Advanced Chiral Resolution for Ramelteon Intermediates: Scalable and Safe Manufacturing Solutions
Advanced Chiral Resolution for Ramelteon Intermediates: Scalable and Safe Manufacturing Solutions
The pharmaceutical landscape for sleep disorder treatments has been significantly shaped by the development of melatonin receptor agonists, with Ramelteon standing out as a pioneering therapeutic agent. Central to the production of this critical medication is the availability of high-optical-purity intermediates, specifically (S)-2-(1,6,7,8-tetrahydro-2H-indeno[5,4-b]furan-8-yl)ethylamine. Patent CN101531647B introduces a transformative resolution method that addresses the longstanding challenges associated with synthesizing this chiral building block. Unlike traditional methods that rely on complex catalytic systems, this innovation utilizes readily available optically pure organic acids to achieve efficient separation of enantiomers. For R&D Directors and Procurement Managers seeking a reliable pharmaceutical intermediate supplier, this technology represents a pivotal shift towards safer, more cost-effective, and industrially viable manufacturing processes that ensure consistent supply chain continuity for global drug production.
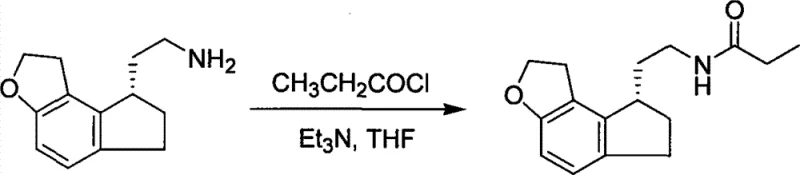
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral amines for pharmaceutical applications has heavily depended on asymmetric catalytic hydrogenation, a technique that, while effective, introduces substantial operational complexities and safety hazards. As illustrated in prior art such as Japanese Patent JP11140073, conventional routes often necessitate the use of expensive homogeneous ruthenium catalysts like Ru2Cl4[(R)-BINAP]2NEt3 under high-pressure hydrogen conditions, sometimes exceeding 100 atm. These stringent requirements demand specialized high-pressure reaction vessels and rigorous safety protocols, which significantly inflate capital expenditure and operational overheads for manufacturing facilities. Furthermore, the removal of trace heavy metal residues from the final product to meet stringent regulatory standards adds additional purification steps, complicating the workflow and potentially reducing overall yield. The reliance on precious metal catalysts also exposes the supply chain to volatility in raw material pricing and availability, creating bottlenecks that can disrupt production schedules for critical API intermediates.
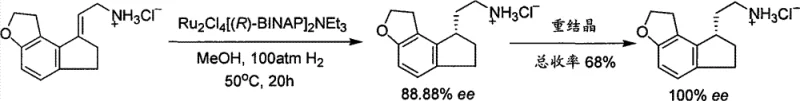
The Novel Approach
In stark contrast to the high-pressure hydrogenation routes, the method disclosed in CN101531647B employs a classical yet optimized chiral resolution strategy that leverages the differential solubility of diastereomeric salts. By reacting the racemic amine with inexpensive, commercially available optically pure organic acids such as L-(-)-dibenzoyltartaric acid or L-(-)-camphorsulfonic acid, the process creates diastereomeric salts that can be separated through simple crystallization. This approach eliminates the need for high-pressure equipment and toxic heavy metal catalysts, thereby drastically simplifying the engineering requirements for commercial scale-up of complex pharmaceutical intermediates. The operational simplicity allows for execution in standard glass-lined or stainless-steel reactors at atmospheric pressure and moderate temperatures, significantly enhancing process safety and reducing the barrier to entry for manufacturing partners. This shift not only mitigates safety risks but also streamlines the purification workflow, making it an ideal candidate for cost reduction in API manufacturing where efficiency and safety are paramount.
Mechanistic Insights into Diastereomeric Salt Crystallization
The core of this technological advancement lies in the precise manipulation of stereochemistry through diastereomeric salt formation, a fundamental concept in chiral separation that is executed here with exceptional efficiency. When the racemic (±)-amine interacts with a single enantiomer of a chiral acid, two distinct diastereomeric salts are formed: one comprising the (S)-amine and the chiral acid, and the other comprising the (R)-amine and the same acid. Unlike enantiomers, which possess identical physical properties in an achiral environment, these diastereomers exhibit different physical characteristics, most notably solubility in specific organic solvents. By carefully selecting solvent systems such as ethanol, acetonitrile, or their mixtures, and controlling the crystallization temperature between 0°C and 30°C, the process selectively precipitates the less soluble diastereomeric salt enriched with the desired (S)-enantiomer. This thermodynamic control ensures that the mother liquor retains the unwanted (R)-isomer, allowing for the isolation of the target compound with high optical purity through simple filtration.
Impurity control is inherently built into this crystallization mechanism, as the lattice energy of the forming crystals favors the incorporation of the matching stereoisomer while excluding the mismatched counterpart. The patent data indicates that through optimization of the molar ratio between the racemate and the resolving agent, typically between 0.5 and 1.5, the enantiomeric excess (ee) can be driven to levels exceeding 93% to 96% without the need for iterative recrystallization steps that often plague other resolution methods. The subsequent neutralization of the isolated salt with a base like sodium hydroxide releases the free amine, which is then extracted into an organic phase. This liquid-liquid extraction step further purifies the product by removing the water-soluble organic acid salt, resulting in a final product that meets the stringent purity specifications required for downstream synthesis of Ramelteon. This robust mechanism ensures batch-to-batch consistency, a critical factor for regulatory compliance in pharmaceutical manufacturing.
How to Synthesize (S)-2-(1,6,7,8-tetrahydro-2H-indeno[5,4-b]furan-8-yl)ethylamine Efficiently
Implementing this resolution method requires a disciplined approach to solvent selection and temperature control to maximize the yield and optical purity of the final intermediate. The process begins with the dissolution of the racemic amine and the chosen chiral acid in a suitable solvent, followed by a heating phase to ensure a homogeneous solution before initiating the crystallization process. Detailed standard operating procedures regarding specific solvent ratios, cooling rates, and filtration techniques are essential for reproducing the high ee values reported in the patent literature. For technical teams looking to adopt this route, understanding the nuances of supersaturation and nucleation is key to preventing the co-crystallization of the unwanted enantiomer. The detailed standardized synthesis steps see the guide below for a comprehensive breakdown of the operational parameters.
- Dissolve the racemic amine and optically pure organic acid in a suitable solvent like ethanol or acetonitrile.
- Heat the mixture to ensure complete dissolution, then cool slowly to induce crystallization of the diastereomeric salt.
- Filter the crystals, neutralize with base, and extract to isolate the high-purity (S)-enantiomer.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the transition from high-pressure hydrogenation to chiral resolution offers profound advantages that resonate deeply with Procurement Managers and Supply Chain Heads focused on stability and cost efficiency. The elimination of expensive ruthenium catalysts removes a significant variable cost component, as precious metals are subject to market fluctuations and require complex recovery processes to be economically viable. Furthermore, the avoidance of high-pressure hydrogenation reduces the need for specialized maintenance and safety certifications for reaction vessels, leading to substantial cost savings in both capital investment and ongoing operational expenditures. This process simplification translates directly into a more resilient supply chain, as the raw materials required—common organic acids and standard solvents—are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks that can halt production lines.
- Cost Reduction in Manufacturing: The economic benefits of this resolution method are driven by the substitution of high-cost catalytic systems with low-cost stoichiometric reagents that are easily sourced. By removing the requirement for precious metal catalysts, manufacturers avoid the significant expenses associated with catalyst procurement, loading, and the rigorous purification steps needed to remove metal traces to ppm levels. Additionally, the ambient pressure conditions reduce energy consumption related to compression and heating, contributing to a lower overall carbon footprint and utility costs. These factors combine to create a manufacturing profile that is significantly more cost-effective than traditional asymmetric hydrogenation, allowing for better margin management in the production of high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply chain continuity is greatly enhanced by the reliance on commodity chemicals rather than specialized catalytic systems that may have long lead times or limited suppliers. The resolving agents used, such as tartaric acid derivatives, are produced at scale for various industries, ensuring a stable and predictable supply even during market disruptions. This availability reduces the lead time for high-purity pharmaceutical intermediates, enabling manufacturers to respond more agilely to fluctuations in demand for the final drug product. The robustness of the process also means that technology transfer between manufacturing sites is smoother, as the equipment requirements are standard across the fine chemical industry, facilitating multi-site production strategies.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of hazardous high-pressure steps and toxic heavy metals. The waste streams generated are primarily organic salts and solvents, which are easier to treat and dispose of compared to heavy metal-contaminated waste, aligning with increasingly strict environmental regulations. The simplicity of the unit operations—dissolution, crystallization, filtration, and extraction—allows for easy scale-up from 100 kgs to 100 MT/annual commercial production without requiring fundamental changes to the process design. This scalability ensures that the supply can grow in tandem with the market demand for Ramelteon, providing a secure foundation for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral resolution technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical principles underlying the method. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the consensus on best practices for achieving optimal yield and purity while maintaining safety and cost standards.
Q: Why is chiral resolution preferred over asymmetric hydrogenation for this intermediate?
A: Chiral resolution avoids the use of expensive ruthenium catalysts and high-pressure hydrogenation equipment, significantly reducing safety risks and capital expenditure while maintaining high optical purity.
Q: What resolving agents are effective for this synthesis?
A: Optically pure organic acids such as L-(-)-dibenzoyltartaric acid, L-(-)-di-p-toluoyltartaric acid, and L-(-)-camphorsulfonic acid have proven effective in forming separable diastereomeric salts.
Q: How is the optical purity verified in this process?
A: The optical purity is typically verified using High-Performance Liquid Chromatography (HPLC) after derivatization with agents like D-camphorsulfonyl chloride to ensure ee values exceed 93%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-2-(1,6,7,8-tetrahydro-2H-indeno[5,4-b]furan-8-yl)ethylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of reliable supply chains for complex pharmaceutical intermediates like the chiral amine precursor for Ramelteon. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch meets the highest standards of quality and safety. Our facility is equipped to handle the specific solvent systems and crystallization protocols required for this resolution method, providing a seamless transition from process development to full-scale manufacturing.
We invite you to collaborate with us to optimize your supply chain for this critical intermediate. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments that demonstrate our capability to deliver this high-value intermediate consistently. By partnering with us, you gain access to a robust manufacturing platform that prioritizes safety, quality, and cost-efficiency, ensuring the uninterrupted production of your life-saving medications.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
