Scalable Manufacturing of High-Purity Adefovir Dipivoxil via Optimized Alkylation
The pharmaceutical industry continuously seeks robust manufacturing pathways that balance high purity with economic efficiency, particularly for critical antiviral agents. Patent CN110483575B introduces a transformative synthetic methodology for Adefovir Dipivoxil, a pivotal prodrug used in the treatment of chronic hepatitis B. This innovative approach fundamentally reengineers the alkylation step by eliminating the reliance on phase transfer catalysts, a common bottleneck in nucleoside analogue synthesis. By leveraging a specific combination of polar aprotic solvents and organic base scavengers, the process achieves superior reaction control and impurity management. For R&D directors and procurement specialists, this represents a significant opportunity to optimize the supply chain for high-value antiviral intermediates. The following analysis dissects the technical merits and commercial viability of this PTC-free route, demonstrating its readiness for industrial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Adefovir Dipivoxil has been plagued by inefficiencies associated with phase transfer catalysis. Prior art, such as the method disclosed in CN105646586A, relies heavily on quaternary ammonium salts like tetrabutylammonium bromide to facilitate the reaction between adefovir and chloromethyl pivalate. While effective on a small scale, this dependency introduces severe complications during commercial scale-up. The presence of these catalysts necessitates rigorous downstream purification to meet stringent residual solvent and heavy metal guidelines, adding substantial time and cost to the manufacturing cycle. Furthermore, conventional protocols often utilize diethyl ether for crystallization, a solvent notorious for its low flash point and explosion hazards, rendering it unsuitable for safe, large-volume production environments. The difficulty in controlling reaction endpoints in the presence of phase transfer agents further exacerbates batch-to-batch variability, posing a risk to consistent product quality.
The Novel Approach
The methodology outlined in CN110483575B offers a decisive break from these legacy constraints by executing the condensation reaction without any phase transfer catalyst. Instead, the process utilizes organic bases such as triethylamine or pyridine, which act simultaneously as acid adsorbents and reaction promoters. This simplification allows for precise thermal control, with the addition of chloromethyl pivalate optimized at 55-65°C to ensure complete conversion while minimizing side reactions. The replacement of hazardous diethyl ether with safer C5-C6 ethers, specifically methyl tert-butyl ether or isopropyl ether, dramatically enhances operational safety profiles. This strategic shift not only mitigates explosion risks but also improves the crystallization kinetics, leading to higher recovery rates of the crude product. By streamlining the reagent profile and solvent system, this novel approach establishes a foundation for a more reliable pharmaceutical intermediate supplier network capable of delivering consistent quality.
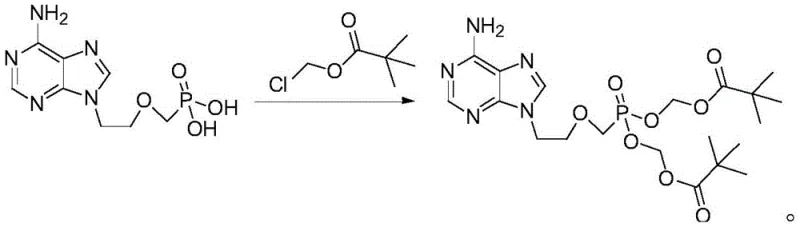
Mechanistic Insights into Base-Promoted Alkylation
The core of this synthetic advancement lies in the mechanistic efficiency of base-promoted nucleophilic substitution. In the absence of a phase transfer catalyst, the reaction relies on the solubility of the adefovir salt in polar aprotic media such as N-methylpyrrolidone (NMP), dimethylformamide (DMF), or dimethyl sulfoxide (DMSO). Triethylamine serves a dual function: it neutralizes the hydrochloric acid byproduct generated during the esterification, driving the equilibrium forward, and it likely facilitates the formation of a more reactive nucleophilic species of adefovir. The careful regulation of temperature between 50°C and 65°C is critical; temperatures below this range result in incomplete conversion, while excessive heat promotes the degradation of the sensitive phosphonate ester linkage. This thermal window ensures that the chloromethyl pivalate reacts selectively with the phosphonic acid groups of adefovir, forming the desired dipivoxil ester with minimal formation of mono-ester impurities.
Impurity control is further refined through the sophisticated purification strategy employed in the final stages. The patent details a mixed solvent recrystallization system, typically combining a good solvent like ethyl acetate or acetone with a poor solvent like methyl tert-butyl ether. This binary solvent system exploits the differential solubility of the target compound versus potential byproducts, such as unreacted starting materials or over-alkylated species. By dissolving the crude product at elevated temperatures (40-50°C) and inducing crystallization upon cooling to 10-15°C, the process effectively excludes structurally similar impurities from the crystal lattice. This results in a final product with HPLC purity exceeding 99.8%, meeting the rigorous standards required for high-purity Adefovir Dipivoxil intended for clinical applications. The ability to consistently achieve such purity levels without complex chromatographic separation underscores the robustness of this chemical design.
How to Synthesize Adefovir Dipivoxil Efficiently
Implementing this synthesis requires strict adherence to the optimized stoichiometry and thermal profiles defined in the patent. The process begins with the dissolution of adefovir in a selected polar aprotic solvent, followed by the addition of the acid scavenger. The key operational parameter is the controlled addition of chloromethyl pivalate at 60°C, maintaining the reaction mixture within the narrow 55-65°C window to maximize yield. Following the reaction, a multi-stage workup involving aqueous washes and organic extraction isolates the crude material. The final purification step utilizes a specific volume ratio of ethyl acetate to methyl tert-butyl ether to precipitate the pure product. For a comprehensive breakdown of the exact quantities, timing, and equipment specifications required for GMP-compliant production, please refer to the standardized protocol below.
- Dissolve Adefovir in a polar aprotic solvent like N-methylpyrrolidone with an acid adsorbent such as triethylamine.
- Add chloromethyl pivalate at a controlled temperature of 55-65°C and monitor the reaction progress via HPLC until completion.
- Purify the crude product using a mixed solvent system of ethyl acetate and methyl tert-butyl ether to achieve high purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the elimination of phase transfer catalysts translates directly into tangible supply chain and cost benefits. Traditional methods incur hidden costs associated with the procurement, handling, and subsequent removal of quaternary ammonium salts. By removing this component entirely, the new process simplifies the bill of materials and reduces the burden on waste treatment facilities. Furthermore, the improved safety profile regarding solvent selection lowers insurance premiums and facility compliance costs, contributing to overall cost reduction in API manufacturing. The robustness of the reaction conditions also means fewer failed batches and less downtime, ensuring a steady flow of material to downstream formulation teams. This reliability is essential for maintaining the continuity of supply for life-saving antiviral medications.
- Cost Reduction in Manufacturing: The most significant economic driver is the complete removal of expensive phase transfer catalysts like tetrabutylammonium bromide. In large-scale production, the cost of these catalysts, combined with the extensive washing procedures required to remove them, represents a substantial portion of the COGS. By switching to a simpler amine-based system, manufacturers can achieve significant raw material savings. Additionally, the reduced molar excess of chloromethyl pivalate required in this method minimizes reagent waste, further optimizing the cost structure without compromising conversion rates.
- Enhanced Supply Chain Reliability: The use of commodity chemicals such as triethylamine and methyl tert-butyl ether ensures that the supply chain is not vulnerable to the shortages often associated with specialized catalysts. These reagents are widely available from multiple global vendors, reducing the risk of procurement bottlenecks. Moreover, the simplified process flow shortens the overall cycle time per batch, allowing for increased throughput and faster response to market demand fluctuations. This agility is crucial for reducing lead time for high-purity antiviral intermediates in a competitive market.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges. This method addresses both by replacing explosive diethyl ether with safer alternatives and eliminating toxic catalyst residues. The ease of reaction monitoring at the specified temperature range allows for seamless technology transfer from pilot plants to multi-ton reactors. Consequently, this supports the commercial scale-up of complex nucleoside analogues while adhering to increasingly strict environmental regulations regarding solvent emissions and hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on process optimization and quality control measures. Understanding these nuances is vital for engineering teams planning to adopt this methodology for commercial production.
Q: Why is the elimination of phase transfer catalysts beneficial for Adefovir Dipivoxil production?
A: Eliminating phase transfer catalysts like tetrabutylammonium bromide removes the need for expensive heavy metal清除 steps, reduces raw material costs significantly, and simplifies the purification process, leading to a cleaner impurity profile.
Q: What are the critical parameters for ensuring high yield in this synthesis?
A: Maintaining the reaction temperature between 55°C and 65°C during the addition of chloromethyl pivalate is crucial. Additionally, optimizing the molar ratio of reagents and utilizing a specific mixed solvent system for crystallization ensures maximum recovery and purity.
Q: How does this method improve safety compared to traditional ether-based crystallization?
A: Traditional methods often rely on diethyl ether, which poses significant explosion hazards during large-scale operations. This optimized protocol utilizes safer C5-C6 ethers like methyl tert-butyl ether or isopropyl ether, drastically reducing flammability risks and enhancing operational safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Adefovir Dipivoxil Supplier
The technical superiority of the PTC-free synthesis route for Adefovir Dipivoxil highlights the importance of partnering with a manufacturer that possesses deep process chemistry expertise. NINGBO INNO PHARMCHEM stands at the forefront of this capability, bridging the gap between laboratory innovation and industrial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the highest international pharmacopeial standards.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized manufacturing protocols, we can help you secure a stable supply of high-quality intermediates while driving down your overall production costs. Contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced synthesis capabilities can strengthen your supply chain resilience.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
