Advanced Iron-Catalyzed Synthesis of Substituted Pyridines for Pharmaceutical Intermediates
Advanced Iron-Catalyzed Synthesis of Substituted Pyridines for Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for complex heterocyclic intermediates, particularly those serving as precursors for potent therapeutic agents like endothelin receptor inhibitors. Patent CN1117746C introduces a groundbreaking methodology for the preparation of substituted pyridines, specifically focusing on a novel carbamoylation process that significantly enhances yield and operational simplicity. This technology represents a pivotal shift from traditional radical carbamoylation techniques, offering a reliable pathway for generating high-purity pharmaceutical intermediates. The core innovation lies in the strategic use of stoichiometric quantities of iron(II) salts within an acidic aqueous medium, which drives the conversion of substituted pyridines into valuable carboxamide derivatives with exceptional efficiency. 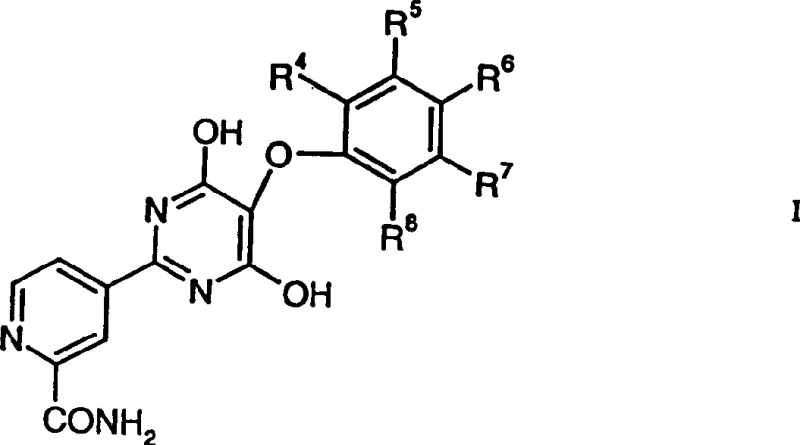 As depicted in the structural representation above, the target compounds of Formula I serve as critical building blocks, where substituents R4 to R8 can be hydrogen, lower-alkoxy, or halogen groups, providing significant chemical diversity for downstream drug development. This patent not only covers the synthesis of these novel compounds but also details their subsequent transformation into biologically active endothelin receptor inhibitors, addressing critical needs in the treatment of circulatory diseases such as hypertension and angina pectoris.
As depicted in the structural representation above, the target compounds of Formula I serve as critical building blocks, where substituents R4 to R8 can be hydrogen, lower-alkoxy, or halogen groups, providing significant chemical diversity for downstream drug development. This patent not only covers the synthesis of these novel compounds but also details their subsequent transformation into biologically active endothelin receptor inhibitors, addressing critical needs in the treatment of circulatory diseases such as hypertension and angina pectoris.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the radical carbamoylation of parent heteroarenes has been described in academic literature, such as the work by F. Minisci et al. in Tetrahedron (1985), which utilized catalytic amounts of iron(II) sulfate. While foundational, these conventional methods often suffer from limited substrate scope and inconsistent yields when applied to complex, highly substituted pyridine systems required for modern drug discovery. The reliance on catalytic rather than stoichiometric metal loading can lead to incomplete conversions or the formation of difficult-to-remove impurities, necessitating extensive purification protocols that increase both production time and cost. Furthermore, traditional organic solvent-based systems pose environmental and safety challenges during commercial scale-up, including flammability risks and the generation of hazardous waste streams that require specialized disposal. For procurement managers and supply chain heads, these inefficiencies translate into higher cost of goods sold (COGS) and potential bottlenecks in securing consistent quality raw materials for API manufacturing.
The Novel Approach
In stark contrast, the process disclosed in CN1117746C leverages a surprising discovery: that employing relatively large amounts of iron(II) salt, specifically ranging from 15-40 mol% relative to the substrate, results in vastly superior yields for specific compounds of Formula I. This novel approach operates in an acidic aqueous media, utilizing formamide as the carbamoyl source and an oxidizing agent such as hydrogen peroxide to drive the radical mechanism. The shift to an aqueous system not only enhances safety by reducing organic solvent usage but also simplifies work-up procedures, as the product can often be isolated via filtration after quenching with reducing agents like sodium metabisulfite. This method effectively overcomes the reactivity barriers associated with sterically hindered or electronically deactivated pyridine rings, ensuring that the synthesis of high-purity pharmaceutical intermediates is both chemically feasible and economically viable. By optimizing the molar ratio of the iron catalyst and controlling the reaction temperature between 0°C and 10°C, the process minimizes side reactions, delivering a crude product with purity levels exceeding 90% directly from the reactor.
Mechanistic Insights into Fe(II)-Mediated Radical Carbamoylation
The mechanistic foundation of this invention rests on the generation of carbamoyl radicals through the oxidation of formamide by hydrogen peroxide, a process that is critically facilitated by the presence of iron(II) ions. In this system, the iron(II) salt acts not merely as a catalyst but as a crucial redox mediator that accelerates the decomposition of peroxide to generate hydroxyl radicals, which subsequently abstract hydrogen from formamide to produce the reactive carbamoyl radical species. 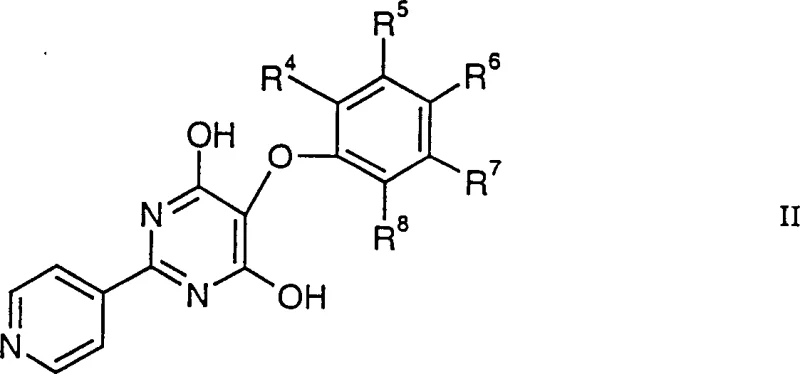 These radicals then attack the electron-deficient pyridine ring of the starting material, shown as Formula II in the diagram above, leading to the formation of the desired carbon-carbon bond at the specific position adjacent to the nitrogen atom. The use of a substantial excess of iron(II) salt ensures a steady state concentration of radicals sufficient to drive the reaction to completion even in the presence of competing side reactions or radical scavengers inherent in the complex molecular architecture. This mechanistic understanding allows R&D directors to appreciate the robustness of the process, as the excess iron serves as a buffer against variations in reagent quality or minor fluctuations in reaction conditions, thereby ensuring batch-to-batch consistency.
These radicals then attack the electron-deficient pyridine ring of the starting material, shown as Formula II in the diagram above, leading to the formation of the desired carbon-carbon bond at the specific position adjacent to the nitrogen atom. The use of a substantial excess of iron(II) salt ensures a steady state concentration of radicals sufficient to drive the reaction to completion even in the presence of competing side reactions or radical scavengers inherent in the complex molecular architecture. This mechanistic understanding allows R&D directors to appreciate the robustness of the process, as the excess iron serves as a buffer against variations in reagent quality or minor fluctuations in reaction conditions, thereby ensuring batch-to-batch consistency.
Furthermore, the control of impurities is intrinsically linked to the precise management of reaction temperature and the stoichiometry of the oxidant. The patent specifies a narrow temperature window of 0°C to 35°C, with a strong preference for 0°C to 10°C, to prevent the over-oxidation of the sensitive pyrimidine-diol moiety or the degradation of the formamide reagent. By maintaining these cryogenic to ambient conditions, the formation of poly-carbamoylated byproducts or oxidative cleavage products is effectively suppressed. The subsequent quenching step with sodium metabisulfite serves a dual purpose: it destroys excess peroxide to ensure safety and reduces any high-valent iron species back to a state that facilitates easy removal during filtration. This rigorous control over the reaction environment results in an impurity profile that is significantly cleaner than those obtained via traditional methods, reducing the burden on downstream purification units and enhancing the overall throughput of the manufacturing line.
How to Synthesize 4-[4,6-Dihydroxy-5-(2-methoxy-phenoxy)-pyrimidin-2-yl]-pyridine-2-carboxamide Efficiently
The synthesis of this key intermediate involves a carefully orchestrated sequence beginning with the carbamoylation of the corresponding dihydroxy-pyrimidine precursor. The process requires the suspension of the starting material in a large volume of formamide, followed by the sequential addition of concentrated sulfuric acid and iron(II) sulfate heptahydrate at low temperatures. The detailed standardized synthesis steps, including specific reagent grades, addition rates, and isolation protocols, are outlined in the guide below to ensure reproducibility and safety during laboratory or pilot plant operations. Adhering to these parameters is critical for achieving the reported 91% theoretical yield and maintaining the stringent purity specifications required for pharmaceutical applications.
- Perform radical carbamoylation of substituted pyridines using formamide and hydrogen peroxide in the presence of 15-40 mol% iron(II) salt at 0-10°C.
- Convert the resulting dihydroxy-pyrimidine intermediate into a dichloro derivative using phosphorus oxychloride (POCl3) and a base.
- Couple the chloro-intermediate with a pyridine sulfonamide derivative under basic conditions to form the final endothelin receptor inhibitor precursor.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this iron-catalyzed carbamoylation technology offers substantial strategic advantages regarding cost stability and operational resilience. The primary driver of cost reduction lies in the substitution of expensive noble metal catalysts or complex organometallic reagents with commodity-grade iron salts and hydrogen peroxide, which are globally available and subject to minimal price volatility. This shift drastically simplifies the raw material sourcing strategy, mitigating the risk of supply disruptions that often plague specialty chemical markets. Additionally, the use of water as the primary reaction medium eliminates the need for large quantities of flammable organic solvents, leading to significant savings in solvent procurement, recovery, and waste disposal costs. The simplified work-up procedure, which relies on filtration rather than complex chromatographic separations, further reduces processing time and labor costs, contributing to a leaner and more efficient manufacturing operation.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and the use of inexpensive, bulk chemicals like formamide and iron sulfate directly lower the variable cost per kilogram of the intermediate. By avoiding the need for specialized ligand synthesis or recovery systems associated with palladium or rhodium catalysis, the capital expenditure for reactor setup is also minimized. The high yield reported in the patent examples implies less raw material waste, meaning more product is generated from the same input mass, effectively stretching the purchasing budget further. Furthermore, the reduced solvent load decreases the energy consumption required for distillation and drying, adding another layer of operational expense savings that accumulates significantly over commercial production volumes.
- Enhanced Supply Chain Reliability: Relying on universally available reagents ensures that production schedules are not held hostage by the lead times of niche suppliers. Iron(II) sulfate and hydrogen peroxide are produced at massive scales for various industries, guaranteeing a continuous supply even during global market fluctuations. This reliability allows for more accurate forecasting and inventory planning, reducing the need for safety stock and freeing up working capital. The robustness of the aqueous process also means that manufacturing can be potentially localized in regions with strict environmental regulations regarding organic solvents, providing flexibility in supply chain network design and reducing logistics costs associated with transporting hazardous materials.
- Scalability and Environmental Compliance: The transition from organic solvents to an aqueous system inherently improves the safety profile of the process, making it easier to scale from pilot batches to multi-ton commercial production without encountering heat transfer or mixing limitations typical of viscous organic media. The reduced generation of hazardous organic waste aligns with increasingly stringent environmental regulations, lowering the compliance burden and potential liability for the manufacturing site. This green chemistry approach not only enhances the corporate sustainability profile but also future-proofs the supply chain against tightening regulatory frameworks on volatile organic compound (VOC) emissions, ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and claims within CN1117746C, providing clarity on the practical aspects of adopting this technology for the production of endothelin receptor inhibitor precursors. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this process into existing manufacturing workflows.
Q: Why does this process require large amounts of iron(II) salt compared to conventional Minisci reactions?
A: Unlike conventional catalytic Minisci reactions that use trace iron, this patented method utilizes 15-40 mol% iron(II) salt to drive the reaction to surprisingly high yields for specific substituted pyridine substrates, overcoming limitations in reactivity and selectivity found in earlier literature.
Q: What are the critical temperature controls for the carbamoylation step?
A: The reaction must be strictly maintained between 0°C and 35°C, with a preferred range of 0°C to 10°C. This tight thermal control is essential to manage the exothermic nature of the hydrogen peroxide addition and to minimize side reactions, ensuring high purity of the carboxamide product.
Q: How does this route improve supply chain reliability for endothelin receptor inhibitors?
A: By utilizing inexpensive, commodity-grade reagents such as formamide, hydrogen peroxide, and iron sulfate in an aqueous medium, the process eliminates reliance on scarce or expensive transition metal catalysts, thereby stabilizing raw material costs and ensuring consistent availability for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Pyridines Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of life-saving cardiovascular medications. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex heterocyclic chemistry, such as the iron-catalyzed carbamoylation described in CN1117746C, positions us as a strategic partner capable of meeting the demanding quality and volume requirements of global pharmaceutical companies.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis route to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this iron-mediated process for your supply chain. We encourage potential partners to contact us directly to obtain specific COA data for our batch-produced intermediates and to receive comprehensive route feasibility assessments that demonstrate our commitment to quality, reliability, and innovation in fine chemical manufacturing.
