Advanced Manufacturing of Roxadustat Key Intermediates for Global Pharmaceutical Supply Chains
Introduction to Next-Generation Roxadustat Manufacturing
The global demand for effective treatments for Chronic Kidney Disease (CKD) has placed Roxadustat at the forefront of pharmaceutical innovation. As a hypoxia-inducible factor prolyl hydroxylase (HIF-PH) inhibitor, its oral administration offers a significant advantage over traditional injectable erythropoietin therapies. However, the commercial viability of this life-saving drug hinges critically on the efficiency and cost-effectiveness of its supply chain, specifically the synthesis of its key heterocyclic intermediates. Patent CN115594631A introduces a groundbreaking synthetic methodology that addresses the longstanding bottlenecks in producing the critical 4-hydroxy-7-phenoxyisoquinoline scaffold. This technical insight report analyzes the novel six-step route starting from m-bromobenzaldehyde, highlighting its potential to redefine cost structures and supply reliability for global API manufacturers seeking a reliable pharmaceutical intermediate supplier.
The significance of this patent lies not merely in a new chemical transformation but in a holistic re-engineering of the production logic. Traditional pathways have been plagued by the use of exotic starting materials, dangerous reagents, and low-yielding steps that necessitate complex purification. By shifting the synthetic entry point to widely available commodity chemicals and employing robust catalytic systems, this new approach offers a pathway to substantial cost reduction in API manufacturing. For procurement leaders and R&D directors alike, understanding the mechanistic nuances and operational advantages of this route is essential for securing a competitive edge in the increasingly crowded anemia treatment market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthesis routes for Roxadustat intermediates, such as those disclosed in early FibroGen patents (e.g., WO2004108681A1), rely heavily on 3,4-dicyanonitrobenzene or 5-bromophthalide as starting materials. These precursors are not only expensive but often lack consistent commercial availability, creating immediate supply chain vulnerabilities. Furthermore, these legacy processes frequently employ hazardous reagents such as n-butyllithium for methylation steps, which poses severe safety risks and requires specialized cryogenic infrastructure, drastically increasing capital expenditure. The reliance on palladium-catalyzed coupling reactions in some alternative routes further exacerbates cost issues due to the high price of noble metals and the stringent requirements for residual metal removal to meet pharmacopeial standards.
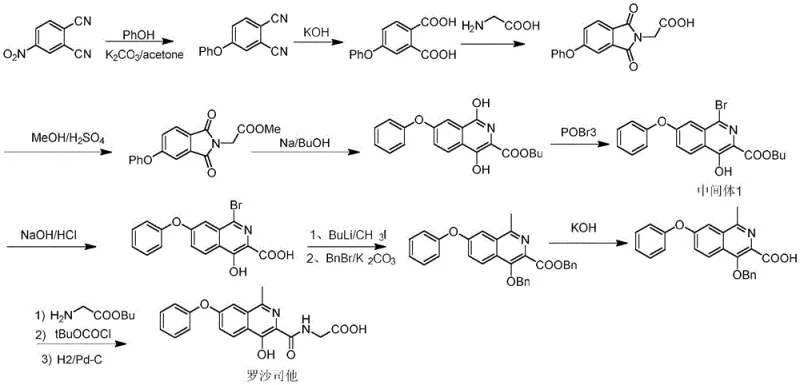
Beyond safety and cost, the purification challenges associated with conventional methods are profound. Many prior art routes generate complex impurity profiles that cannot be resolved through simple crystallization, necessitating column chromatography. In an industrial setting, chromatography is a major bottleneck that limits throughput and increases solvent consumption, negatively impacting both the environmental footprint and the overall production timeline. The cumulative effect of these factors—high raw material costs, dangerous operating conditions, and inefficient purification—renders many traditional routes economically unfeasible for large-scale commercial production, driving the urgent need for the innovative approach detailed in the subject patent.
The Novel Approach
The synthetic strategy outlined in patent CN115594631A represents a paradigm shift by utilizing m-bromobenzaldehyde and phenol as the foundational building blocks. This route elegantly constructs the isoquinoline core through a sequence of etherification, reduction, halogenation, substitution, cyclization, and aromatization. The brilliance of this design is its modularity and reliance on robust, well-understood chemical transformations that avoid the pitfalls of the prior art. By eliminating the need for precious metal catalysts and cryogenic reagents, the process inherently lowers the barrier to entry for manufacturing while enhancing operational safety.
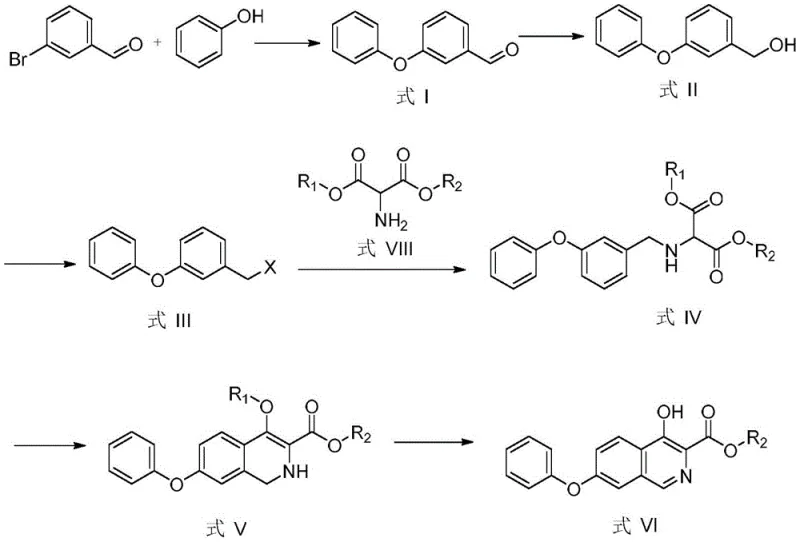
Crucially, this novel approach achieves high purity and yield without resorting to chromatographic purification. The intermediate compounds, particularly the key Formula IV and Formula V species, can be isolated and purified through standard workup procedures such as extraction and recrystallization. This simplification of downstream processing is a game-changer for commercial scale-up, as it allows for continuous or large-batch processing with minimal equipment downtime. The ability to produce high-purity intermediates (>99%) using such a streamlined workflow demonstrates a sophisticated understanding of process chemistry that directly translates to improved margins and supply chain resilience for pharmaceutical partners.
Mechanistic Insights into the Cyclization and Aromatization Strategy
The core of this synthetic innovation lies in the efficient construction of the isoquinoline ring system and the precise installation of the 4-hydroxy group. The cyclization step (Step S5) utilizes phosphorus oxychloride (POCl3) or phosphorus pentoxide (P2O5) in a high-boiling solvent like DMF or xylene. This conditions facilitate an intramolecular electrophilic substitution that closes the ring to form the dihydroisoquinoline structure (Formula V). The choice of cyclizing agent is critical; POCl3 acts both as a dehydrating agent and a chlorinating source if necessary, driving the equilibrium towards ring closure with high efficiency. The reaction temperature is carefully controlled between 150-160°C to ensure complete conversion while minimizing thermal degradation of the sensitive phenoxy moiety.
Following cyclization, the aromatization step (Step S6) is equally pivotal. The dihydroisoquinoline intermediate is subjected to acidic hydrolysis followed by oxidation using manganese dioxide (MnO2). This oxidative aromatization is highly selective, converting the 4-position into the required hydroxyl group while establishing the fully aromatic isoquinoline system (Formula VI). The use of MnO2 is particularly advantageous as it is a mild oxidant that avoids over-oxidation of other functional groups, ensuring the integrity of the ester side chain. This two-step sequence (cyclization followed by oxidative aromatization) effectively bypasses the need for difficult direct hydroxylation of the isoquinoline ring, a common challenge in heterocyclic chemistry that often leads to regioisomeric impurities.
How to Synthesize Roxadustat Intermediate Efficiently
The practical implementation of this synthesis involves six distinct operational stages, each optimized for yield and purity. The process begins with the copper-catalyzed etherification of m-bromobenzaldehyde, followed by a mild reduction to the alcohol and subsequent halogenation to activate the benzylic position. The resulting benzyl halide is then coupled with a protected aminomalonate derivative, setting the stage for the critical ring-closing event. The detailed standardized synthetic steps, including specific molar ratios, solvent choices, and temperature profiles for each stage, are provided in the guide below to ensure reproducibility and quality control.
- Etherification: React m-bromobenzaldehyde with phenol using a copper-based catalyst system at 150-190°C to form m-phenoxybenzaldehyde.
- Reduction & Halogenation: Reduce the aldehyde to benzyl alcohol using sodium borohydride, then convert to benzyl bromide using PBr3.
- Substitution: React the benzyl bromide with diethyl aminomalonate in acetonitrile with sodium carbonate to form the substituted malonate.
- Cyclization: Heat the substituted malonate with phosphorus oxychloride (POCl3) in DMF at 150-160°C to close the isoquinoline ring.
- Aromatization: Hydrolyze the intermediate with HCl and oxidize with manganese dioxide (MnO2) to achieve the final aromatic hydroxy-isoquinoline structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible strategic benefits beyond mere technical elegance. The primary advantage is the decoupling of production from volatile specialty chemical markets. By relying on m-bromobenzaldehyde and phenol, manufacturers can source raw materials from a broad base of suppliers, mitigating the risk of single-source dependency and price spikes. This diversification of the supply base enhances the overall reliability of the API supply chain, ensuring that production schedules remain uninterrupted even during global raw material shortages.
- Cost Reduction in Manufacturing: The elimination of expensive palladium catalysts and hazardous organolithium reagents results in significant direct cost savings. Furthermore, the avoidance of chromatographic purification reduces solvent consumption and waste disposal costs, while the high yields at each step minimize the loss of valuable intermediates. These factors combine to lower the overall cost of goods sold (COGS), allowing for more competitive pricing in the final drug market.
- Enhanced Supply Chain Reliability: The use of commodity starting materials ensures a stable and continuous supply of inputs. Unlike exotic precursors that may have long lead times or limited production capacity, the raw materials for this route are produced at scale by the global chemical industry. This availability translates to shorter lead times for the intermediate itself, enabling faster response to market demand fluctuations and more agile inventory management.
- Scalability and Environmental Compliance: The process is inherently green, avoiding heavy metals and toxic reagents that complicate regulatory compliance and waste treatment. The mild reaction conditions and standard solvent systems make the technology easily transferable from pilot plant to full commercial scale without the need for specialized high-pressure or cryogenic reactors. This ease of scale-up accelerates time-to-market and reduces the capital investment required for manufacturing infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on purity profiles, safety considerations, and scalability potential for potential manufacturing partners.
Q: What are the primary cost advantages of this new Roxadustat intermediate synthesis route?
A: The route utilizes readily available commodity chemicals like m-bromobenzaldehyde and phenol, avoiding expensive starting materials like 5-bromophthalide. It also eliminates the need for costly transition metal catalysts (like Palladium) and dangerous reagents (like n-butyllithium), significantly reducing raw material and safety compliance costs.
Q: How does this process improve purity and downstream processing compared to traditional methods?
A: Unlike prior art routes that require column chromatography for purification, this method relies on crystallization and standard extraction techniques. The specific reaction conditions minimize side reactions, resulting in high purity (over 99%) intermediates without the need for complex separation technologies.
Q: Is this synthetic route scalable for industrial production?
A: Yes, the process is designed for industrial scalability. It operates under mild reaction conditions, avoids high-pressure hydrogenation, and uses common solvents. The elimination of hazardous reagents and the robustness of the cyclization and aromatization steps make it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Roxadustat Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceuticals like Roxadustat depends on a partner who can bridge the gap between laboratory innovation and industrial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics seen in patent literature are realized in actual manufacturing campaigns. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the exacting standards required for API synthesis, providing you with a secure foundation for your drug product manufacturing.
We invite you to engage with our technical procurement team to discuss how this advanced synthetic route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this greener, more efficient methodology. We are prepared to provide specific COA data and route feasibility assessments tailored to your volume requirements, helping you secure a sustainable and cost-effective supply of high-purity Roxadustat intermediates for the global market.
