Advanced Purification Strategy for Indacaterol Maleate Intermediates Enhancing Commercial Scalability
Advanced Purification Strategy for Indacaterol Maleate Intermediates Enhancing Commercial Scalability
The pharmaceutical landscape for Chronic Obstructive Pulmonary Disease (COPD) treatments demands exceptionally high standards for active pharmaceutical ingredients, particularly for long-acting beta2-adrenergic agonists like Indacaterol Maleate. As detailed in patent CN111808021B, a significant technological breakthrough has been achieved in the preparation method of Indacaterol and its salts, addressing critical bottlenecks in purity and yield that have historically plagued manufacturers. This innovation centers on a novel purification strategy utilizing m-chlorobenzoic acid to isolate key intermediates, offering a robust alternative to traditional chromatographic or less efficient salting-out processes. For global procurement teams and R&D directors seeking a reliable API intermediate supplier, understanding this shift towards high-selectivity crystallization is vital for securing supply chains that prioritize both quality and cost-efficiency.
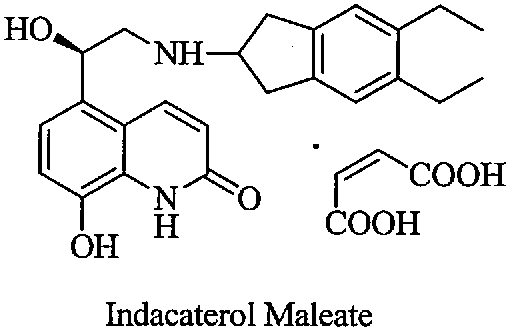
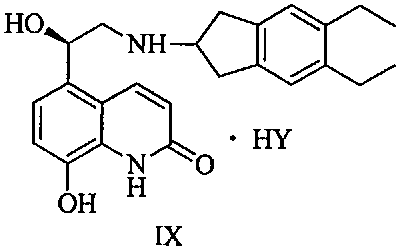
The structural complexity of Indacaterol Maleate, characterized by its specific stereochemistry and functional groups, necessitates precise synthetic control to ensure therapeutic efficacy. The patent outlines a comprehensive pathway that not only synthesizes the core structure but also rigorously controls the impurity profile through a specialized salt formation step. By transitioning from vague purification hopes to a defined crystalline solid of intermediate Formula II-1, this method sets a new benchmark for high-purity Indacaterol intermediate production. This report delves into the mechanistic advantages and commercial implications of adopting this refined synthesis route for commercial scale-up of complex respiratory drug intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Indacaterol has been hindered by the inherent lack of selectivity in the initial ring-opening substitution reaction. As illustrated in the prior art routes, reacting 8-phenylmethoxy-5-(R)-oxiranyl-1H-quinolin-2-one with 2-amino-5,6-diethylindan generates a chaotic mixture of products. This reaction suffers from poor regioselectivity and stereoselectivity, leading to the formation of significant amounts of isomeric substitution byproducts (Formula III), disubstituted impurities (Formula IV), and deethylated derivatives (Formula V). In many documented cases, the content of isomeric substitution byproducts can reach as high as 7.8%, while disubstituted byproducts account for approximately 12.4% of the crude product, creating a formidable purification challenge.
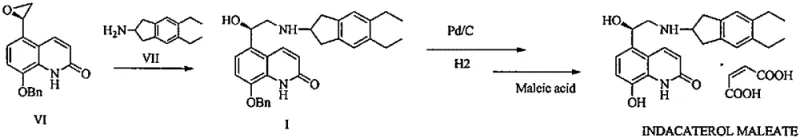
Furthermore, the purification of the resulting intermediate (Formula I) in conventional methods has relied heavily on inefficient techniques. Some prior patents disclose the use of column chromatography, a method that is notoriously difficult to scale industrially due to high solvent consumption, low throughput, and significant product loss. Alternative salifying crystallization processes using agents like benzoic acid or salicylic acid have shown marginal improvements but still suffer from suboptimal yields, often hovering around 60%, with purities struggling to exceed 96% even after multiple recrystallizations. These limitations result in substantial material waste and increased production costs, making cost reduction in pharmaceutical manufacturing difficult to achieve without a fundamental process redesign.
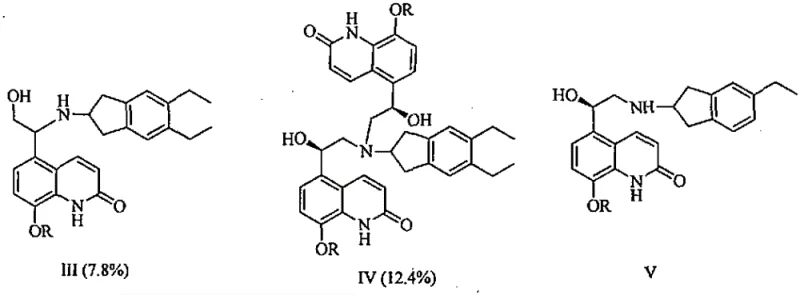
The Novel Approach
The innovative method disclosed in CN111808021B introduces a transformative step by treating the crude reaction mixture directly with m-chlorobenzoic acid. This specific acid acts as a highly selective resolving agent, reacting with the mixture containing Formula I and its impurities to form a distinct crystalline salt, identified as Formula II-1. This approach leverages the unique solubility and crystallization properties of the m-chlorobenzoate salt to effectively exclude the troublesome isomeric and disubstituted impurities, which remain in the mother liquor. The result is a dramatic improvement in process metrics, with the yield of the ring-opening substitution and purification sequence reaching approximately 75%, a significant leap from the historical 60% baseline.
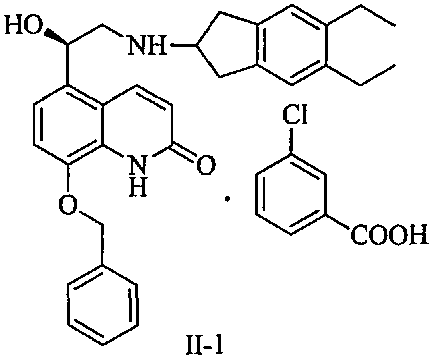
Beyond yield improvements, the purity profile of the isolated intermediate is exceptionally clean, achieving HPLC purity levels of 99.8% with single impurities controlled to less than 0.05%. Crucially for chiral drugs, the content of the unwanted S-isomer is reduced to below 0.1%, ensuring the final API meets stringent regulatory requirements for enantiomeric excess. This method eliminates the need for column chromatography entirely, replacing it with a straightforward filtration and washing protocol that is inherently safer and more scalable. For supply chain managers, this translates to a more predictable and robust manufacturing process that minimizes the risk of batch failures and ensures consistent availability of high-purity Indacaterol intermediates.
Mechanistic Insights into Selective Salt Formation and Deprotection
The success of this novel preparation method lies in the precise thermodynamic control exerted during the salt formation step. When m-chlorobenzoic acid is introduced to the reaction mixture in solvents such as ethanol or isopropanol at elevated temperatures (50-90°C), it selectively complexes with the desired (R)-enantiomer of the intermediate. The resulting crystal lattice of Formula II-1 is energetically favorable compared to the salts of the impurities or the S-enantiomer, driving the equilibrium towards the precipitation of the pure product. This phenomenon, often referred to as chiral resolution via diastereomeric salt formation, is optimized here by the specific electronic and steric properties of the meta-chloro substituent on the benzoic acid, which appears to enhance the differentiation between the target molecule and its structural analogs.
Following the isolation of the pure salt, the process proceeds to the deprotection phase, where the benzyl protecting group (R) is removed. This is typically achieved through catalytic hydrogenation using palladium on carbon (Pd/C) in alcoholic solvents like methanol or ethanol. The mild conditions (30-60°C) ensure that the sensitive quinolinone core remains intact while the benzyl ether is cleanly cleaved to reveal the phenolic hydroxyl group. The final step involves the conversion of the free base into the therapeutically active maleate salt. By neutralizing the free base or treating it directly with maleic acid, the final Indacaterol Maleate is obtained with high crystallinity and stability. This seamless transition from protected intermediate to final salt underscores the chemical elegance of the route.
How to Synthesize Indacaterol Intermediate Efficiently
To implement this advanced synthesis route effectively, manufacturers must adhere to specific operational parameters regarding solvent choice, temperature control, and stoichiometry. The process begins with the substitution reaction in n-butanol at temperatures between 80-120°C, followed immediately by the critical purification step. The detailed standardized synthesis steps, including specific reagent ratios and crystallization timelines, are outlined below to guide process engineers in replicating these high-yield results.
- Perform a ring-opening substitution reaction between 8-phenylmethoxy-5-(R)-oxiranyl-1H-quinolin-2-one and 2-amino-5,6-diethylindan to obtain a crude mixture.
- Treat the crude mixture with m-chlorobenzoic acid in a suitable solvent to selectively form the m-chlorobenzoate salt of the desired intermediate.
- Crystallize and isolate the pure salt, followed by catalytic hydrogenation to remove the protecting group and final salt formation with maleic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers compelling strategic advantages that extend beyond simple chemistry. The elimination of column chromatography represents a massive reduction in operational complexity and solvent usage, directly impacting the cost of goods sold (COGS). By shifting to a crystallization-based purification, the process becomes amenable to large-scale batch processing in standard reactors, removing the bottleneck of slow, labor-intensive column packing and elution. This operational simplification leads to substantial cost savings in terms of both labor and materials, making the production of Indacaterol intermediates significantly more economically viable.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the significant increase in yield from approximately 60% in prior art methods to roughly 75% in this new method. This improvement means that for every ton of starting material purchased, manufacturers obtain substantially more usable intermediate, drastically reducing the effective cost per kilogram of the final API. Furthermore, the removal of expensive silica gel and the vast quantities of solvents required for chromatography results in lower waste disposal costs and reduced raw material expenditure. The use of readily available reagents like m-chlorobenzoic acid, rather than specialized or exotic resolving agents, further stabilizes the supply chain against price volatility.
- Enhanced Supply Chain Reliability: Reliability in the pharmaceutical supply chain is often compromised by processes that are sensitive to minor variations. The robust crystallization protocol described in the patent offers a high degree of reproducibility, ensuring that batch-to-batch variability is minimized. The ability to consistently achieve 99.8% purity reduces the need for re-processing or re-crystallization, which can delay shipments and disrupt inventory planning. By securing a manufacturing partner capable of executing this stable process, buyers can mitigate the risk of supply interruptions caused by failed quality control tests, ensuring a steady flow of materials for downstream formulation.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns perfectly with green chemistry principles. The avoidance of column chromatography significantly reduces the volume of hazardous organic waste generated, simplifying compliance with increasingly strict environmental regulations. The process utilizes common solvents like ethanol and methanol, which are easier to recover and recycle compared to the complex solvent mixtures often found in chromatographic fractions. This ease of scale-up ensures that production can be ramped from pilot plant quantities to multi-ton commercial volumes without the need for specialized equipment, facilitating rapid response to market demand surges.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Indacaterol synthesis method. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on performance metrics and operational feasibility.
Q: What is the primary advantage of using m-chlorobenzoic acid in Indacaterol synthesis?
A: The use of m-chlorobenzoic acid allows for the selective crystallization of the desired intermediate salt (Formula II-1), effectively removing isomeric and disubstituted impurities that are difficult to separate via traditional methods, thereby achieving purity levels up to 99.8%.
Q: How does this new method improve upon prior art purification techniques?
A: Unlike prior art methods that rely on low-yield column chromatography or less efficient salifying agents like benzoic acid (yielding ~60%), this novel approach utilizes a specific crystallization process that boosts yield to approximately 75% while significantly reducing S-isomer content to below 0.1%.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process is designed for industrialization by eliminating complex chromatographic steps and utilizing robust crystallization and catalytic hydrogenation techniques, which ensures consistent quality and operational simplicity essential for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indacaterol Maleate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated purification techniques described in CN111808021B can be seamlessly integrated into your supply chain. We maintain stringent purity specifications and operate rigorous QC labs equipped to verify the low S-isomer content and high HPLC purity essential for respiratory APIs, guaranteeing that every batch meets global regulatory standards.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits tailored to your volume needs. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to delivering high-quality Indacaterol Maleate intermediates that drive your business forward.
