Advanced Biocatalytic Synthesis of Chiral Alcohol Intermediates for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to produce optically pure chiral building blocks, which are essential for the synthesis of active pharmaceutical ingredients. A significant breakthrough in this domain is documented in patent CN110592035B, which discloses a novel mutant of carbonyl reductase, specifically derived from the EbSDR8 amino acid sequence. This innovation addresses the critical limitations of traditional chemical synthesis by offering a biocatalytic route that operates under mild conditions while delivering exceptional stereoselectivity. The patent details specific single-point and multi-point mutations, such as at positions 94, 145, 153, 188, 199, and 202, which collectively enhance the enzyme's affinity and specificity towards prochiral ketones. For R&D directors and procurement specialists, this technology represents a paradigm shift towards more sustainable and efficient manufacturing processes that eliminate the reliance on harsh chemical reagents and expensive metal catalysts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of optically active chiral alcohols, such as (R)-(3)-chloro-(1)-phenylpropanol, has relied heavily on chemical asymmetric reduction using chiral metal derivatives. While these methods have been partially adopted in industrial settings, they are fraught with significant operational difficulties and environmental concerns. The reaction conditions are often severe, requiring strict temperature and pressure controls that increase energy consumption and operational risks. Furthermore, the use of heavy metal catalysts introduces the persistent risk of metal residues in the final product, necessitating complex and costly purification steps to meet regulatory standards for pharmaceutical intermediates. These downstream processing requirements not only extend the production lead time but also substantially inflate the overall manufacturing costs, making the supply chain less agile and more vulnerable to raw material price fluctuations.
The Novel Approach
In stark contrast, the biocatalytic approach utilizing the engineered EbSDR8 mutants offers a streamlined and environmentally friendly alternative that directly addresses the痛点 of conventional chemistry. The mutant enzymes function effectively in aqueous buffer systems at moderate temperatures ranging from 20°C to 50°C, significantly reducing the energy footprint of the reaction. Crucially, the recombinant cells can catalyze the asymmetric reduction in the presence of isopropanol without the addition of any external coenzymes, simplifying the reaction system and reducing reagent costs. This self-sufficient cofactor regeneration mechanism ensures that the process remains economically viable even at high substrate concentrations, with the patent reporting substrate loads of 100-300 g/L. By bypassing the need for heavy metals, this method inherently produces a cleaner crude product, thereby simplifying the isolation and purification workflow and enhancing the overall process safety profile.
Mechanistic Insights into EbSDR8-Catalyzed Asymmetric Reduction
The core of this technological advancement lies in the rational protein design of the carbonyl reductase EbSDR8, where specific amino acid substitutions alter the enzyme's active site geometry to favor the production of the desired chiral alcohol. The mutations, including G94A, S153L, Y188A, and others, are strategically located near the substrate binding pocket, enhancing the binding affinity and directional orientation of the prochiral ketone. This structural optimization allows the enzyme to discriminate effectively between enantiomers, achieving optical purity values greater than 99% ee, which is critical for downstream drug synthesis. The kinetic parameters reveal a marked improvement in catalytic efficiency, with certain mutants demonstrating conversion rates approaching 99.9% and yields between 70% and 98%. This high level of performance is maintained even under high substrate loading conditions, indicating a robust enzyme variant capable of withstanding industrial process stresses.
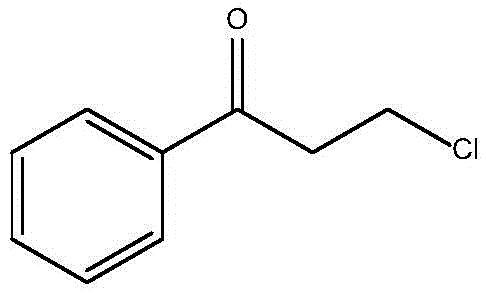
Controlling the impurity profile is another critical aspect where this enzymatic route excels, particularly for applications requiring high-purity pharmaceutical intermediates. The high regioselectivity and stereoselectivity of the mutant enzymes minimize the formation of by-products and unwanted isomers that are common in chemical reduction processes. This specificity ensures that the resulting chiral alcohol, such as the (R)-(3)-chloro-(1)-phenylpropanol shown in the structural analysis, meets stringent quality specifications with minimal downstream purification effort. For supply chain managers, this translates to a more predictable and consistent product quality, reducing the risk of batch failures and ensuring a reliable supply of critical intermediates. The ability to operate without heavy metals also means that the final product is free from toxic metal contaminants, a key requirement for regulatory compliance in the global pharmaceutical market.
How to Synthesize (R)-(3)-chloro-(1)-phenylpropanol Efficiently
Implementing this biocatalytic route involves a series of well-defined steps that leverage the recombinant expression system described in the patent data. The process begins with the construction of the mutant gene and its insertion into a suitable expression vector, followed by transformation into a host microorganism like E. coli BL21(DE3). Once the engineered strain is established, fermentation conditions are optimized to maximize enzyme expression, often using induction strategies with IPTG or lactose to trigger high-level protein production. The subsequent biotransformation step utilizes the whole cells or purified enzyme in a buffer system with isopropanol, where the reaction progress is monitored to ensure complete conversion of the substrate. Detailed standardized synthesis steps see the guide below.
- Construct recombinant expression vectors containing specific EbSDR8 mutant genes (e.g., G94A/S153L/Y188A) and transform into E. coli BL21(DE3) host cells.
- Cultivate the engineered bacteria in fermentation media, induce protein expression with IPTG or lactose, and harvest the recombinant cells or purified enzyme.
- Perform asymmetric reduction of prochiral ketones in a buffer system with isopropanol as the hydrogen donor, maintaining pH 5.5-10.5 and temperatures between 20-50°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this mutant carbonyl reductase technology offers substantial strategic advantages that go beyond mere technical performance. The elimination of expensive heavy metal catalysts and the simplification of the reaction system directly contribute to a significant reduction in raw material costs and waste disposal expenses. Furthermore, the mild reaction conditions reduce the need for specialized high-pressure or high-temperature equipment, lowering capital expenditure and maintenance costs for manufacturing facilities. The robustness of the enzyme at high substrate concentrations also means that production throughput can be increased without proportionally increasing reactor volume, leading to better asset utilization and operational efficiency.
- Cost Reduction in Manufacturing: The biocatalytic process eliminates the need for costly chiral metal catalysts and complex cofactor regeneration systems, which are major cost drivers in traditional chemical synthesis. By using isopropanol as a hydrogen donor within the reaction system, the process reduces reagent consumption and simplifies the workup procedure, leading to substantial cost savings in utilities and waste treatment. The high conversion rates and yields minimize the loss of valuable starting materials, ensuring that the overall material cost per kilogram of product is significantly optimized compared to conventional methods.
- Enhanced Supply Chain Reliability: The use of recombinant microorganisms for enzyme production ensures a scalable and consistent supply of the biocatalyst, reducing dependency on scarce or volatile chemical reagents. The mild operating conditions and high substrate tolerance of the mutants allow for flexible manufacturing schedules and faster turnaround times, which is crucial for meeting tight delivery deadlines in the pharmaceutical supply chain. This reliability is further bolstered by the enzyme's stability, which reduces the risk of production delays due to catalyst deactivation or process upsets.
- Scalability and Environmental Compliance: The process is inherently green, avoiding the use of toxic heavy metals and generating less hazardous waste, which simplifies environmental compliance and reduces the burden of waste disposal regulations. The scalability of the fermentation and biotransformation steps has been demonstrated at high substrate concentrations, indicating that the technology can be seamlessly transferred from laboratory to commercial scale without significant process re-engineering. This ease of scale-up ensures that production capacity can be rapidly expanded to meet market demand while maintaining strict environmental and safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this carbonyl reductase mutant technology. These answers are derived directly from the patent specifications and experimental data, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for making informed decisions about integrating this biocatalytic route into existing manufacturing workflows.
Q: What are the advantages of this mutant carbonyl reductase over wild-type enzymes?
A: The mutant EbSDR8 enzymes exhibit significantly improved catalytic activity and substrate tolerance, achieving conversion rates up to 99.9% and optical purity exceeding 99% ee without the need for expensive external coenzymes.
Q: Does this biocatalytic process require complex cofactor regeneration systems?
A: No, the recombinant cells can efficiently catalyze the reaction in an isopropanol-containing system, which serves as both the solvent and the hydrogen donor, eliminating the need for additional cofactor regeneration enzymes.
Q: How does this technology impact the purity profile of the final chiral alcohol?
A: By avoiding heavy metal catalysts and utilizing highly specific enzymatic reduction, the process minimizes side reactions and metal residues, resulting in a cleaner impurity profile suitable for stringent pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this enzyme engineering technology for the production of high-value chiral intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab to market is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of chiral alcohol meets the highest industry standards. We are committed to leveraging advanced biocatalytic solutions to drive innovation and efficiency in your supply chain.
We invite you to collaborate with our technical procurement team to explore how this mutant carbonyl reductase technology can optimize your specific manufacturing needs. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits and process improvements for your project. We encourage you to contact us for specific COA data and route feasibility assessments, allowing us to tailor a solution that aligns perfectly with your quality and timeline requirements.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
