Advanced Manufacturing of Chiral Piperazine Derivatives for Pharmaceutical Applications
Advanced Manufacturing of Chiral Piperazine Derivatives for Pharmaceutical Applications
The pharmaceutical industry continuously demands more efficient and cost-effective pathways for synthesizing complex active pharmaceutical ingredient (API) intermediates, particularly those exhibiting potent biological activity such as tachykinin antagonism. Patent CN1451003A discloses a groundbreaking methodology for the preparation of a specific piperazine derivative, identified as Compound I, which serves as a critical intermediate in the development of neurokinin antagonists. This compound exhibits significant pharmacological potential, including antagonism against Substance P, Neurokinin A, and Neurokinin B, making it a high-value target for drug discovery programs focused on central nervous system disorders. The disclosed process represents a substantial technological leap forward by streamlining the synthetic route, thereby addressing long-standing challenges related to yield optimization and operational simplicity in the production of high-purity pharmaceutical intermediates.
For research and development directors and procurement specialists, understanding the nuances of this synthesis is vital for securing a reliable supply chain. The innovation lies not merely in the final molecule but in the strategic manipulation of functional groups to bypass unnecessary synthetic steps. By leveraging selective reactivity, the inventors have created a pathway that is not only chemically elegant but also commercially robust. This report delves deep into the mechanistic underpinnings and commercial implications of this technology, providing a comprehensive analysis for stakeholders looking to integrate this high-value intermediate into their portfolio. The ability to produce such complex chiral molecules with high stereochemical fidelity and minimal waste is a key differentiator in the competitive landscape of fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of piperazine derivatives bearing both phenolic hydroxyl groups and secondary amines has been plagued by chemoselectivity issues. Conventional methodologies, such as those described in prior art documents like WO00/35915A1, typically necessitate the introduction of a protecting group on the phenolic hydroxyl moiety before any alkylation can occur on the piperazine nitrogen. This protective strategy, while chemically sound, introduces significant inefficiencies into the manufacturing process. It requires additional reagents, extra reaction vessels, and extended processing times for both the installation and subsequent removal of the protecting group. Furthermore, each additional step inherently carries a risk of yield loss and impurity generation, compounding the cost and reducing the overall throughput of the synthesis. In an industrial setting, these cumulative inefficiencies translate to higher production costs and longer lead times, which are critical pain points for supply chain managers.
The Novel Approach
In stark contrast, the process detailed in patent CN1451003A offers a streamlined alternative that elegantly circumvents the need for phenolic protection. The inventors discovered that under specific conditions, it is possible to achieve selective N-alkylation of the piperazine ring even in the presence of a free phenolic hydroxyl group. This breakthrough eliminates two entire synthetic steps—the protection and deprotection of the phenol—thereby shortening the overall preparation sequence significantly. The impact on efficiency is profound, with the patent data indicating a dramatic improvement in overall yield from a conventional 41.5% to an impressive 76.7%. This enhancement is not merely a statistical improvement but a fundamental shift in process economics, reducing raw material consumption and waste generation. For a reliable pharmaceutical intermediates supplier, adopting such a route ensures a more competitive pricing structure and a more resilient supply capability.
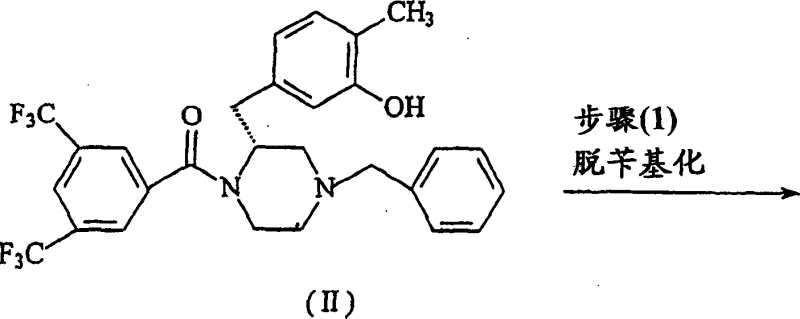
Mechanistic Insights into Selective N-Alkylation and Catalytic Debenzylation
The success of this novel synthesis hinges on two critical chemical transformations: the catalytic debenzylation and the subsequent selective N-alkylation. The first step involves the removal of the benzyl protecting group from the piperazine nitrogen of Compound II. This is achieved through a catalytic reduction process utilizing palladium on carbon (Pd/C) as the catalyst and ammonium formate as the hydrogen donor. The reaction is typically conducted in a mixed solvent system of methanol and water at a controlled temperature range of 50-55°C. This specific thermal window is crucial; it provides sufficient energy to drive the reduction to completion within 4 hours while preventing the degradation of sensitive functional groups. The use of ammonium formate as a transfer hydrogenation agent is particularly advantageous for safety and scalability, avoiding the need for high-pressure hydrogen gas equipment. The result is the clean formation of the secondary amine intermediate, Compound III, with a remarkable isolated yield of 94.8%, demonstrating the high efficiency of this catalytic system.
Following debenzylation, the process moves to the pivotal N-alkylation step, where the secondary amine of Compound III reacts with (2S)-4-(2-chloroethyl)-2-(methoxymethyl)morpholine hydrochloride (Compound IV). The mechanistic elegance here lies in the chemoselectivity; despite the presence of the nucleophilic phenolic oxygen, the reaction conditions favor alkylation at the piperazine nitrogen. This is facilitated by the use of a non-nucleophilic base, such as N,N-diisopropylethylamine (DIPEA), in a polar aprotic solvent like N,N-dimethylformamide (DMF). The addition of potassium iodide acts as a catalyst to enhance the reactivity of the chloroethyl leaving group via in situ formation of the more reactive iodo-analogue. The reaction proceeds smoothly at 50-55°C, yielding the free base Compound Ia. Finally, the conversion to the dihydrochloride salt is achieved by treatment with hydrochloric acid in a solvent like acetone or ethyl acetate, inducing crystallization of the stable sesquihydrate form. This final salt formation step is critical for ensuring the physical stability and handling properties required for pharmaceutical applications.
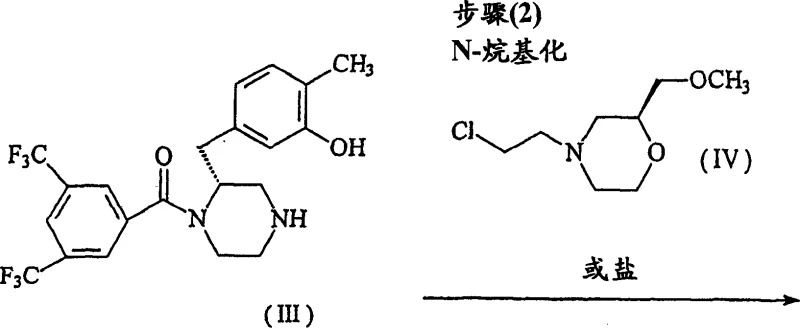
How to Synthesize (2R)-1-[3,5-bis(trifluoromethyl)benzoyl] Piperazine Derivatives Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and purity. The process begins with the charging of the benzyl-protected precursor into a reactor containing methanol and water, followed by the addition of the palladium catalyst and ammonium formate. Maintaining the internal temperature strictly between 50-55°C is essential for optimal kinetics. Upon completion, the catalyst is removed by filtration, and the product is induced to crystallize by the addition of water, a technique that leverages solubility differences to achieve high purity without extensive chromatography. The subsequent alkylation step demands anhydrous conditions and careful stoichiometry of the base to neutralize the hydrochloride salt of the alkylating agent while avoiding side reactions. The final isolation of the dihydrochloride salt involves a controlled acidification and cooling crystallization protocol, which locks the molecule into its thermodynamically stable crystal lattice. Detailed standardized operating procedures for these steps are essential for technology transfer and regulatory compliance.
- Perform catalytic debenzylation of the benzyl-protected precursor using palladium on carbon and ammonium formate at 50-55°C to remove the benzyl group.
- Conduct selective N-alkylation of the resulting piperazine intermediate with (2S)-4-(2-chloroethyl)-2-(methoxymethyl)morpholine hydrochloride in the presence of a base.
- Convert the free base into the stable dihydrochloride sesquihydrate salt through acidification and crystallization to ensure pharmaceutical grade purity.
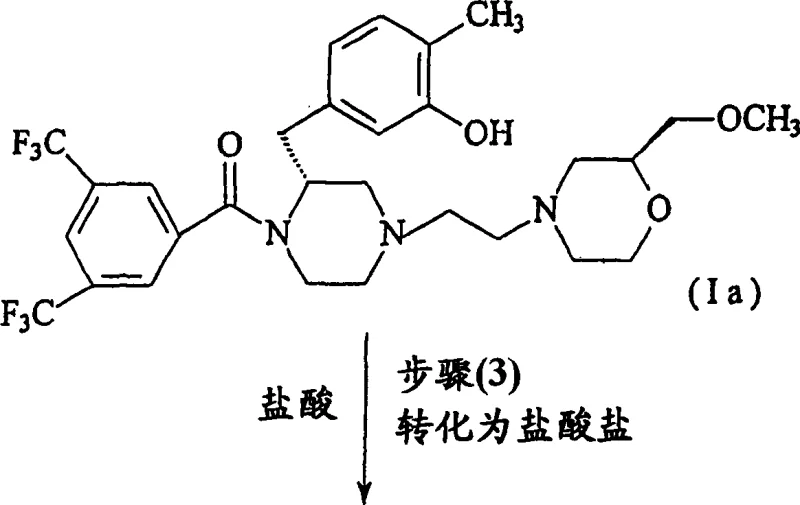
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible strategic benefits that extend beyond simple chemistry. The primary value proposition lies in the drastic simplification of the manufacturing workflow. By eliminating the protection and deprotection steps associated with the phenolic hydroxyl group, the process significantly reduces the number of unit operations required. This reduction directly correlates to lower capital expenditure on equipment usage, reduced labor hours, and decreased utility consumption per kilogram of product. Furthermore, the shorter cycle time enhances the agility of the supply chain, allowing for faster response to market demand fluctuations. The high yields reported in the patent examples suggest a robust process that minimizes raw material waste, contributing to a more sustainable and cost-effective production model. These factors collectively position this synthetic route as a superior choice for long-term sourcing strategies.
- Cost Reduction in Manufacturing: The elimination of phenolic protection steps removes the need for expensive protecting group reagents and the solvents required for their removal. This simplification leads to substantial cost savings in raw materials and waste disposal. Additionally, the high yield of the debenzylation step (94.8%) and the overall improved efficiency mean that less starting material is required to produce the same amount of final product. The use of common, commodity-grade solvents like methanol, water, and ethyl acetate further drives down operational costs compared to processes requiring exotic or highly regulated solvents. This economic efficiency allows for a more competitive pricing structure in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: A shorter synthetic route inherently reduces the number of potential failure points in the manufacturing process. With fewer steps, there is less accumulation of impurities and a lower risk of batch failures due to intermediate instability. The process utilizes widely available catalysts like palladium on carbon and standard reagents, mitigating the risk of supply shortages for specialized chemicals. The ability to crystallize intermediates and the final product effectively ensures that high-purity material can be consistently produced, which is critical for meeting the stringent quality specifications of downstream API manufacturers. This reliability fosters stronger partnerships between suppliers and pharmaceutical clients.
- Scalability and Environmental Compliance: The process has been demonstrated to be scalable, with patent examples showing successful execution on a 30 kg scale. The use of catalytic hydrogenation with ammonium formate avoids the safety hazards associated with high-pressure hydrogen gas, making it safer to scale up in standard multipurpose reactors. Moreover, the reduction in chemical steps leads to a lower E-factor (mass of waste per mass of product), aligning with modern green chemistry principles. The simplified workup procedures, primarily involving filtration and crystallization rather than complex chromatographic separations, facilitate easier waste management and reduce the environmental footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of this piperazine derivative. These insights are derived directly from the experimental data and claims presented in the patent literature. Understanding these details helps stakeholders assess the feasibility and advantages of integrating this technology into their supply chains. The answers reflect the specific conditions and outcomes observed during the development of this process, providing a realistic view of its capabilities.
Q: What is the primary advantage of this synthesis method over conventional routes?
A: The primary advantage is the elimination of phenolic hydroxyl protection and deprotection steps. Conventional methods require protecting the phenol group before alkylation, which adds complexity and reduces overall yield. This novel process allows for selective N-alkylation in the presence of the free phenol, significantly simplifying the workflow and improving the overall yield from approximately 41.5% to 76.7%.
Q: What are the critical reaction conditions for the debenzylation step?
A: The debenzylation step is optimally performed using catalytic reduction with 10% palladium on carbon and ammonium formate as the hydrogen source. The reaction is conducted in a methanol and water mixture at a controlled internal temperature of 50-55°C for 4 hours. These specific conditions ensure high conversion rates (94.8% yield) while maintaining the integrity of the chiral centers.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is highly scalable. The patent data demonstrates successful execution on a multi-kilogram scale (e.g., 30.0 kg input in Example 1), achieving consistent yields and high purity. The use of common solvents like methanol, DMF, and ethyl acetate, along with standard filtration and crystallization techniques, facilitates easy technology transfer to industrial manufacturing facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Piperazine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern pharmaceutical landscape. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and high-quality supply of complex intermediates. Our state-of-the-art facilities are equipped to handle the specific requirements of this synthesis, including catalytic hydrogenation and precise crystallization processes. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest international standards. Our commitment to technical excellence allows us to deliver cost reduction in pharmaceutical intermediates manufacturing without compromising on quality or regulatory compliance.
We invite you to collaborate with us to leverage this advanced technology for your drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments for this piperazine derivative. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain that is not only reliable but also optimized for speed and efficiency, enabling you to bring your therapeutic candidates to market faster.
