Advanced Manufacturing of Dabigatran Etexilate: A Cost-Effective Route for Global Supply Chains
Advanced Manufacturing of Dabigatran Etexilate: A Cost-Effective Route for Global Supply Chains
The pharmaceutical landscape for anticoagulant therapy has been revolutionized by the introduction of direct thrombin inhibitors, with Dabigatran Etexilate standing as a cornerstone molecule in modern cardiovascular care. As detailed in patent CN108373466B, a novel preparation method has emerged that addresses critical bottlenecks in the traditional synthesis of this vital Active Pharmaceutical Ingredient (API). This technical insight report analyzes the breakthrough methodology which utilizes stable orthoester derivatives to construct the complex benzimidazole scaffold, offering a robust alternative to legacy processes plagued by hazardous reagents and low throughput. For R&D directors and supply chain leaders, understanding this shift is paramount, as it represents a transition from fragile, high-maintenance chemistry to a streamlined, industrially friendly protocol. The strategic adoption of this route not only enhances the purity profile of the final drug substance but also fundamentally reshapes the cost structure and reliability of the global supply chain for this high-value therapeutic.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
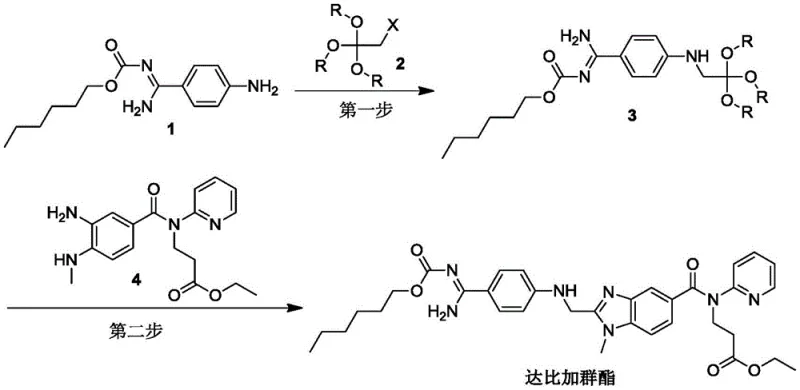
Historically, the industrial synthesis of Dabigatran Etexilate, as pioneered by major pharmaceutical entities in the early 2000s, relied heavily on the use of highly reactive and problematic acylating agents such as acid anhydrides and acyl chlorides. These conventional pathways, while chemically feasible, introduced severe operational challenges that persist as pain points for generic manufacturers today. The primary defect lies in the physical properties of these reagents; they possess pungent, irritating odors that necessitate expensive containment systems and rigorous personal protective equipment (PPE) protocols to ensure worker safety. Furthermore, acid chlorides are notoriously hygroscopic and unstable in the presence of moisture, demanding strictly anhydrous reaction conditions that drive up utility costs for solvent drying and nitrogen blanketing. From a process chemistry perspective, these harsh conditions often lead to the formation of complex impurity profiles, requiring extensive downstream purification steps that erode overall yield. The cumulative effect is a synthetic route that is not only environmentally burdensome due to corrosive waste streams but also economically inefficient, with historical total yields reported as low as 6.3%, rendering the process marginally viable for cost-sensitive markets.
The Novel Approach
In stark contrast, the methodology disclosed in CN108373466B introduces a paradigm shift by employing stable orthoester derivatives, specifically compounds like trimethyl 2-chloroorthoacetate, as the key building blocks. This innovative approach effectively circumvents the volatility and sensitivity issues associated with traditional acyl chlorides. The new route is characterized by the use of reagents that are stable at room temperature, non-hygroscopic, and devoid of the noxious odors that complicate facility operations. By shifting the reaction mechanism to utilize these robust precursors, the process eliminates the critical need for absolute anhydrous environments, allowing for more flexible and forgiving reaction parameters. This simplification translates directly into operational excellence; the reaction process becomes significantly easier to operate and control, reducing the risk of batch failures due to minor fluctuations in humidity or temperature. Moreover, the synthetic route is notably shorter, minimizing the number of unit operations and intermediate isolations required, which inherently reduces material loss and solvent consumption. The result is a cleaner reaction profile with fewer byproducts, facilitating a more straightforward purification process and ultimately delivering a higher quality API intermediate with superior economic metrics.
Mechanistic Insights into Orthoester-Mediated Cyclization
The core of this technological advancement lies in the nuanced mechanistic pathway facilitated by the orthoester functionality. In the first critical step, the nucleophilic attack of the amidine nitrogen on the orthoester carbon is catalyzed by iodide ions, such as those provided by potassium iodide, which act as a potent nucleophilic catalyst to enhance the leaving group ability of the halide. This reaction proceeds smoothly in polar aprotic solvents like DMF at moderate temperatures ranging from 50 to 70°C. The stability of the orthoester allows for a controlled release of the reactive species, preventing the runaway exotherms often seen with acid chlorides. This controlled reactivity is crucial for maintaining high selectivity, ensuring that the desired cyclization occurs without significant degradation of the sensitive carbamate protecting groups present on the hexyl chain. The use of mild inorganic bases like sodium bicarbonate further buffers the reaction medium, neutralizing any generated acid without promoting hydrolysis of the ester moieties, thereby preserving the integrity of the molecular skeleton throughout the transformation.
Following the formation of the key intermediate, the second step involves a condensation reaction with a functionalized benzimidazole amine. This step is catalyzed by organic acids such as p-toluenesulfonic acid monohydrate in dichloromethane at ambient temperatures (20-25°C). The mild acidic conditions promote the dehydration necessary for the final ring closure and bond formation without inducing decomposition of the thermally labile components of the molecule. From an impurity control standpoint, this low-temperature approach is vital; it suppresses side reactions such as over-alkylation or hydrolysis of the ethyl ester side chains, which are common failure modes in harsher thermal processes. The combination of specific solvent systems and precise pH control ensures that the impurity spectrum remains narrow and predictable, greatly simplifying the analytical burden on quality control teams and ensuring that the final product consistently meets the stringent specifications required for regulatory submission and commercial release.
How to Synthesize Dabigatran Etexilate Efficiently
The implementation of this synthesis strategy requires precise adherence to the optimized reaction parameters defined in the patent to maximize yield and purity. The process is designed to be scalable, moving seamlessly from laboratory benchtop experiments to pilot plant and full commercial production. Operators must focus on the stoichiometric balance between the orthoester derivative and the amine precursor, as well as the efficient removal of byproducts during the aqueous workup phases. The following guide outlines the critical operational phases, emphasizing the specific conditions that drive the high efficiency of this route. For detailed standard operating procedures and safety data sheets regarding the specific reagents mentioned, please refer to the technical documentation provided below.
- Step 1: React hexyl carbamate derivative (Compound 1) with trimethyl 2-chloroorthoacetate (Compound 2) using potassium iodide and sodium bicarbonate in DMF at 60°C to form the intermediate amidine precursor.
- Step 2: Condense the resulting intermediate (Compound 3) with the benzimidazole amine derivative (Compound 4) using p-toluenesulfonic acid catalyst in dichloromethane at ambient temperature (20-25°C).
- Purification: Isolate the final crude product via aqueous workup and purify using column chromatography with a dichloromethane/methanol gradient to achieve high-purity Dabigatran Etexilate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthesis route offers tangible strategic benefits that extend far beyond simple chemical curiosity. The elimination of hazardous, pungent reagents fundamentally alters the risk profile of the manufacturing process, leading to significant reductions in insurance premiums, waste disposal costs, and facility maintenance expenses associated with corrosion control. By removing the dependency on strictly anhydrous conditions, the process becomes more resilient to environmental variables, reducing the likelihood of costly batch rejections due to moisture ingress. This robustness translates into a more reliable supply of high-purity pharmaceutical intermediates, ensuring that downstream formulation timelines are met without interruption. Furthermore, the shortened synthetic sequence reduces the overall cycle time per batch, enhancing the agility of the supply chain to respond to fluctuating market demands for anticoagulant therapies.
- Cost Reduction in Manufacturing: The economic impact of substituting acid chlorides with stable orthoesters is profound. By eliminating the need for specialized corrosion-resistant reactors and complex off-gas scrubbing systems required for handling pungent acid chlorides, capital expenditure (CAPEX) and operational expenditure (OPEX) are drastically reduced. Additionally, the higher yields achieved in each step mean that less raw material is wasted, directly lowering the cost of goods sold (COGS). The simplified purification process also reduces solvent consumption and energy usage for distillation, contributing to a leaner, more cost-effective manufacturing model that allows for competitive pricing in the global generic drug market.
- Enhanced Supply Chain Reliability: The stability of the starting materials used in this route, such as trimethyl 2-chloroorthoacetate, ensures a consistent and secure supply of raw materials. Unlike sensitive acyl chlorides that require cold chain logistics and have short shelf lives, these orthoester derivatives can be stored under standard conditions, reducing inventory management complexity and the risk of raw material spoilage. This stability mitigates supply chain disruptions, ensuring continuous production capability even during logistical challenges. The ease of operation also means that the process can be transferred between manufacturing sites with greater fidelity, diversifying the supply base and reducing single-point-of-failure risks for critical API supplies.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of toxic, volatile byproducts make this process inherently greener and easier to scale. Regulatory bodies increasingly favor manufacturing processes that minimize the use of hazardous reagents and reduce waste generation. This route aligns perfectly with green chemistry principles, facilitating smoother regulatory approvals and reducing the environmental footprint of production. The scalability is further supported by the use of common, commercially available solvents like DMF and dichloromethane, which are easily sourced in bulk quantities globally. This ensures that as demand for Dabigatran Etexilate grows, production capacity can be expanded rapidly without encountering bottlenecks related to exotic reagent availability or complex engineering constraints.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and technical disclosures within the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the long-term viability of the supply partnership.
Q: What are the primary advantages of this synthesis route over traditional methods?
A: This method replaces volatile, pungent, and moisture-sensitive acid anhydrides or acyl chlorides with stable, odorless orthoester derivatives. This significantly simplifies operational control, reduces equipment corrosion risks, and eliminates the need for stringent anhydrous conditions, thereby lowering production costs and improving worker safety.
Q: What yields can be expected from this novel preparation method?
A: Experimental data indicates a substantial improvement in efficiency. The first step achieves yields around 80%, and the second step yields approximately 70%. This represents a dramatic increase compared to the historical 6.3% total yield reported in earlier Boehringer Ingelheim routes, making it highly viable for commercial scale-up.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process utilizes common solvents like DMF and dichloromethane and operates at mild temperatures (0-70°C). The use of stable starting materials and the avoidance of hazardous acylating agents make the reaction easier to control and safer to scale from kilogram to multi-ton production levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dabigatran Etexilate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex molecules like Dabigatran Etexilate requires more than just a patent; it demands deep process expertise and unwavering commitment to quality. Our team of expert chemists has extensively analyzed this novel orthoester-mediated route and possesses the technical capability to optimize it for maximum efficiency. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, delivering an API intermediate that exceeds global regulatory standards.
We invite you to collaborate with us to leverage this advanced synthesis technology for your portfolio. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and regional compliance needs. We encourage you to contact us today to request specific COA data and comprehensive route feasibility assessments, allowing you to make informed decisions that will strengthen your supply chain and enhance your market competitiveness in the anticoagulant sector.
