Advanced Manufacturing of High-Purity Quetiapine Intermediates for Global Pharmaceutical Supply Chains
Advanced Manufacturing of High-Purity Quetiapine Intermediates for Global Pharmaceutical Supply Chains
The pharmaceutical landscape for atypical antipsychotics is undergoing a significant transformation driven by the need for stricter impurity control and more sustainable manufacturing processes. Patent CN103724294A introduces a groundbreaking methodology for the preparation of Quetiapine, specifically targeting the critical intermediate 1-[2-(2-hydroxyethoxy)ethyl]piperazine hydrochloride. This innovation addresses long-standing challenges in the industry regarding the removal of toxic piperazine derivatives that often persist through traditional high-temperature purification steps. By shifting the paradigm from free base distillation to hydrochloride salt recrystallization, this technology offers a pathway to achieve purity levels exceeding 99.5% while drastically simplifying the operational workflow. For global stakeholders, this represents a pivotal opportunity to enhance the quality profile of generic Quetiapine fumarate while simultaneously reducing the environmental footprint associated with energy-intensive distillation protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Quetiapine has been plagued by inefficient routes that rely heavily on the use of protected piperazine derivatives or harsh purification techniques. Traditional methodologies often employ protecting groups such as Boc, Cbz, or Ac to mask one amino group of the piperazine ring, necessitating a lengthy sequence of protection, substitution, and deprotection steps that significantly erode overall yield. Furthermore, existing industrial practices frequently utilize high-temperature vacuum distillation to purify the free base form of the key intermediate, a process that demands expensive, specialized equipment capable of withstanding extreme thermal and pressure conditions. This aggressive thermal treatment is not only energy-prohibitive but also chemically detrimental, as it promotes the formation of unknown piperazine impurities that are notoriously difficult to separate in downstream processing. These residual impurities pose a severe risk to patient safety, as they can carry through to the final Active Pharmaceutical Ingredient (API), potentially leading to unforeseen toxic side effects and regulatory non-compliance. Consequently, the reliance on these archaic techniques creates a bottleneck for manufacturers seeking to balance cost-efficiency with the rigorous quality standards demanded by modern pharmacopeias.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent data utilizes a direct alkylation strategy that bypasses the need for amino protection entirely, streamlining the synthesis into a more concise and robust operation. The core innovation lies in the generation and purification of the intermediate as a hydrochloride salt rather than a free base, allowing for effective purification via low-temperature recrystallization instead of destructive high-heat distillation. This strategic shift ensures that thermally sensitive impurities are never formed in the first place, resulting in an intermediate with exceptional chemical integrity that serves as a superior building block for the final coupling reaction. The subsequent reaction with 11-chloro-dibenzo[b,f][1,4]thiazepine proceeds with high efficiency in common solvents like toluene, utilizing simple inorganic bases and catalytic amounts of sodium iodide to drive the nucleophilic substitution to completion. By integrating these improvements, the process achieves crude Quetiapine purity levels greater than 99% and final fumarate salt purity exceeding 99.5%, with single impurities controlled below 0.1%.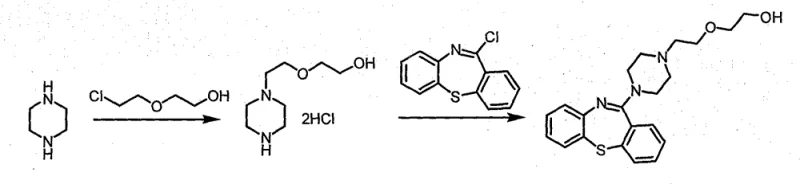
Mechanistic Insights into Direct Alkylation and Salt Formation
The mechanistic foundation of this improved synthesis rests on a highly selective nucleophilic substitution reaction where the piperazine nitrogen attacks the electrophilic carbon of 2-(2-chloroethoxy)ethanol. Under optimized thermal conditions ranging from 120 to 140 degrees Celsius, the reaction kinetics favor the mono-substitution product without the need for steric or electronic protection of the second nitrogen atom. The presence of piperazine hydrochloride in the reaction mixture acts as a buffer and a source of chloride ions, which helps to stabilize the transition state and suppress the formation of di-substituted byproducts. Following the alkylation, the immediate conversion of the crude reaction mixture into the hydrochloride salt form leverages the differential solubility properties of the salt versus the impurities in alcoholic solvents. This crystallization step is thermodynamically driven at low temperatures, effectively excluding organic contaminants that would otherwise co-distill in a free base purification scenario. The result is a crystalline solid with a defined lattice structure that inherently rejects amorphous impurities, providing a level of purity that distillation simply cannot match without significant product loss.
Furthermore, the coupling mechanism with the dibenzothiazepine core is facilitated by the use of sodium iodide, which acts as a catalyst to enhance the leaving group ability of the chlorine atom on the heterocyclic ring through the Finkelstein reaction principle. This in-situ generation of a more reactive iodide species accelerates the rate of the nucleophilic attack by the piperazine intermediate, allowing the reaction to proceed to completion within a reasonable timeframe of approximately 12 hours under reflux. The use of anhydrous sodium carbonate as the base ensures that the reaction medium remains sufficiently alkaline to deprotonate the piperazine nitrogen for attack, without being so strong as to degrade the sensitive thiazepine ring system. This careful balance of reagents creates a clean reaction profile where the primary pathway dominates, minimizing the generation of regio-isomers or degradation products. The final conversion to the fumarate salt capitalizes on the specific acid-base interaction between the basic Quetiapine molecule and fumaric acid, precipitating the therapeutic salt form in high yield while leaving neutral organic impurities in the mother liquor.
How to Synthesize 1-[2-(2-hydroxyethoxy)ethyl]piperazine Hydrochloride Efficiently
The execution of this synthesis requires precise control over thermal parameters and stoichiometric ratios to maximize the formation of the desired mono-alkylated species. Operators must maintain the reaction temperature within the optimal window of 120 to 140 degrees Celsius to ensure complete conversion of the chloro-ethoxy-ethanol while preventing thermal degradation of the piperazine ring. The detailed standardized synthesis steps, including specific workup procedures and recrystallization protocols, are outlined in the guide below to ensure reproducibility across different manufacturing sites. Adherence to these parameters is critical for achieving the high purity specifications required for pharmaceutical grade intermediates.
- React piperazine with 2-(2-chloroethoxy)ethanol at 120-140°C to form the hydrochloride salt intermediate, purifying via low-temperature recrystallization.
- Couple the purified intermediate with 11-chloro-dibenzo[b,f][1,4]thiazepine in toluene using sodium carbonate and sodium iodide under reflux.
- Convert the crude Quetiapine base to Quetiapine Fumarate by reacting with fumaric acid in isopropanol to achieve >99.5% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route translates into tangible strategic advantages that extend far beyond simple chemical yield metrics. The elimination of complex protection and deprotection steps significantly reduces the number of unit operations required, thereby lowering the overall consumption of raw materials and solvents per kilogram of finished product. This simplification of the process flow directly correlates to a reduction in manufacturing lead times, allowing suppliers to respond more agilely to fluctuations in market demand for antipsychotic medications. Moreover, the avoidance of high-temperature vacuum distillation removes a major bottleneck in production capacity, as this equipment is often a shared resource that limits throughput in multi-product facilities. By shifting to a crystallization-based purification, manufacturers can decouple their production schedules from the constraints of specialized distillation units, enhancing overall facility utilization rates and supply continuity.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the removal of expensive protecting group reagents and the associated waste disposal costs. By operating under solvent-free or low-solvent conditions for the initial alkylation step, the process drastically cuts down on solvent purchase and recovery expenses, which are typically a major component of variable manufacturing costs. Additionally, the energy savings achieved by replacing high-vacuum distillation with low-temperature recrystallization are substantial, as the thermal load on the plant utilities is significantly decreased. These cumulative efficiencies allow for a more competitive pricing structure for the final API, providing a distinct margin advantage in the generic pharmaceutical market without compromising on quality standards.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this method mitigates the risk of batch failures caused by the formation of intractable impurities. The consistent production of high-purity intermediates ensures that downstream coupling reactions proceed predictably, reducing the incidence of out-of-specification batches that can disrupt inventory planning. Furthermore, the raw materials required for this process, such as piperazine and chloro-ethoxy-ethanol, are commodity chemicals with stable global availability, reducing the risk of supply shortages associated with exotic or custom-synthesized reagents. This reliability is crucial for maintaining uninterrupted production lines and meeting the stringent delivery commitments expected by large-scale pharmaceutical partners.
- Scalability and Environmental Compliance: The environmental profile of this synthesis is markedly improved, aligning with the increasing regulatory pressure for greener chemical manufacturing. The reduction in solvent usage and the elimination of high-energy distillation steps contribute to a lower carbon footprint for the production of Quetiapine. The process generates less hazardous waste, particularly avoiding the tarry residues often associated with high-thermal stress reactions, which simplifies effluent treatment and lowers disposal costs. This alignment with green chemistry principles not only facilitates easier regulatory approval in environmentally strict jurisdictions but also enhances the corporate sustainability credentials of the manufacturing entity, a factor of growing importance to international buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented Quetiapine synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation to assist decision-makers in evaluating the feasibility of adoption. Understanding these nuances is essential for R&D teams planning technology transfer and for procurement teams assessing supplier capabilities.
Q: How does the new hydrochloride salt purification method improve safety compared to traditional distillation?
A: Traditional methods rely on high-temperature vacuum distillation of the free base, which generates unknown piperazine impurities that are toxic and difficult to remove. The novel method utilizes low-temperature recrystallization of the hydrochloride salt, effectively eliminating these hazardous byproducts and ensuring a safer final API profile.
Q: What are the yield improvements observed in the patented Quetiapine synthesis route?
A: The optimized process demonstrates significant efficiency gains, with total recovery rates reaching up to 84% in scaled examples. By avoiding multi-step protection and deprotection sequences, the overall material throughput is maximized while maintaining crude product purity above 99%.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the process is designed for industrial scalability by utilizing solvent-free or common solvent systems like toluene and avoiding harsh high-vacuum equipment. The mild reaction conditions and robust purification steps make it highly adaptable for metric-ton manufacturing campaigns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quetiapine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthesis routes requires a partner with deep technical expertise and proven scale-up capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent can be fully realized in a practical industrial setting. We operate stringent purity specifications and maintain rigorous QC labs equipped to detect trace impurities at the ppm level, guaranteeing that every batch of Quetiapine intermediate meets the highest global regulatory standards. Our commitment to quality assurance means that we can consistently deliver the high-purity hydrochloride salt intermediate necessary for producing safe and effective final drug products.
We invite you to engage with our technical procurement team to discuss how this innovative manufacturing process can be integrated into your supply chain to drive value. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both the quality and cost-efficiency of your Quetiapine sourcing strategy.
