydrogen source rather than pure hydrogen gas, this methodology offers a robust solution for manufacturers seeking to enhance yield and simplify downstream purification. For a reliable API intermediate supplier, mastering such nuanced reaction conditions is paramount to delivering high-purity materials that meet stringent global regulatory requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for Indacaterol typically involve the coupling of a quinolinone epoxide with an indanamine derivative, followed by a crucial deprotection step to reveal the active phenolic hydroxyl group. In many prior art methods, such as those disclosed in US6878721, this deprotection is achieved through catalytic hydrogenation using pure hydrogen gas over a palladium catalyst. While effective at removing benzyl protecting groups, this aggressive condition often lacks the necessary selectivity. The conjugated double bond within the quinolinone ring system is susceptible to reduction under these conditions, leading to the formation of Impurity A, where the alkene is saturated. This side reaction is particularly problematic because Impurity A possesses physical and chemical properties very similar to the target product, rendering it notoriously difficult to separate via standard purification techniques like column chromatography or recrystallization. Consequently, batches produced via these conventional routes often suffer from lower yields and require extensive, costly purification efforts to meet pharmacopeial standards.
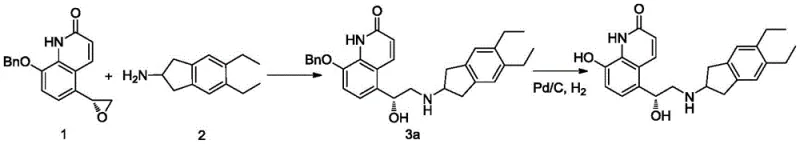
The Novel Approach
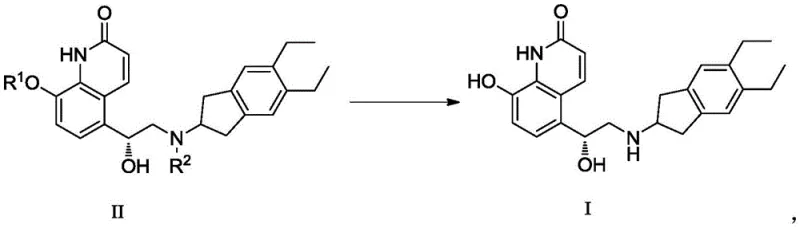
The innovation presented in patent CN108101841B addresses this selectivity issue head-on by modifying the thermodynamic and kinetic environment of the hydrogenation reaction. Instead of utilizing 100% hydrogen, the process employs a hydrogen-containing mixed gas where the hydrogen content is strictly controlled to be not higher than 90% (V/V), with a preferred range of 40-80% (V/V). This dilution with an inert gas, such as nitrogen or argon, effectively lowers the partial pressure of hydrogen available at the catalyst surface. This subtle yet powerful adjustment creates a kinetic window where the hydrogenolysis of the benzyl-oxygen bond proceeds efficiently, while the reduction of the electron-deficient quinolinone double bond is significantly suppressed. By tuning the gas composition, manufacturers can achieve a reaction profile that favors the desired deprotection pathway, drastically reducing the generation of Impurity A to levels as low as 0.06%, thereby simplifying the isolation of the final API salt.
Mechanistic Insights into Controlled Catalytic Hydrogenation
The core mechanism driving this improvement lies in the competitive adsorption and reaction rates on the palladium catalyst surface. In a standard hydrogenation environment with high hydrogen partial pressure, the catalyst is saturated with atomic hydrogen, promoting the rapid addition of hydrogen across any accessible unsaturated bonds, including the sensitive C=C double bond of the quinolinone ring. By introducing a diluted hydrogen stream, the concentration of adsorbed hydrogen species is reduced. The cleavage of the benzylic C-O bond (hydrogenolysis) generally has a different activation energy and mechanistic requirement compared to the simple addition of hydrogen across a conjugated alkene. The diluted environment appears to selectively retard the rate of alkene reduction more significantly than it retards the debenzylation process. Furthermore, the patent data indicates that this effect is robust across different catalyst loadings (1-20% by mass) and types (Pd/C, Pd(OH)2), suggesting that the gas composition is the primary driver of selectivity. This mechanistic understanding allows for precise process control, ensuring that even on a large commercial scale, the reaction remains within the "safe zone" where impurity formation is minimized.
Additionally, the process incorporates specific solvent systems and temperature controls to further refine the outcome. The use of protic solvents like methanol or ethanol, potentially mixed with esters or aromatic hydrocarbons, facilitates the solubility of the intermediate while maintaining a favorable environment for the catalytic cycle. Operating at moderate temperatures (10-30°C) prevents thermal acceleration of side reactions. The combination of diluted hydrogen, optimized solvent choice, and controlled temperature creates a multi-faceted barrier against impurity formation. This holistic approach to reaction engineering ensures that the final crude product is of sufficiently high quality to undergo direct salt formation with maleic acid, bypassing the need for complex intermediate purification steps that would otherwise erode overall process efficiency and increase production costs.
How to Synthesize Indacaterol Maleate Efficiently
The synthesis of high-purity Indacaterol Maleate begins with the preparation of the protected intermediate, typically involving the ring-opening of a quinolinone epoxide with a protected indanamine. Once the intermediate (Formula II) is secured, the critical deprotection step is executed using the patented diluted hydrogen protocol. The detailed standardized synthesis steps, including specific molar ratios, stirring rates, and work-up procedures, are outlined in the guide below to ensure reproducibility and compliance with the patent's specifications.
- Prepare the protected intermediate (Formula II) via epoxy ring-opening reaction using appropriate solvents like DMF or DMSO.
- Conduct catalytic hydrogenation using a metal catalyst (e.g., 5% Pd/C) and a diluted hydrogen source (40-80% H2 in inert gas) at 10-30°C.
- React the resulting free base with maleic acid in a solvent system (e.g., methanol/ethanol) to crystallize the final Indacaterol Maleate salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this refined synthesis route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the significant enhancement of process robustness and yield stability. By effectively suppressing the formation of hard-to-remove Impurity A, the process eliminates the need for resource-intensive purification methods such as preparative HPLC or multiple recrystallization cycles, which are often bottlenecks in API manufacturing. This streamlining translates directly into improved throughput and reduced cycle times, allowing suppliers to respond more agilely to market demand fluctuations without compromising on quality standards.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the drastic reduction in material loss associated with purification. In conventional processes, the struggle to separate Impurity A often leads to significant yield losses during chromatography or repeated crystallizations. By preventing the impurity from forming in the first place, the new method maximizes the conversion of valuable starting materials into the final API. Furthermore, the elimination of complex purification steps reduces the consumption of solvents, silica gel, and energy, leading to substantial operational cost savings. The ability to use standard, commercially available catalysts like 5% Pd/C without requiring exotic ligands or specialized equipment further keeps capital and operational expenditures low, ensuring a competitive pricing structure for the final product.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by processes that are sensitive to minor variations in raw material quality or reaction conditions. The diluted hydrogen method described in the patent demonstrates remarkable tolerance, with data showing consistent impurity control across different catalyst batches and loading percentages. This robustness minimizes the risk of batch failures or off-spec production runs, which can disrupt supply schedules. Additionally, the use of diluted hydrogen gas enhances operational safety by reducing the flammability risks associated with handling pure hydrogen under pressure, thereby lowering insurance costs and facilitating smoother regulatory inspections. A safer, more predictable process inherently leads to a more reliable supply chain for downstream pharmaceutical partners.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to multi-ton production often exacerbates impurity issues due to heat and mass transfer limitations. However, the kinetic control offered by gas composition is highly scalable, as gas-liquid mass transfer can be precisely managed in large-scale reactors. The reduction in solvent usage and the avoidance of silica-based chromatography significantly decrease the volume of hazardous waste generated per kilogram of product. This aligns with modern green chemistry principles and helps manufacturers meet increasingly stringent environmental regulations regarding solvent discharge and solid waste disposal. A cleaner process not only reduces disposal costs but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis method, derived from the specific experimental data and claims within the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: How does the patented method reduce Impurity A in Indacaterol synthesis?
A: The method utilizes a diluted hydrogen source (e.g., 70% H2 mixed with nitrogen) instead of pure hydrogen. This lowers the hydrogen partial pressure, kinetically favoring the removal of the benzyl protecting group while suppressing the over-reduction of the quinolinone double bond, which forms Impurity A.
Q: What catalysts are suitable for this deprotection reaction?
A: The patent specifies several effective metal catalysts, with a strong preference for Palladium on Carbon (Pd/C) ranging from 1% to 20% palladium content. Palladium hydroxide is also cited as a viable alternative for this specific transformation.
Q: Why is controlling Impurity A critical for commercial production?
A: Impurity A is structurally similar to the target molecule due to the saturation of the double bond, making it extremely difficult to remove via standard purification techniques like recrystallization or column chromatography. Minimizing its formation at the source is essential for meeting strict regulatory purity standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indacaterol Maleate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and state-of-the-art infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the delicate balance of the diluted hydrogenation process is maintained perfectly at every scale. We operate stringent purity specifications and utilize rigorous QC labs equipped with advanced analytical tools to verify that every batch of Indacaterol Maleate meets the lowest possible impurity profiles, specifically targeting Impurity A levels well below regulatory thresholds.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced manufacturing capability. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
