Advanced Racemization Technology for High-Purity Fluoroquinolone Intermediates and Commercial Scale-Up Capabilities
Advanced Racemization Technology for High-Purity Fluoroquinolone Intermediates and Commercial Scale-Up Capabilities
The pharmaceutical industry constantly seeks efficient pathways to maximize atom economy, particularly in the synthesis of complex chiral antibiotics like fourth-generation fluoroquinolones. Patent CN101429199B introduces a groundbreaking methodology for the preparation of racemic cis-8-benzyl-7,9-dioxo-2,8-diazabicyclo[4.3.0]nonane, a pivotal intermediate in the manufacture of Moxifloxacin and related antifungal agents. This technology addresses a critical bottleneck in chiral synthesis by providing a robust mechanism to recycle the unwanted (1R,6S)-enantiomer, which is typically discarded as waste during optical resolution processes. By converting this low-value byproduct back into a usable racemic mixture through a mild two-step catalytic sequence, the process significantly enhances the overall utilization rate of expensive starting materials. The innovation lies in its departure from harsh thermal dehydrogenation, utilizing instead a selective manganese dioxide catalyzed oxidation followed by controlled high-pressure hydrogenation. This approach not only simplifies the operational workflow but also ensures the structural integrity of the sensitive bicyclic lactam system is maintained throughout the transformation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the conversion of saturated nitrogen heterocycles to their aromatic or partially aromatic counterparts required extreme reaction conditions that posed significant risks to product quality and operational safety. Prior art, such as that described in EP61982 and US4051140, relied heavily on gas-phase dehydrogenation using precious metal catalysts like platinum, palladium, or vanadium at temperatures ranging from 200 to 500 degrees Celsius. These violent thermal conditions frequently induced uncontrolled oxidation side reactions, leading to the degradation of the delicate lactam rings inherent in the diazabicyclo nonane structure. Furthermore, the high energy consumption associated with maintaining such elevated temperatures, combined with the complexity of gas-phase reactor engineering, rendered these methods economically unviable for large-scale fine chemical manufacturing. The lack of selectivity in these high-temperature processes often resulted in a mixture of isomers and degradation products, necessitating costly and time-consuming purification steps that further eroded profit margins. Consequently, the industry lacked a practical, scalable solution for recycling chiral waste, forcing manufacturers to absorb the cost of discarding nearly half of their synthesized material.
The Novel Approach
In stark contrast, the methodology disclosed in CN101429199B employs a sophisticated liquid-phase strategy that operates under remarkably mild conditions, fundamentally altering the economic landscape of intermediate production. The process initiates with an oxidative dehydrogenation step using commercially available manganese dioxide in common organic solvents such as tetrahydrofuran or toluene at temperatures between 60 and 110 degrees Celsius. This gentle oxidation effectively removes the chirality at the bridgehead carbon by forming an achiral tetrahydro-pyrrolo-pyridine intermediate without compromising the rest of the molecular framework. Following isolation, this intermediate undergoes a highly stereoselective hydrogenation using Pd/C or PtO2 catalysts at moderate pressures of 5 to 9 MPa and temperatures of 70 to 100 degrees Celsius. 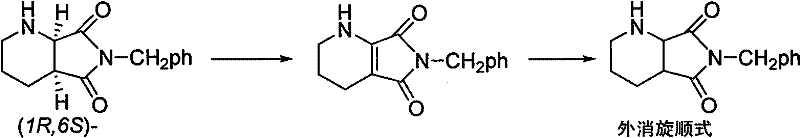 This sequence ensures that the regenerated product is exclusively the desired cis-isomer, completely avoiding the formation of the thermodynamically less stable but difficult-to-separate trans-isomer. The result is a streamlined, high-yield process that transforms a waste management problem into a value-generating opportunity, with overall yields consistently exceeding 90 percent across multiple experimental examples.
This sequence ensures that the regenerated product is exclusively the desired cis-isomer, completely avoiding the formation of the thermodynamically less stable but difficult-to-separate trans-isomer. The result is a streamlined, high-yield process that transforms a waste management problem into a value-generating opportunity, with overall yields consistently exceeding 90 percent across multiple experimental examples.
Mechanistic Insights into MnO2-Catalyzed Oxidative Dehydrogenation
The core of this technological breakthrough resides in the unique reactivity of manganese dioxide as a selective oxidant for allylic and benzylic positions, adapted here for the dehydrogenation of the piperidine ring within the bicyclic system. Unlike radical-based oxidations that can attack various sites on the molecule, activated MnO2 facilitates a surface-mediated hydride abstraction mechanism that specifically targets the C-H bonds adjacent to the nitrogen atom in the piperidine ring. This specificity is crucial because it allows for the formation of the conjugated enamine system in the tetrahydro-pyrrolo-pyridine intermediate while leaving the adjacent pyrrolidine-dione ring untouched. 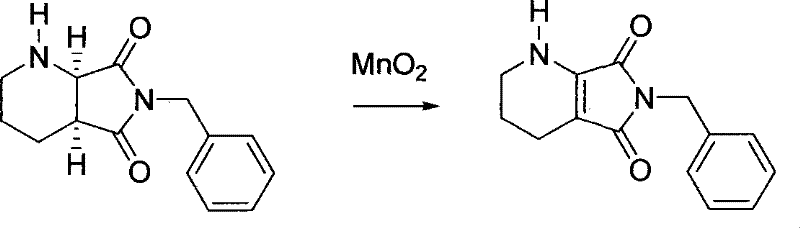 The reaction proceeds through a transition state where the manganese surface coordinates with the nitrogen lone pair, lowering the activation energy for hydrogen removal and ensuring that the reaction stops at the desired oxidation state rather than proceeding to full aromatization or ring cleavage. This controlled oxidation is the key to erasing the stereochemical information at the C-1 and C-6 positions, rendering the molecule achiral and ready for the subsequent reset of stereochemistry. The use of a heterogeneous catalyst also implies that the reaction kinetics are governed by surface area and diffusion, allowing for precise control over the reaction extent simply by monitoring time and temperature, thereby preventing over-oxidation.
The reaction proceeds through a transition state where the manganese surface coordinates with the nitrogen lone pair, lowering the activation energy for hydrogen removal and ensuring that the reaction stops at the desired oxidation state rather than proceeding to full aromatization or ring cleavage. This controlled oxidation is the key to erasing the stereochemical information at the C-1 and C-6 positions, rendering the molecule achiral and ready for the subsequent reset of stereochemistry. The use of a heterogeneous catalyst also implies that the reaction kinetics are governed by surface area and diffusion, allowing for precise control over the reaction extent simply by monitoring time and temperature, thereby preventing over-oxidation.
Following the oxidation, the control of stereochemistry during the reduction phase is equally critical for ensuring the utility of the final product in downstream resolution processes. The hydrogenation step utilizes the thermodynamic stability of the cis-fused ring system to drive the selectivity, as the cis-isomer is significantly more stable than the trans-isomer due to reduced steric strain in the fused bicyclic framework. Under the specified conditions of 5 to 9 MPa hydrogen pressure and the presence of Pd/C or PtO2, the catalyst surface promotes the syn-addition of hydrogen to the double bond from the less hindered face of the molecule. This mechanistic pathway inherently favors the formation of the cis-configured product, effectively suppressing the generation of trans-impurities that would otherwise complicate the subsequent chiral resolution. By eliminating the trans-isomer at the source, the process ensures that the recycled material possesses the same high purity profile as virgin material, making it fully compatible with existing downstream synthesis lines for Moxifloxacin without requiring additional purification protocols. This level of impurity control is vital for meeting the stringent regulatory standards required for active pharmaceutical ingredient (API) manufacturing.
How to Synthesize Racemic Cis-8-Benzyl-7,9-dioxo-2,8-diazabicyclo[4.3.0]nonane Efficiently
Implementing this racemization protocol requires careful attention to catalyst activation and solvent selection to maximize the recovery of the valuable intermediate. The process begins with the suspension of the unwanted (1R,6S)-enantiomer in a dehydrogenation solvent, followed by the addition of manganese dioxide in a mass ratio of approximately 1:5 relative to the substrate. The mixture is then heated to reflux, typically between 60 and 110 degrees Celsius depending on the solvent boiling point, for a duration of 3 to 10 hours to ensure complete conversion to the yellow tetrahydro-pyrrolo-pyridine intermediate. After filtration to recover the reusable manganese dioxide, the second stage involves dissolving the intermediate in a hydrogenation solvent and subjecting it to high-pressure hydrogen in the presence of a noble metal catalyst.
- Perform oxidative dehydrogenation of the (1R,6S)-enantiomer using manganese dioxide catalyst in a suitable solvent like tetrahydrofuran or toluene at 60-110°C to form the achiral tetrahydro-pyrrolo-pyridine intermediate.
- Conduct high-pressure hydrogenation of the intermediate using Pd/C or PtO2 catalyst at 70-100°C and 5-9MPa hydrogen pressure to selectively reduce the double bond and regenerate the racemic cis-bicyclic structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this racemization technology represents a strategic shift from a linear consumption model to a circular economy model within the manufacturing plant. By enabling the recycling of the (1R,6S)-enantiomer, which was previously considered a low-value byproduct, manufacturers can effectively double the yield of usable intermediate from the same amount of initial raw materials. This drastic improvement in material efficiency translates directly into substantial cost savings, as the demand for expensive chiral starting materials is significantly reduced without compromising output volumes. Furthermore, the reliance on commodity chemicals like manganese dioxide and standard hydrogenation catalysts eliminates the supply chain risks associated with sourcing specialized or scarce reagents. The robustness of the process also means that production schedules are less susceptible to delays caused by complex purification steps or low-yield batches, ensuring a more consistent and reliable flow of intermediates to the API synthesis units.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the elimination of waste disposal costs and the reduction of raw material procurement expenses associated with discarding 50 percent of the resolution output. By converting the unwanted enantiomer back into the racemic pool, the effective cost per kilogram of the usable (1S,6R)-isomer is dramatically lowered, enhancing the overall margin profile of the final antibiotic product. Additionally, the mild reaction conditions reduce energy consumption compared to traditional high-temperature dehydrogenation methods, contributing to lower utility costs per batch. The ability to recover and reuse the manganese dioxide and noble metal catalysts further amplifies these savings, minimizing the recurring expenditure on consumable catalytic materials.
- Enhanced Supply Chain Reliability: Integrating this recycling loop creates a buffer against fluctuations in the global supply of chiral building blocks, as the internal inventory of recyclable waste acts as a secondary source of raw material. This self-sufficiency reduces dependency on external suppliers for the specific (1S,6R)-enantiomer, mitigating the risk of shortages that could halt production lines. The simplicity of the workup procedures, involving basic filtration and solvent removal, ensures that the turnaround time between batches is minimized, allowing for faster response to market demand spikes. Moreover, the high purity of the recycled product ensures that it does not introduce variability into the downstream process, maintaining consistent quality assurance metrics.
- Scalability and Environmental Compliance: The use of liquid-phase reactions in standard stirred-tank reactors makes this process highly scalable from pilot plant to multi-ton commercial production without the need for specialized gas-phase infrastructure. The absence of toxic heavy metal catalysts like vanadium and the avoidance of high-temperature combustion processes align with modern green chemistry principles and environmental regulations. Waste generation is minimized not only through material recycling but also through the simplicity of the effluent stream, which primarily consists of recoverable organic solvents. This environmental compatibility simplifies the permitting process for capacity expansion and reduces the long-term liability associated with hazardous waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this racemization technology in a commercial setting. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for feasibility assessments. Understanding these details is essential for R&D teams evaluating the integration of this route into existing manufacturing workflows.
Q: Why is racemization critical for Moxifloxacin intermediate production?
A: Chiral resolution processes typically utilize only 50% of the racemic starting material. Racemizing the unwanted (1R,6S)-enantiomer back into the racemic mixture allows for its re-entry into the resolution cycle, theoretically doubling the yield from the initial raw materials and drastically reducing waste.
Q: What are the advantages of using MnO2 over traditional dehydrogenation catalysts?
A: Traditional methods often require harsh gas-phase conditions with temperatures exceeding 200°C and precious metal catalysts like Platinum. The MnO2 method operates under mild liquid-phase conditions (60-110°C), minimizing side reactions, preserving the sensitive lactam rings, and allowing for easy catalyst recovery and reuse.
Q: Does this process generate trans-isomer impurities?
A: No, the patented hydrogenation conditions are specifically optimized to favor the thermodynamic cis-configuration. The process effectively avoids the generation of trans-products, ensuring the resulting racemate is suitable for direct optical resolution without complex purification steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 8-Benzyl-7,9-dioxo-2,8-diazabicyclo[4.3.0]nonane Supplier
At NINGBO INNO PHARMCHEM, we recognize that the efficient production of fluoroquinolone intermediates requires not just chemical expertise but a deep understanding of process scalability and quality control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial manufacturing is seamless and risk-free. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation to verify every batch. Our facility is designed to handle complex catalytic reactions safely, including high-pressure hydrogenation, guaranteeing a consistent supply of critical building blocks for your antibiotic synthesis needs.
We invite you to collaborate with us to leverage this advanced racemization technology for your specific production requirements. Contact our technical procurement team today to request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of integrating this recycling loop into your supply chain. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can enhance your operational efficiency and reduce your overall cost of goods sold.
