Advanced Green Synthesis of Budesonide: Technical Insights for Commercial Scale-Up and Procurement
The pharmaceutical industry is constantly seeking more sustainable and efficient pathways for the production of high-value corticosteroids, and the technology disclosed in patent CN101717428A represents a significant leap forward in this domain. This patent introduces a novel method for synthesizing Budesonide, a potent anti-inflammatory agent widely used in asthma and allergy treatments, by utilizing acidic ionic liquids as a dual-function medium and catalyst. Unlike traditional methods that rely on corrosive mineral acids and volatile organic solvents, this approach leverages the unique physicochemical properties of ionic liquids to create a greener, more controllable reaction environment. For R&D directors and process chemists, this technology offers a compelling alternative that addresses critical pain points regarding waste management, catalyst recovery, and product purity. By shifting the paradigm from homogeneous acid catalysis to a tunable ionic liquid system, manufacturers can achieve superior control over the acetalization reaction, ensuring consistent quality while adhering to increasingly stringent environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Budesonide via the acetalization of 16α-hydroxyprednisolone with n-butyraldehyde has relied heavily on traditional Brønsted acids such as sulfuric acid or p-toluenesulfonic acid dissolved in volatile organic solvents. These conventional processes suffer from inherent drawbacks that complicate large-scale manufacturing and increase operational costs. The use of strong mineral acids often leads to severe equipment corrosion, necessitating expensive specialized reactors and frequent maintenance. Furthermore, the separation of the acid catalyst from the final product typically requires energy-intensive neutralization steps followed by extensive washing, which generates substantial amounts of saline wastewater. This not only burdens the facility's waste treatment infrastructure but also poses risks of product degradation due to exposure to harsh pH conditions during workup. Additionally, the volatility of traditional organic solvents contributes to VOC emissions, creating safety hazards and environmental compliance challenges that modern pharmaceutical facilities strive to eliminate.
The Novel Approach
In stark contrast, the methodology outlined in the patent utilizes acidic ionic liquids, specifically imidazolium-based salts functionalized with sulfonic acid or carboxyl groups, to drive the reaction forward with remarkable efficiency. This innovative approach transforms the reaction medium itself into the catalyst, eliminating the need for additional acid additives and volatile solvents. The ionic liquid provides a stable, non-volatile environment that enhances the solubility of the steroid substrate while facilitating the protonation of the aldehyde carbonyl group. As illustrated in the reaction scheme below, the process converts 16α-hydroxyprednisolone (I) and n-butyraldehyde directly into Budesonide (III) under mild thermal conditions.
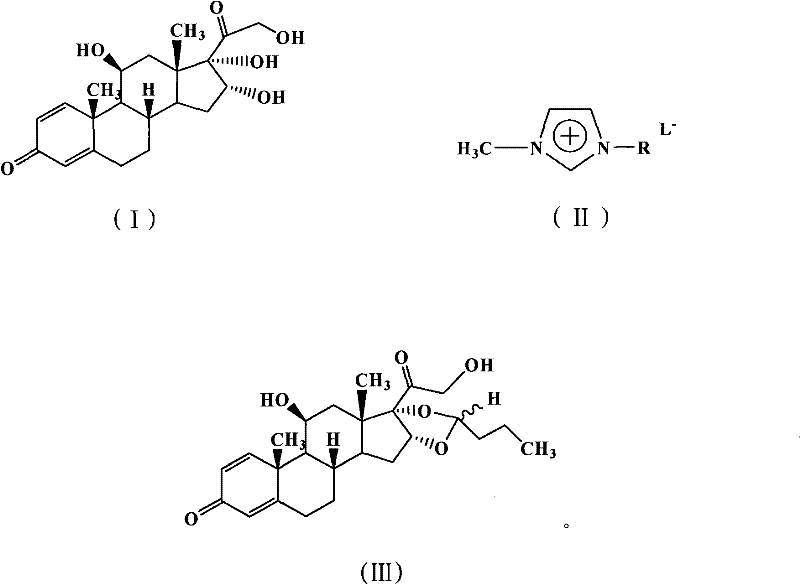
The ability to tune the acidity of the ionic liquid by selecting different anions, such as HSO4-, BF4-, or CF3SO3-, allows for precise optimization of reaction kinetics without compromising selectivity. Moreover, the negligible vapor pressure of ionic liquids drastically reduces solvent loss and emission risks, while their thermal stability ensures safe operation even at elevated temperatures. Perhaps most critically for supply chain sustainability, the ionic liquid can be recovered and reused multiple times without significant loss of catalytic activity, fundamentally altering the economics of the synthesis by reducing raw material consumption.
Mechanistic Insights into Acidic Ionic Liquid Catalyzed Acetalization
To fully appreciate the technical superiority of this method, one must understand the mechanistic role played by the acidic ionic liquid in facilitating the formation of the cyclic acetal structure at the C16 and C17 positions of the steroid backbone. The reaction proceeds through a classic acid-catalyzed mechanism where the acidic protons provided by the functionalized cation or the acidic anion of the ionic liquid protonate the carbonyl oxygen of the n-butyraldehyde. This protonation significantly increases the electrophilicity of the carbonyl carbon, making it more susceptible to nucleophilic attack by the 16α-hydroxyl group of the prednisolone derivative. The unique solvation environment of the ionic liquid stabilizes the charged intermediates and transition states involved in this process, lowering the activation energy barrier compared to non-polar organic solvents. Furthermore, the hydrogen bonding network within the ionic liquid matrix may assist in orienting the reactants favorably, thereby enhancing the stereochemical outcome and minimizing the formation of unwanted byproducts.
Impurity control is another critical aspect where this technology excels, particularly concerning the suppression of side reactions that commonly plague steroid chemistry. In traditional acidic media, steroids are prone to dehydration, isomerization, or oxidative degradation due to the harsh conditions. However, the mild yet effective acidity of the ionic liquid system allows the reaction to proceed at moderate temperatures, typically between 50°C and 80°C, which preserves the integrity of the sensitive diene-one chromophore in the A-ring of the steroid. The patent data indicates that conversion rates reach 100% with yields consistently above 80%, suggesting that the ionic liquid effectively suppresses competing pathways. The ease of separation—simply extracting the product with dichloromethane while leaving the ionic liquid behind—further ensures that no residual acid catalyst contaminates the final API intermediate, simplifying downstream purification and ensuring high-purity specifications are met without extensive chromatography.
How to Synthesize Budesonide Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the stoichiometry and physical parameters defined in the patent to maximize efficiency. The process begins by charging the reactor with the steroid substrate and the aldehyde in a specific molar ratio, followed by the addition of the ionic liquid which serves as the reaction medium. The mixture is then heated to the optimal temperature range to initiate the acetalization. While the general concept is straightforward, the devil is in the details regarding the specific choice of ionic liquid anion and the precise control of reaction time to prevent over-reaction or degradation. For a comprehensive understanding of the operational parameters, the detailed standardized synthesis steps are provided in the guide below.
- Charge the reactor with 16α-hydroxyprednisolone and n-butyraldehyde in a molar ratio of 1: 2 to 1:5, along with the acidic ionic liquid catalyst.
- Maintain the reaction mixture at a temperature between 50°C and 80°C for a duration of 3 to 5 hours to ensure complete conversion.
- Perform post-treatment by extracting with dichloromethane, washing with distilled water, drying over anhydrous magnesium sulfate, and evaporating the solvent.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this ionic liquid-based technology translates into tangible strategic advantages that go beyond mere technical novelty. The primary value proposition lies in the drastic simplification of the downstream processing workflow. By eliminating the need for neutralization agents and the associated salt waste streams, the facility can significantly reduce its environmental footprint and the costs associated with wastewater treatment and disposal. The reusability of the ionic liquid catalyst is another major economic driver; since the catalyst does not get consumed in the reaction and can be separated easily from the organic product phase, the recurring cost of catalyst purchase is minimized over the lifecycle of the production campaign. This creates a more predictable cost structure and reduces dependency on fluctuating markets for traditional acid reagents.
- Cost Reduction in Manufacturing: The elimination of volatile organic solvents and the reduction in water usage for washing steps lead to substantial operational expenditure savings. Traditional processes often require large volumes of solvents for extraction and crystallization, which incur high procurement and recovery costs. In this novel process, the ionic liquid acts as the solvent, and since it is non-volatile, losses due to evaporation are virtually non-existent. Furthermore, the absence of corrosive mineral acids extends the lifespan of reactor vessels and piping, reducing capital expenditure on maintenance and equipment replacement. The overall process intensification means that more product can be produced per unit volume of reactor space, improving asset utilization rates.
- Enhanced Supply Chain Reliability: Sourcing high-purity reagents with consistent quality is a perennial challenge in pharmaceutical supply chains. This method relies on robust, chemically stable ionic liquids that have a long shelf life and are less sensitive to moisture or handling errors compared to sensitive Lewis acid catalysts. The simplified workup procedure reduces the number of unit operations required, thereby shortening the overall manufacturing cycle time. A shorter cycle time implies faster turnaround for batch production, allowing the supply chain to respond more agilely to market demand fluctuations. Additionally, the reduced generation of hazardous waste simplifies logistics and compliance reporting, removing potential bottlenecks related to waste disposal permits and transportation.
- Scalability and Environmental Compliance: As regulatory bodies worldwide tighten restrictions on VOC emissions and hazardous waste discharge, this technology positions manufacturers ahead of the compliance curve. The non-volatile nature of the ionic liquid ensures that workplace exposure limits are easier to maintain, enhancing operator safety. From a scalability perspective, the exothermic nature of the acetalization reaction is easier to manage in an ionic liquid medium due to its high heat capacity, reducing the risk of thermal runaway during scale-up from kilogram to tonne scales. This thermal safety margin is crucial for obtaining regulatory approval for commercial manufacturing sites and ensures continuous, uninterrupted supply of this critical respiratory medicine intermediate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ionic liquid catalyzed synthesis. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into existing production lines or for assessing the quality of suppliers who utilize this advanced methodology.
Q: What are the advantages of using acidic ionic liquids over traditional mineral acids in Budesonide synthesis?
A: Acidic ionic liquids offer tunable acidity, negligible vapor pressure, and thermal stability. Unlike traditional mineral acids, they act as both solvent and catalyst, simplifying separation and allowing for catalyst recycling, which significantly reduces waste generation and corrosion issues.
Q: What is the typical yield and conversion rate for this ionic liquid catalyzed process?
A: According to the experimental data in patent CN101717428A, the process achieves a raw material conversion rate of 100% with isolated yields ranging from 80% to 95%, depending on the specific ionic liquid anion and reaction conditions employed.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the method is designed for scalability. The mild reaction conditions (50-80°C), the reusability of the ionic liquid catalyst, and the straightforward workup procedure involving simple extraction make it highly suitable for commercial scale-up of complex steroid intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Budesonide Supplier
The technological advancements described in patent CN101717428A highlight the evolving landscape of steroid synthesis, where green chemistry principles are becoming synonymous with commercial viability. At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting such innovative pathways to deliver high-quality intermediates to the global market. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of materials. Our commitment to excellence is underpinned by stringent purity specifications and rigorous QC labs that validate every batch against the highest industry standards, guaranteeing that the complex stereochemistry of molecules like Budesonide is preserved throughout the manufacturing process.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage these advanced synthetic capabilities for your next project. Whether you are looking to optimize an existing route or develop a new process from scratch, our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and profitability in your supply chain.
