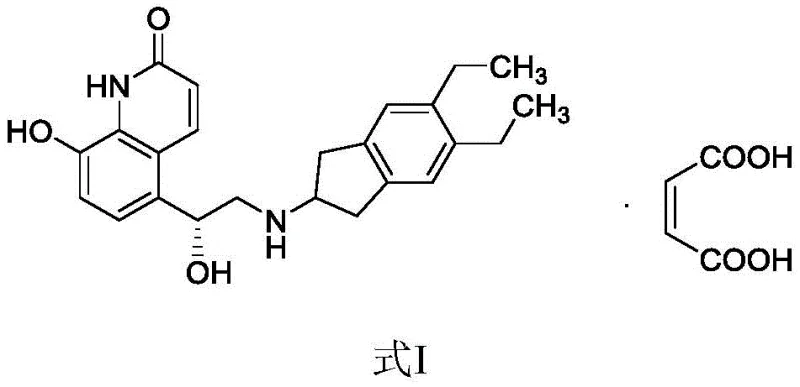
Advanced Base-Free Alkylation Strategy for Commercial Scale-Up of Indacaterol Maleate
The pharmaceutical landscape for Chronic Obstructive Pulmonary Disease (COPD) treatments demands rigorous standards for long-acting beta-agonists (LABAs), particularly regarding optical purity and impurity control. Patent CN108250140B introduces a transformative preparation method for Indacaterol Maleate, a potent bronchodilator, addressing critical bottlenecks in traditional synthetic routes. This innovation pivots away from complex epoxide-opening strategies toward a streamlined, base-free alkylation protocol that significantly enhances process safety and environmental compatibility. By eliminating the need for alkaline solutions in the initial coupling step, the process not only simplifies operational procedures but also drastically reduces the formation of stubborn byproducts like regioisomers and dimers. The method achieves a remarkable isolated yield of 93% for the key intermediate, with HPLC purity exceeding 97.8%, setting a new benchmark for industrial feasibility.
For procurement and supply chain leaders, this technological leap represents a substantial opportunity for cost reduction in respiratory API manufacturing. The ability to secure high-purity intermediates through a shorter, safer synthetic sequence directly correlates to reduced production lead times and lower waste disposal costs. As a reliable pharmaceutical intermediates supplier, understanding these mechanistic advantages is crucial for evaluating long-term vendor partnerships. The following analysis dissects the technical superiority of this route, offering deep insights into how it resolves historical challenges in scaling complex quinolinone derivatives for commercial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Indacaterol has relied heavily on the condensation of (R)-5-oxiranyl-8-benzyloxy-quinolin-2(1H)-one with 5,6-diethyl-2,3-dihydro-1H-indene-2-amine, as disclosed in various prior art documents such as WO200476422 and US6878721. While chemically viable, this epoxide-opening pathway is plagued by significant selectivity issues. The nucleophilic attack on the epoxide ring often lacks precision, leading to the formation of regioisomeric impurities and disubstituted dimers that can account for approximately 20% of the crude reaction mixture.
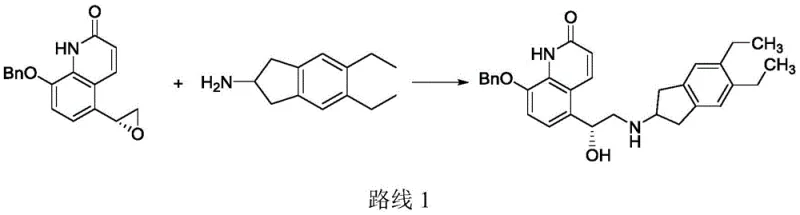
These impurities are notoriously difficult to remove, requiring extensive chromatographic purification or multiple recrystallization steps that erode overall yield and increase solvent consumption. Furthermore, alternative methods reported in WO2013132514 attempt to mitigate these issues by employing mixed base systems (e.g., triethylamine/NaI or K2CO3/NaI) in organic solvents. However, even with optimized base combinations, yields typically plateau between 76% and 84%. The reliance on inorganic bases introduces additional unit operations for aqueous workups and wastewater treatment, complicating the commercial scale-up of complex pharmaceutical intermediates and increasing the environmental footprint of the manufacturing process.
The Novel Approach
In stark contrast, the methodology outlined in CN108250140B employs a direct alkylation strategy that bypasses the instability of epoxide intermediates entirely. The core innovation involves reacting a bromo-ketone derivative (Formula IIB-1) directly with the amine component (Formula IIA) in a high-boiling alcohol solvent, specifically n-butanol, at temperatures ranging from 100°C to 110°C. Crucially, this reaction proceeds efficiently without the addition of any external alkali solution or inorganic base. The thermal energy provided by the refluxing n-butanol is sufficient to drive the nucleophilic substitution to completion, resulting in a clean conversion to the ketone intermediate (Formula II-1).
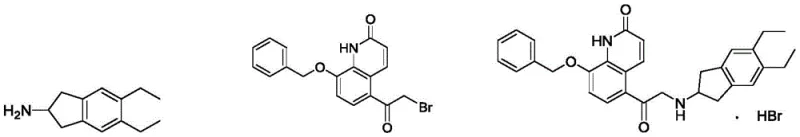
This base-free condition is a game-changer for process chemistry, as it eliminates the generation of inorganic salts that often complicate filtration and drying steps. The result is a robust process that delivers the coupled product in a superior 93% yield with an HPLC purity of 97.82%. By avoiding the harsh conditions and selectivity pitfalls of epoxide ring-opening, this novel approach ensures a much cleaner impurity profile from the very first step, laying a solid foundation for the subsequent stereoselective transformations required to produce the final active pharmaceutical ingredient.
Mechanistic Insights into Base-Free N-Alkylation and Asymmetric Reduction
The success of this synthetic route hinges on the careful selection of reaction parameters that favor the desired nucleophilic substitution while suppressing side reactions. In the coupling step, the use of n-butanol serves a dual purpose: it acts as a polar protic solvent capable of dissolving both the organic halide and the amine hydrochloride salt (after free basing), and its boiling point (117°C) allows the reaction to be conducted at 100-110°C. At this elevated temperature, the kinetic energy of the molecules overcomes the activation barrier for the SN2-type displacement of the bromide leaving group by the secondary amine. The absence of a strong base prevents the potential deprotonation of other acidic sites on the quinolinone ring, which could otherwise lead to O-alkylation or polymerization byproducts.
Following the coupling, the establishment of the critical chiral center is achieved through an asymmetric reduction using the Corey-Bakshi-Shibata (CBS) protocol. The ketone intermediate is treated with a chiral oxazaborolidine catalyst, preferably (R)-2-methyl-CBS-oxazaborolidine, in the presence of a borane reducing agent such as borane-tetrahydrofuran complex. This reaction is typically conducted at low temperatures (0-5°C) under nitrogen protection to maintain catalyst integrity and stereocontrol. The CBS catalyst coordinates with the carbonyl oxygen and the borane hydride, creating a rigid transition state that directs the hydride delivery specifically to the re-face (or si-face, depending on catalyst configuration) of the ketone. This precise stereochemical control ensures the formation of the (R)-hydroxy ethyl moiety with high enantiomeric excess, which is essential for the potent beta-2 adrenergic activity of the final drug substance.
How to Synthesize Indacaterol Maleate Efficiently
The synthesis of Indacaterol Maleate via this patented route involves a logical sequence of coupling, reduction, deprotection, and salt formation. The process begins with the preparation of the free amine from its hydrochloride salt, followed by the thermal coupling in n-butanol. Subsequent steps involve the stereoselective reduction of the ketone, catalytic hydrogenation to remove the benzyl protecting group, and final crystallization with maleic acid. Each step is optimized for high yield and ease of isolation, making it highly suitable for multi-kilogram production.
- Coupling Reaction: React 5,6-diethyl-indan-2-amine (Formula IIA) with 2-bromo-1-(8-benzyloxy-2-oxo-1,2-dihydroquinolin-5-yl)ethanone (Formula IIB-1) in n-butanol at 100-110°C without added base to obtain the ketone intermediate.
- Asymmetric Reduction: Reduce the ketone intermediate using a chiral CBS catalyst (e.g., (R)-2-methyl-CBS-oxazaborolidine) and borane-THF complex at 0-5°C to establish the (R)-chiral center.
- Deprotection and Salt Formation: Remove the benzyl protecting group via Pd/C catalytic hydrogenation, followed by crystallization with maleic acid in methanol to yield high-purity Indacaterol Maleate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this base-free alkylation technology offers tangible benefits that extend beyond mere chemical elegance. The elimination of inorganic bases and the associated aqueous workup steps translates directly into simplified processing infrastructure. Without the need for large-scale washing tanks to remove salts like sodium bromide or potassium carbonate, manufacturers can reduce water consumption and wastewater treatment loads significantly. This streamlining of the post-reaction processing not only accelerates the batch cycle time but also lowers the operational expenditure related to utility consumption and waste disposal compliance.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the dramatic improvement in yield and purity. Achieving a 93% yield in the coupling step, compared to the 76-84% typical of base-catalyzed methods, means that less raw material is required to produce the same amount of intermediate. Furthermore, the high purity (>97.8%) reduces the burden on downstream purification, minimizing product loss during recrystallization. The removal of expensive inorganic bases and the simplification of the workup procedure also contribute to substantial cost savings in terms of reagent procurement and labor hours.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain continuity by reducing the risk of batch failures due to impurity excursions. Traditional methods prone to generating 20% impurities often require reprocessing or result in scrapped batches, leading to unpredictable delivery schedules. By contrast, the predictable and clean nature of the base-free alkylation ensures consistent output quality. Additionally, the reliance on common, commodity solvents like n-butanol and methanol, rather than specialized or hazardous reagents, mitigates the risk of raw material shortages and price volatility.
- Scalability and Environmental Compliance: From a scale-up perspective, the exothermic profile of the base-free reaction is easier to manage than the potentially vigorous epoxide-opening reactions. The absence of solid inorganic bases eliminates issues related to mixing efficiency and heat transfer in large reactors. Environmentally, the process aligns with green chemistry principles by reducing the E-factor (mass of waste per mass of product). The simplified workflow generates less hazardous waste, facilitating easier compliance with increasingly stringent environmental regulations governing pharmaceutical manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing clarity on critical process parameters and quality attributes.
Q: How does the new base-free alkylation method improve impurity profiles compared to prior art?
A: Conventional epoxide-opening routes often generate up to 20% regioisomers and dimers. The novel base-free alkylation described in CN108250140B operates at 100-110°C in n-butanol, suppressing side reactions and achieving HPLC purity greater than 97.8% for the intermediate.
Q: What is the role of the CBS catalyst in this synthesis?
A: The chiral oxazaborolidine catalyst (specifically (R)-2-methyl-CBS) directs the stereoselective reduction of the ketone intermediate by borane. This ensures the formation of the critical (R)-hydroxy ethyl moiety required for beta-2 adrenoceptor agonist activity.
Q: Why is n-butanol preferred as the solvent for the coupling step?
A: n-Butanol serves as both a solvent and a reaction medium that supports the necessary temperature range (100-110°C) for the nucleophilic substitution to proceed efficiently without the need for external inorganic bases, thereby simplifying downstream processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indacaterol Maleate Supplier
The technical advancements detailed in CN108250140B underscore the complexity and precision required to manufacture high-quality respiratory APIs. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production necessary to bring such sophisticated chemistry to life. Our facility is equipped with rigorous QC labs and stringent purity specifications that ensure every batch of Indacaterol intermediate meets the exacting standards required for global regulatory filings. We understand that consistency in chiral purity and impurity profiles is non-negotiable for your final drug product.
We invite you to leverage our technical expertise to optimize your supply chain for this critical COPD medication. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your long-term commercial goals with reliability and efficiency.
