Advanced Catalytic Synthesis of 2-Mercaptobenzimidazole for Commercial Scale-Up
The chemical industry is constantly seeking more efficient pathways to synthesize critical heterocyclic compounds, and the recent disclosure in patent CN113773261A represents a significant leap forward in the production of 2-mercaptobenzimidazole derivatives. This innovative preparation method addresses long-standing challenges associated with the oxidative instability of o-phenylenediamine precursors, a issue that has historically plagued manufacturers aiming for high-purity outputs. By integrating a specialized antioxidant system alongside a high-efficiency tertiary amine catalyst, this technology achieves product yields exceeding 99% while virtually eliminating the generation of saline wastewater. For R&D directors and procurement specialists alike, this patent offers a compelling blueprint for cost reduction in fine chemical manufacturing, particularly for applications ranging from proton pump inhibitor intermediates to advanced rubber vulcanization accelerators. The ability to recycle reaction mother liquor directly back into the process not only enhances atom economy but also drastically simplifies the downstream purification workflow, marking a departure from the resource-intensive protocols of the past.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for 2-mercaptobenzimidazole compounds have typically relied on the condensation of o-phenylenediamine with carbon disulfide in the presence of strong alkalis such as liquid caustic soda. While literature reports indicate yields around 96.3% for some variations, these conventional methods suffer from inherent defects that compromise both economic efficiency and environmental compliance. A primary drawback is the susceptibility of the o-phenylenediamine raw material to oxidative discoloration during the reaction process; the electron-rich amino groups readily react with atmospheric oxygen to form colored by-products, necessitating rigorous and costly purification steps such as activated carbon decolorization and acid precipitation. Furthermore, these legacy processes generate substantial quantities of salt-containing wastewater due to the use of inorganic bases, imposing a heavy burden on effluent treatment facilities and increasing the overall operational expenditure for chemical plants. Alternative methods using ethanol solvents have attempted to mitigate these issues but often result in lower yields, such as the 90.5% reported in certain academic studies, and still fail to completely prevent the formation of organic by-products that contaminate the mother liquor.
The Novel Approach
In stark contrast to these outdated techniques, the novel approach detailed in the patent introduces a paradigm shift by employing a protective antioxidant strategy coupled with organic amine catalysis. By creatively adding antioxidants like sodium hydrosulfite, thiourea dioxide, or L-ascorbic acid to the reaction matrix, the method effectively shields the o-phenylenediamine starting material from oxidative degradation, ensuring that the final product remains white and high-purity without the need for complex refining. The selection of high-efficiency catalysts, specifically tertiary amines such as urotropin or triethylamine, significantly accelerates the reaction kinetics, allowing for rapid conversion of raw materials into the desired mercaptobenzimidazole structure. This synergistic combination not only pushes product yields to over 99% but also enables the direct reuse of the reaction mother liquor and catalyst system, thereby creating a closed-loop manufacturing process that minimizes waste discharge and maximizes resource utilization for sustainable chemical production.
Mechanistic Insights into Antioxidant-Assisted Condensation
The core mechanistic advantage of this synthesis lies in the precise control of the reaction environment to suppress side reactions that typically degrade product quality. The o-phenylenediamine structure is inherently prone to oxidation due to the activating effect of the benzene ring on the amino groups, which can lead to the formation of complex polymeric impurities if left unchecked. The introduction of reducing agents acts as a sacrificial shield, preferentially reacting with dissolved oxygen or radical species before they can attack the diamine substrate. This preservation of the starting material integrity is crucial for maintaining high optical purity and consistent batch-to-batch reproducibility, which are critical parameters for pharmaceutical intermediate suppliers. Additionally, the use of tertiary amine catalysts facilitates the nucleophilic attack of the amino group on the carbon disulfide, lowering the activation energy required for the cyclization step and ensuring that the reaction proceeds to completion even under relatively mild thermal conditions.
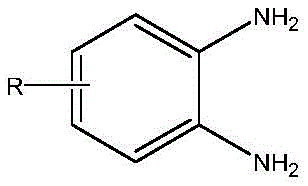
As illustrated in the structural framework above, the versatility of this method extends to various substituted o-phenylenediamines where R can be hydrogen, alkyl, or alkoxy groups, allowing for the synthesis of a diverse library of mercaptobenzimidazole derivatives. The mechanism ensures that regardless of the electronic nature of the substituent R, the antioxidant protection remains effective, preventing the formation of colored impurities that are common in unsubstituted or alkyl-substituted variants. This robustness is further enhanced by the solvent system, which can utilize water or low-carbon alcohols; interestingly, when using alcoholic media, the saturation of the product in the mother liquor allows for easy crystallization upon cooling, while the recycled solvent retains its catalytic activity. The absorption of hydrogen sulfide gas, a toxic by-product of the condensation, by multistage alkali scrubbing converts a hazardous waste stream into a valuable sodium hydrosulfide solution, demonstrating a holistic approach to green chemistry that integrates safety, efficiency, and environmental stewardship into the molecular design of the process.
How to Synthesize 2-Mercaptobenzimidazole Efficiently
The operational protocol for this synthesis is designed for seamless integration into existing fine chemical infrastructure, requiring only standard reactor setups equipped with reflux condensers and gas absorption units. The process begins with the uniform mixing of the o-phenylenediamine compound, the chosen medium, the antioxidant, and the catalyst, creating a homogeneous reaction phase that is ready for the introduction of carbon disulfide. Detailed standardized synthesis steps see the guide below.
- Uniformly mix the o-phenylenediamine compound with a medium (water or alcohol), a specific antioxidant (e.g., sodium hydrosulfite), and a tertiary amine catalyst.
- Slowly add carbon disulfide at controlled temperatures (20-80°C) while maintaining reflux to prevent volatilization.
- Heat the mixture to 80-100°C for 0.5 to 10 hours to complete the reaction, then filter and recycle the mother liquor.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology translates into tangible strategic advantages that go beyond simple yield improvements. The elimination of oxidative by-products means that the crude product often meets high-purity specifications directly after filtration, removing the need for expensive recrystallization solvents or chromatographic purification columns. This simplification of the downstream processing significantly reduces the consumption of auxiliary materials and shortens the overall production cycle time, allowing facilities to increase throughput without expanding their physical footprint. Moreover, the ability to recycle the reaction mother liquor and catalyst directly back into subsequent batches creates a compounding effect on cost savings, as the effective consumption of high-value reagents is minimized over the lifespan of the production campaign.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the drastic reduction in waste treatment costs and raw material consumption. By avoiding the generation of large volumes of salt-containing wastewater, manufacturers can bypass the capital and operational expenditures associated with complex effluent neutralization and disposal systems. The high conversion efficiency ensures that nearly every mole of the expensive o-phenylenediamine starting material is converted into saleable product, minimizing the financial loss associated with unreacted feedstock. Furthermore, the replacement of harsh inorganic bases with recyclable organic amine catalysts reduces the corrosion load on reactor vessels, extending equipment lifespan and lowering maintenance budgets for the facility.
- Enhanced Supply Chain Reliability: From a sourcing perspective, the raw materials required for this synthesis—o-phenylenediamines, carbon disulfide, and common antioxidants—are commodity chemicals with stable global supply chains, reducing the risk of production stoppages due to material shortages. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent qualities, ensures that production can continue reliably even if minor fluctuations in utility supplies occur. This resilience is critical for maintaining continuous supply to downstream customers in the pharmaceutical and polymer industries, where interruptions can have cascading effects on their own manufacturing schedules and market commitments.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from laboratory glassware to pilot-scale reactors without loss of efficiency or selectivity. The use of water as a preferred solvent aligns perfectly with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions, making it easier for manufacturers to obtain and maintain operating permits. The closed-loop nature of the mother liquor recycling and the safe capture of hydrogen sulfide gas demonstrate a commitment to sustainable manufacturing practices, which is becoming a key differentiator for suppliers seeking contracts with environmentally conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this antioxidant-assisted synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the technology's capabilities. Understanding these details is essential for evaluating the feasibility of adopting this method for your specific production needs.
Q: How does the new method prevent oxidative discoloration of raw materials?
A: The process incorporates specific antioxidants such as sodium hydrosulfite or L-ascorbic acid directly into the reaction mixture. These agents effectively scavenge oxygen and prevent the electron-rich amino groups of the o-phenylenediamine from oxidizing, thereby eliminating colored by-products and ensuring high product purity without extensive decolorization steps.
Q: What are the environmental advantages of this synthesis route?
A: Unlike traditional methods that generate large volumes of salt-containing wastewater requiring neutralization, this novel approach allows for the direct recycling of reaction mother liquor and catalysts. Furthermore, hydrogen sulfide by-products are captured via multistage alkali absorption to produce reusable sodium hydrosulfide solutions, resulting in a process with virtually zero wastewater discharge.
Q: Can this method be scaled for industrial production of rubber additives?
A: Yes, the method is highly scalable. It utilizes water or low-carbon alcohols as solvents rather than hazardous organic media, and the reaction conditions (80-100°C) are easily manageable in standard stainless steel reactors. The high conversion rate (>99%) and simplified purification process make it economically viable for large-scale manufacturing of rubber accelerators and pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Mercaptobenzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced synthesis technology for the global supply of high-performance chemical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in an industrial setting. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-mercaptobenzimidazole meets the exacting standards required for pharmaceutical and specialty polymer applications. We are committed to leveraging our technical expertise to optimize this green chemistry route for maximum efficiency and sustainability.
We invite you to engage with our technical procurement team to discuss how this innovative process can be tailored to your specific volume requirements and quality targets. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this superior manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will strengthen your supply chain and enhance your competitive position in the market.
