Revolutionizing Ramelteon Intermediate Production via Safe One-Step Catalytic Hydrogenation
Revolutionizing Ramelteon Intermediate Production via Safe One-Step Catalytic Hydrogenation
The pharmaceutical industry is constantly seeking more efficient and safer pathways for the synthesis of complex active pharmaceutical ingredients (APIs) and their key precursors. A significant breakthrough in this domain is documented in patent CN111116531A, which discloses a novel preparation method for the Ramelteon intermediate, specifically 2-(1,6,7,8-tetrahydro-2H-indeno[5,4-b]furan-8-yl)ethylamine hydrochloride. This innovation addresses long-standing challenges in the synthetic route of this sleep disorder therapeutic by replacing harsh, multi-step protocols with a streamlined, one-step catalytic hydrogenation process. By utilizing advanced platinum or palladium-based catalytic systems under mild conditions, this method not only enhances reaction efficiency but also drastically improves the impurity profile of the final product. For global procurement teams and R&D directors, understanding this technological shift is crucial for securing a reliable supply chain of high-purity pharmaceutical intermediates that meet stringent regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this critical tricyclic intermediate has been plagued by significant technical hurdles that impact both yield and purity. Traditional methodologies predominantly relied on the use of Raney Nickel as a catalyst under alkaline conditions, a process known for its operational hazards and inconsistent outcomes. The primary defect of this legacy approach lies in the generation of substantial amounts of polymeric byproducts, specifically identified as dimeric Impurity A and trimeric Impurity B, which complicate downstream purification efforts. Furthermore, these conventional routes often suffer from incomplete conversion of the reaction intermediate (Intermediate III), leaving residual levels exceeding 0.1 percent, which can carry through to the final API. The structural similarity between Intermediate III and the target molecule makes separation exceptionally difficult, often requiring costly and time-consuming chromatographic techniques that are undesirable for large-scale manufacturing.
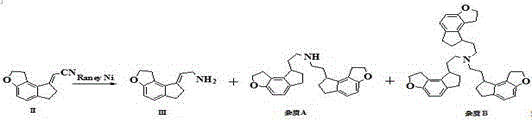
In addition to purity concerns, the reliance on Raney Nickel introduces severe safety risks due to its pyrophoric nature, necessitating specialized handling protocols and increasing the overall cost of compliance and insurance for manufacturing facilities. Previous attempts to optimize this pathway, such as the method described in WO2008062468, attempted to mitigate these issues by splitting the reduction into two distinct steps involving different catalysts. While this two-step approach improved the conversion of Intermediate III, it failed to eliminate the fundamental safety risks associated with explosive reagents and did not fully resolve the issue of polymerization impurities. Consequently, the industry has been in urgent need of a unified process that combines safety, efficiency, and superior impurity control into a single operational unit.
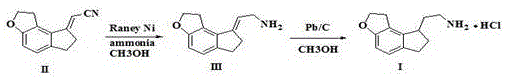
The Novel Approach
The methodology presented in patent CN111116531A represents a paradigm shift by consolidating the reduction of both the nitrile group and the olefinic double bond into a single, cohesive reaction step. This novel approach utilizes a sophisticated catalytic system comprising platinum oxide, platinum chloride, or palladium carbon, dispersed within a specific solvent matrix of chloroform and alcohol compounds. By carefully tuning the solvent ratio, typically maintaining a volume ratio of alcohol to chloroform between 1:1 and 10:1, the process achieves exceptional selectivity. This one-step hydrogenation effectively bypasses the accumulation of Intermediate III, reducing its content to negligible levels of less than or equal to 0.05 percent. The result is a dramatic simplification of the workflow, where the starting material, 2-(1,2,6,7-tetrahydro-8H-indeno[5,4-b]furan-8-ylidene)acetonitrile, is directly converted to the target amine hydrochloride with high fidelity.
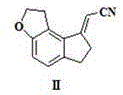
Beyond the chemical elegance, this new route offers profound operational advantages by operating under remarkably mild conditions, with reaction temperatures ranging from 0 to 80 degrees Celsius and hydrogen pressures as low as 0.3 to 0.5 MPa. These gentle parameters stand in stark contrast to the harsh environments required by older methods, significantly reducing energy consumption and equipment stress. The elimination of strong acids and the avoidance of unstable reagents like lithium aluminum hydride further enhance the safety profile, making this process ideally suited for commercial scale-up. For supply chain managers, this translates to a more robust manufacturing capability with reduced risk of batch failures due to thermal runaways or catalyst deactivation, ensuring a consistent flow of high-quality intermediates for the production of Ramelteon.
Mechanistic Insights into Platinum-Catalyzed Hydrogenation
The success of this one-step synthesis hinges on the unique electronic and steric properties of the chosen noble metal catalysts, particularly platinum oxide and palladium carbon, within the chloroform-alcohol solvent system. Mechanistically, the catalyst facilitates the simultaneous adsorption and reduction of the carbon-nitrogen triple bond of the nitrile group and the carbon-carbon double bond of the exocyclic alkene. The presence of chloroform in the solvent mixture is believed to play a critical role in modulating the catalyst surface activity, preventing the over-reduction or hydrogenolysis of sensitive functional groups while promoting the desired saturation. This precise control over the hydrogenation landscape is what allows the reaction to proceed cleanly to the saturated amine without stalling at the unsaturated amine stage (Intermediate III), which is the bottleneck in traditional routes. The kinetic favorability of this dual-reduction ensures that the concentration of Intermediate III remains transient and low, thereby kinetically suppressing its ability to act as a nucleophile in side reactions that lead to dimerization.
Furthermore, the suppression of polymeric impurities (Impurity A and B) is a direct consequence of the rapid and complete conversion of the reactive nitrile species. In slower, less efficient catalytic systems, the prolonged presence of partially reduced species increases the probability of intermolecular condensation reactions, leading to the formation of higher molecular weight oligomers. By driving the reaction to completion rapidly under optimized pressure and temperature, the novel process minimizes the residence time of these reactive intermediates. Additionally, the specific interaction between the tricyclic indeno-furan scaffold and the catalyst surface appears to favor the formation of the desired stereochemistry and prevent the aggregation of molecules that leads to trimerization. This mechanistic clarity provides R&D directors with the confidence that the process is not merely empirical but is grounded in sound chemical principles that ensure reproducibility and scalability across different batch sizes.
How to Synthesize 2-(1,6,7,8-tetrahydro-2H-indeno[5,4-b]furan-8-yl)ethylamine Hydrochloride Efficiently
Implementing this advanced synthesis route requires strict adherence to the optimized parameters defined in the patent to achieve the reported yields of over 96 percent and chemical purities exceeding 93 percent. The process begins with the dissolution of the starting nitrile in a methanol-chloroform mixture, followed by the addition of the catalyst under a controlled hydrogen atmosphere. The reaction is monitored to ensure complete consumption of the starting material, after which the catalyst is removed via filtration, and the product is isolated through concentration and crystallization using n-heptane. This straightforward workup procedure avoids the need for complex extractions or column chromatography, which are often cost-prohibitive at an industrial scale. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis protocol below.
- Dissolve the starting nitrile compound (II) in a mixed solvent system comprising methanol and chloroform to ensure optimal solubility and reaction kinetics.
- Introduce a heterogeneous catalyst such as platinum oxide or palladium carbon (5-20 wt%) into the reaction mixture under an inert atmosphere.
- Conduct hydrogenation at mild temperatures (30-40°C) and low pressure (0.3-0.5 MPa) until complete conversion is achieved, followed by filtration and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel one-step hydrogenation technology offers tangible strategic benefits that extend beyond simple chemical yield. The primary advantage lies in the significant reduction of manufacturing complexity, which directly correlates to lower operational expenditures and reduced lead times. By collapsing a multi-step sequence into a single reactor operation, manufacturers can drastically reduce the labor hours, utility consumption, and equipment occupancy time required per batch. This efficiency gain allows for a more agile response to market demand fluctuations, ensuring that the supply of this critical sleep aid intermediate remains stable even during periods of high global demand. Furthermore, the simplified process flow reduces the number of potential failure points, enhancing the overall reliability of the supply chain.
- Cost Reduction in Manufacturing: The elimination of hazardous and expensive reagents such as Raney Nickel and Lithium Aluminum Hydride results in substantial raw material cost savings. Additionally, the removal of the intermediate isolation and purification steps significantly lowers solvent consumption and waste disposal costs. The use of standard hydrogenation equipment rather than specialized high-pressure or cryogenic reactors further reduces capital expenditure requirements for production facilities. These cumulative efficiencies translate into a more competitive pricing structure for the final intermediate without compromising on quality standards.
- Enhanced Supply Chain Reliability: The mild reaction conditions and the use of stable, commercially available catalysts like Platinum Oxide ensure that production is not susceptible to the supply volatility often associated with niche or hazardous reagents. The robustness of the process against minor variations in temperature and pressure means that batch-to-batch consistency is significantly improved, reducing the risk of out-of-specification materials that could disrupt downstream API synthesis. This reliability is paramount for long-term supply agreements with major pharmaceutical companies who prioritize continuity of supply above all else.
- Scalability and Environmental Compliance: From an environmental perspective, the process generates significantly less hazardous waste compared to traditional methods, aligning with increasingly strict global environmental regulations. The absence of heavy metal contaminants like nickel in the final product simplifies the purification burden and reduces the environmental footprint of the manufacturing site. The inherent safety of the process, characterized by low pressure and moderate temperature, facilitates easier scale-up from pilot plant to commercial tonnage, allowing suppliers to rapidly increase capacity to meet growing market needs for Ramelteon APIs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of this Ramelteon intermediate using the patented one-step method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering a transparent view of the technology's capabilities. Understanding these details helps stakeholders make informed decisions regarding vendor selection and process validation.
Q: Why is the one-step hydrogenation method superior to traditional Raney Nickel processes?
A: The one-step method eliminates the formation of difficult-to-remove polymeric impurities (A and B) and ensures complete conversion of the intermediate amine, significantly improving final product purity and safety by avoiding pyrophoric catalysts.
Q: What represents the critical quality attribute in this synthesis regarding impurity control?
A: The critical attribute is the minimization of Intermediate III residue (<0.05%) and the prevention of Impurity C formation, which is structurally similar to the final product and notoriously difficult to separate in downstream processing.
Q: Is this process scalable for commercial API manufacturing?
A: Yes, the process utilizes mild reaction conditions (0-80°C) and standard hydrogenation equipment, making it highly suitable for scale-up from pilot plants to multi-ton commercial production without requiring specialized high-pressure infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ramelteon Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to deliver superior pharmaceutical intermediates. Our technical team has thoroughly analyzed the advancements presented in patent CN111116531A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to implement this efficient one-step hydrogenation process. We are committed to maintaining stringent purity specifications and utilizing our rigorous QC labs to ensure that every batch of 2-(1,6,7,8-tetrahydro-2H-indeno[5,4-b]furan-8-yl)ethylamine hydrochloride meets the highest international standards for impurity control, specifically targeting the minimization of Intermediate III and polymeric byproducts.
We invite global partners to collaborate with us to leverage this advanced technology for your Ramelteon supply chain. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis that details how switching to our optimized manufacturing route can reduce your overall procurement costs. We encourage you to reach out today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a seamless integration of our high-quality intermediates into your API production workflow.
