Advanced Synthetic Route for Quetiapine Hemifumarate: Enhancing Purity and Scalability for Global API Markets
Advanced Synthetic Route for Quetiapine Hemifumarate: Enhancing Purity and Scalability for Global API Markets
The pharmaceutical industry constantly seeks robust, scalable, and environmentally sustainable pathways for the production of critical active pharmaceutical ingredients (APIs). Patent CN103772319A introduces a significant technological breakthrough in the synthesis of Quetiapine Hemifumarate, a widely prescribed atypical antipsychotic agent. This innovative methodology departs from traditional, hazardous synthetic routes by utilizing o-bromonitrobenzene and thiosalicylic acid as primary starting materials. The process encompasses a streamlined sequence of displacement, reduction, condensation, halogenation, and final salt formation, offering a superior alternative for manufacturers aiming to optimize their supply chains. By focusing on mild reaction conditions and simplified operational procedures, this technology addresses the growing demand for high-purity intermediates while mitigating the environmental footprint associated with legacy manufacturing processes.
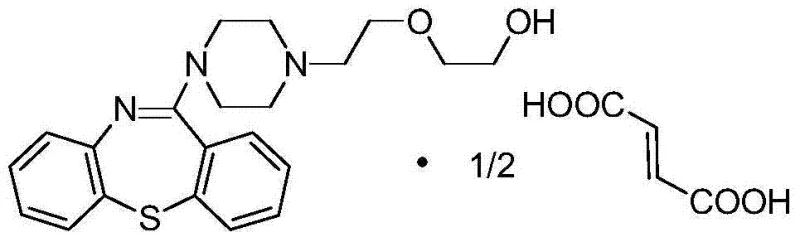
For R&D directors and procurement specialists, understanding the structural integrity and synthetic accessibility of the target molecule is paramount. The chemical architecture of Quetiapine Hemifumarate, as depicted above, relies on a complex dibenzothiazepine core fused with a piperazine ring and a hydroxyethoxy side chain. Achieving this structure efficiently requires precise control over regioselectivity and functional group tolerance. The patented route described herein provides a clear roadmap for constructing this scaffold without resorting to expensive or dangerous reagents, thereby positioning it as a highly attractive option for reliable quetiapine intermediate supplier networks seeking to enhance their portfolio offerings.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Quetiapine and its salts has relied heavily on starting materials such as 2-diaminodiphenyl sulfide. While chemically feasible, these traditional pathways often necessitate the use of highly toxic reagents like phenyl chloroformate or triphosgene to effect esterification and subsequent cyclization. These reagents pose severe safety risks to personnel and require specialized containment infrastructure, driving up capital expenditure and operational costs. Furthermore, intermediates generated in these older routes are frequently unstable, leading to complex impurity profiles that are difficult to purge in later stages. The violent operational conditions often required for these transformations can result in lower overall yields and significant generation of hazardous waste, creating substantial bottlenecks for cost reduction in API manufacturing.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages a substitution-reduction-cyclization strategy that fundamentally alters the risk profile of the synthesis. By initiating the sequence with o-bromonitrobenzene and thiosalicylic acid, the process bypasses the need for hazardous carbonylating agents entirely. The reaction conditions are notably milder, utilizing common solvents like ethanol and toluene which are readily available and easier to recover. This shift not only simplifies the operational workflow but also enhances the stability of intermediates throughout the synthesis. The result is a cleaner reaction profile with fewer by-products, directly translating to higher purity specifications for the final API and a more streamlined downstream processing workflow that appeals to supply chain heads focused on efficiency.
Mechanistic Insights into FeCl2-Catalyzed Reduction and Cyclization
The core of this synthetic innovation lies in the strategic manipulation of the nitro and carboxyl functionalities to build the tricyclic system. The initial step involves a nucleophilic aromatic substitution where the thiol group of thiosalicylic acid displaces the bromine atom on the nitrobenzene ring under basic conditions. This forms the critical diphenyl sulfide backbone (Intermediate Z1). Subsequently, the reduction of the nitro group to an amine (Intermediate Z2) is achieved using ferrous sulfate and ammonia. This choice of reducing agent is particularly advantageous for large-scale operations as it avoids the high pressure and safety concerns associated with catalytic hydrogenation, while still providing excellent conversion rates. The mechanistic pathway ensures that the carboxylic acid moiety remains intact and positioned correctly for the subsequent intramolecular cyclization.
Following the reduction, the formation of the dibenzothiazepinone ring (Intermediate Z3) is driven by acid-catalyzed dehydration cyclization. The use of concentrated sulfuric acid in a toluene-water biphasic system facilitates the removal of water, pushing the equilibrium towards the cyclic ketone. This step is crucial for establishing the rigid core of the molecule. Finally, the activation of the ketone with phosphorus oxychloride (POCl3) generates a reactive imidoyl chloride species in situ, which undergoes nucleophilic attack by the piperazine derivative. This sequence minimizes side reactions and ensures high regioselectivity, effectively controlling the impurity spectrum. For technical teams, this level of mechanistic control is essential for validating the process during technology transfer and regulatory filings.
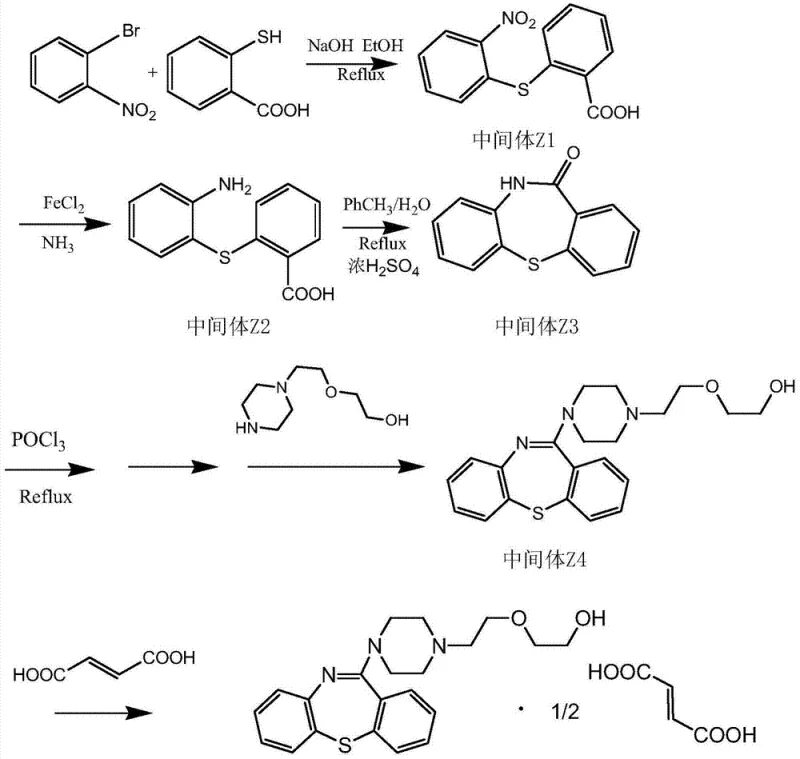
How to Synthesize Quetiapine Hemifumarate Efficiently
The synthesis of this complex psychotropic intermediate is broken down into five distinct, manageable unit operations that are highly amenable to batch processing. The protocol begins with the formation of the sulfide linkage, followed by reduction, ring closure, side-chain attachment, and final salification. Each step has been optimized to maximize yield and minimize work-up complexity. For example, the crystallization of Intermediate Z2 is carefully controlled by temperature gradients to ensure high purity before proceeding to the cyclization step. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and isolation techniques, are outlined in the guide below to assist process engineers in replicating this high-efficiency route.
- Perform nucleophilic substitution between o-bromonitrobenzene and thiosalicylic acid using NaOH in ethanol to form Intermediate Z1.
- Reduce Intermediate Z1 using ferrous sulfate and ammonia in water to obtain Intermediate Z2 (2-amino-2'-carboxyl diphenyl sulfide).
- Cyclize Intermediate Z2 in a toluene/water mixture with concentrated sulfuric acid to generate the tricyclic ketone Intermediate Z3.
- Activate Intermediate Z3 with POCl3 followed by condensation with N-(2-hydroxyethyl)piperazine derivative to form the free base Intermediate Z4.
- Dissolve Intermediate Z4 in acetone and react with fumaric acid at room temperature to crystallize the final Quetiapine Hemifumarate product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers compelling advantages that directly impact the bottom line and supply chain resilience. By eliminating the reliance on scarce or heavily regulated toxic reagents, manufacturers can secure a more stable supply of raw materials. The simplified operational parameters reduce the need for exotic reactor configurations, allowing for production in standard glass-lined or stainless steel vessels. This flexibility is critical for maintaining continuity of supply in a volatile global market. Furthermore, the ability to recover and reuse solvents such as toluene and ethanol contributes to a greener manufacturing profile, aligning with increasingly stringent environmental regulations and corporate sustainability goals.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like triphosgene significantly lowers the direct material costs associated with production. Additionally, the high yields reported in the patent embodiments, particularly in the initial substitution and final salt formation steps, reduce the overall consumption of starting materials per kilogram of finished product. The simplified post-treatment procedures, such as straightforward filtration and crystallization rather than complex chromatographic purifications, further decrease labor and utility costs, resulting in substantial cost savings for the final API.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as o-bromonitrobenzene, thiosalicylic acid, and ferrous sulfate ensures that the supply chain is not vulnerable to the bottlenecks often seen with specialty reagents. These materials are produced globally in large volumes, guaranteeing consistent availability. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in raw material quality or environmental conditions, thereby reducing lead time for high-purity antipsychotic APIs and ensuring reliable delivery schedules to downstream customers.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with reaction steps that generate minimal hazardous waste. The avoidance of heavy metal catalysts and toxic gases simplifies the waste treatment process, lowering the cost of environmental compliance. The ability to operate under reflux conditions with common solvents allows for easy transition from pilot plant to commercial scale production. This scalability ensures that manufacturers can rapidly respond to market demand surges without compromising on product quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this specific synthetic methodology. They are derived from the patent's background analysis and the demonstrated beneficial effects of the new route. Understanding these nuances is vital for stakeholders evaluating the feasibility of adopting this technology for their own manufacturing lines or sourcing strategies.
Q: What are the key advantages of this synthesis route over traditional methods?
A: This route avoids highly toxic reagents like phenyl chloroformate and triphosgene used in conventional methods. It utilizes milder reaction conditions, simpler operations, and achieves higher yields with significantly reduced three-waste pollution, making it more environmentally friendly and economically viable for large-scale production.
Q: How is impurity control managed during the reduction step?
A: Impurity control is rigorously managed during the reduction of Intermediate Z1 to Z2 by monitoring the reaction until the content of Z2 is below 1%. The process involves precise temperature control (cooling to 40-50°C before adding methanol) and extended crystallization times to ensure high purity of the off-white solid intermediate.
Q: Is this process suitable for commercial scale-up?
A: Yes, the process is specifically designed for industrialization. It features simple reaction operations, mild conditions that are easy to control, and the potential for solvent recovery. The elimination of complex post-treatment steps and the use of common solvents like ethanol, toluene, and acetone facilitate easy batch production and supply chain stability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quetiapine Hemifumarate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN103772319A are fully realized in practical manufacturing. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic potential of switching to this greener, more efficient route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring a seamless integration of high-quality intermediates into your supply chain.
